Abstract
BACKGROUND AND PURPOSE:When using detachable coils to treat intracranial aneurysms, thromboembolism is the most feared and frequently reported complication during or after endovascular therapy. The purpose of this study was to document the therapeutic effect of tirofiban on patency of the parent vessel, rate of rebleedings, and outcome of the patients in the setting of acute subarachnoidal hemorrhage.
METHODS:A patient data base was retrospectively reviewed to identify patients in whom thrombus occurred during endovascular treatment of ruptured cerebral aneurysms within a 34-month period and who were treated with tirofiban. All patients underwent anticoagulation with heparin during endovascular treatment procedures. Sixteen patients (age range, 52.9 ± 10.7 years; 10 women, 6 men) were identified with intraprocedural thrombus formation. The patency of the parent vessel was assessed in a retrospective analysis blinded to outcome. Eight patients received ventriculostomy and had a follow-up CT.
RESULTS: Local nonocclusive thrombus at the coil surface was detected in 5 patients, in all of whom the thrombus was dissolved. In 10 patients, partial or total occlusion of the parent vessel occurred during the intervention; in 8 of these, the vessel was recanalized completely and in 2 drug administration was assisted by mechanical means. In 1 patient, however, the occlusion persisted. No periprocedural rebleedings of the ruptured aneurysm occurred; 3 of 8 ventriculostomies had clinically silent small local bleedings.
CONCLUSION: The use of tirofiban in the setting of endovascular treatment of ruptured intracranial aneurysms to dissolve platelet aggregation seems relatively safe and effective.
Thromboembolism is the most frequently reported complication during or after an endovascular therapy with detachable coils for the treatment of ruptured intracranial aneurysms.1-4 The reported incidence of thrombus formation at the site of the interface of coil and the parent vessel is on the order of 7%, based on retrospective analysis.5 In part, these thrombi lead to permanent deficits as a result of thromboembolism.6
The potential sources of emboli vary. A pre-existing intra-aneurysmal thrombus may be displaced during coil placement, or a new thrombus may form on the coil surface. A temporary stenosis of an adjacent artery as a result of coil herniation may also trigger thrombus formation. Last but not least, thrombus may form on the catheters during the procedure. Early detection of thrombus formation at the coil-parent artery interface is very important because this is the prerequisite for early intervention with probably good outcome.
The current regimen of endovascular management in acute thrombus formation during coil placement includes the increase of intravenous (IV) heparin (measured by the activated clotting time [ACT]). Because, however, this strategy does not achieve the desired optimal results, various centers have begun with the periprocedural application of antiplatelet drugs, such as intravenous aspirin. Occlusions are sometimes treated by intraarterial thrombolysis with urokinase or recombinant tissue plasminogen activator, often causing severe rebleeding at the aneurysm site and elsewhere. The benefit of controlling the thromboembolism by local or systemic thrombolytic medication must be balanced against the increased risk of intracranial hemorrhage. A prerequisite for such a treatment option is easy application and short half-life of the drug. The glycoprotein IIb/IIIa antagonist tirofiban seems to fulfill these requirements, at least in part.7
Therefore, we retrospectively analyzed all recent endovascular treatments and searched for thrombus formation as a complication during the procedure. The results as to (1) patency of the parent vessel, (2) any intracranial rebleeding, and (3) outcome of the patients after the intravenous administration of tirofiban were then analyzed.
Materials and Methods
Patients
A 34-month section of our data base, from January 2002 through October 2004, was retrospectively analyzed for patients with a symptomatic intracranial aneurysm who had been treated with coil embolization at our institution (225 cases). All patients were identified by the keywords thrombus or clot. Patients who had received tirofiban for reasons other than primary thrombus formation at the coil-vessel interface (eg, for an intracranial stent) were excluded. Patients with (1) a subarachnoid hemorrhage more than 7 days before the intervention, (2) nonsymptomatic incidental aneurysm findings, or (3) recurrent disease were also excluded, as were patients with incomplete records (n = 2).
The 16 patients comprised 10 women and 6 men with a median age of 52.9 ± 10.7 years (range, 36–78 years). All had acutely symptomatic ruptured aneurysms ranging in size from 3 to 18 mm, and all had had acute thrombus formation during the coil-placement procedure (Table 1).
Summary of anatomic and clinical data
The digital subtraction angiographic (DSA) images, the discharge letters, and the peri-interventional documentation were reviewed. The DSA images were retrospectively analyzed for the location, size of dome, and neck size of the aneurysm; the procedure charts were reviewed to determine the type of coil, medication, dose, route, and for the ACT recorded during the intervention. Finally, the discharge letters were analyzed to ascertain the outcome.
Aneurysm Location
The location, the size of the neck, and the dome of each aneurysm are given in detail in Table 1. The parent artery given in the Table is the artery that is contiguous with the opening of the aneurysm sac. The coil-parent artery interface is the surface of the coil pack, which interacts with inflowing blood and is located at the aneurysm neck in a completely coiled aneurysm.
Interventional Procedure
All patients were treated under general anesthesia. Three operators performed the endovascular coil treatment procedures using similar techniques. A 6F guiding catheter (Envoy; Cordis Neurovascular, Miami Lakes, Fla) was placed in either of the internal carotid arteries or the vertebral arteries, and the aneurysm sac was accessed with an appropriate microcatheter (eg, Excelsior SL 10; Boston Scientific, Natick, Mass). The technique for Guglielmi detachable coil (GDC) therapy (Boston Scientific) has been described previously6 and was used in 13 patients. Three aneurysms were packed with hydraulically detachable coils (Cordis Neurovascular). If a ventricular drainage was required, it was placed before the endovascular therapy in 8 patients (based on the degree of hydrocephalus in the CT scan and the clinical status before the intervention).
All patients received IV-administered heparin after placement of a vascular sheath in the common femoral artery with a targeted ACT of 250. Systemic heparin was delayed until the guiding catheter was successfully placed. In general, patients then received an intravenous bolus of heparin of approximately 2000–3000 IU. ACT was measured in seconds before and during the procedure by using a Hemochron Junior ACT machine (KRAUTH Medical KG Cardiovascular, Hamburg, Germany). In addition, pressurized flushing of the arterial sheath and guide catheter was carried out with a sodium chloride solution including 1000–2000 U of heparin per liter. Intravenous heparin infusion was continued after the intervention for 24–48 hours until a target partial thromboplastin time of 50–70 seconds was reached.
Aspirin (500 mg) was given to 11 of 16 patients intraoperatively at the operator’s discretion before administering tirofiban. Because of the selection criteria of the retrospective analysis, only patients who subsequently received tirofiban were included in the analysis.
Tirofiban was then given intravenously in a standardized scheme that is recommended for myocardial infarctions.8,9 The loading dose of 0.4 μg/kg/min was administered for 30 minutes, then the infusion was reduced to 0.1 μg/kg/min and maintained at least until the thrombus dissolved. On average, tirofiban was given intravenously for a duration of 31.1 ± 70.3 hours.
The administration of tirofiban was assisted in 2 patients by mechanical means. When no spontaneous recanalization occurred in patient 1, mechanical recanalization was attempted by using a X-pedion Microwire (MicroTherapeutics, Irvine, Calif) and an Excelsior SL 10 microcatheter and subsequently a Rebar 27 microcatheter (MicroTherapeutics). The pericallosal artery was recanalized approximately 50 minutes later. In patient 14, the protruding coil package was compressed with the help of a HyperGlide balloon (MicroTherapeutics).
For follow-up, the modified Rankin scale (mRS) was assessed within 2 months after the intervention by using the last available suitable documentation (eg, discharge letter).
Results
The 16 patients with acute subarachnoid hemorrhage (mean Fisher grade, 2.7 ± 0.9) had an initial median World Federation of Neurological Surgeons (WFNS) score of 2.5 ± 1.8 (range, 1–5), and the outcome as measured by the mRS was on average 2.4 (median, 1.0 ± 2.6; range, 0–6). The aneurysms were located in the anterior cerebral artery in 7 cases (of these, 6 were in the anterior communicating branch and 1 in the pericallosal area). In the 9 remaining patients, there were 2 aneurysms of the internal carotid artery, 3 of the posterior communicating artery, 1 of the middle cerebral artery, and 3 of the posterior circulation (2 basilar tip aneurysms and 1 posterior cerebral artery aneurysm).
There was an 88% patency rate, 6% partial recanalization, and 1 vessel remained occluded. Only local thrombus was detected at the coil surface in 5 patients, in all of whom the thrombus was locally monitored; these thrombi dissolved without thromboembolic complications. In 8 patients, there was already a partial or total occlusion of the parent vessel, but the combination of tirofiban and heparin dissolved the thrombus completely under angiographic control, and the vessel size normalized. In 2 patients, the administration of tirofiban and heparin did not result in reopening of the vessel within 30 minutes, and the drug administration was assisted by mechanical means. In 1 patient, however, the M2 parent vessels did not become patent.
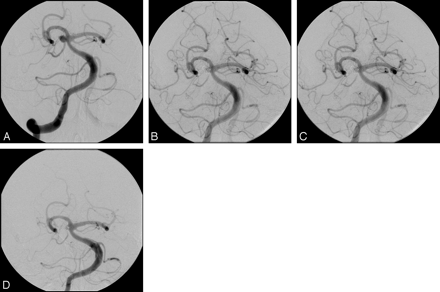
The outcome was excellent in the 6 patients with an initial WFNS 1 in all patients (mRS of 0). An example of a small local thrombosis with subsequent occlusion of the parent vessel in a patient of this group is shown in Fig 1 (patient 6).
This 49-year-old female patient was admitted to the hospital in good clinical condition (WFNS 1). A 5 × 4-mm aneurysm of the basilar tip involving the right superior cerebellar artery (SCA) was found on diagnostic angiography (A). Despite care taken not to compromise the ostium of the SCA with the coil package, the SCA was occluded during the coiling procedure (B). After administration of tirofiban, the vessel reopened within 15 minutes (C). Follow-up angiography 4 months later confirmed patency of the SCA, and the aneurysm remained occluded (D).
Of the 4 patients with moderate WFNS grades of 2 or 3, the outcome was excellent in 2 patients, moderate in 1 patient (mRS 2), and severe in the last (mRS 5). Fig 2 (patient 8) provides an example of a good outcome despite local thrombosis. The patient with the severe outcome (mRS 4) had a large aneurysm of the anterior communicating artery. During the attempt to place the fifth coil into the aneurysm sac, the coil fractured and a free fragment remained in the right A1 segment. Despite immediate rescue maneuvers, thrombosis occurred. At the time tirofiban administration was initiated, the thrombus extended into the left A1 segment and the communicating branch. After tirofiban, both anterior cerebral arteries were recanalized. The patient awoke with a persistent right hemiparesis but after a prolonged recovery was able to get up with assistance.
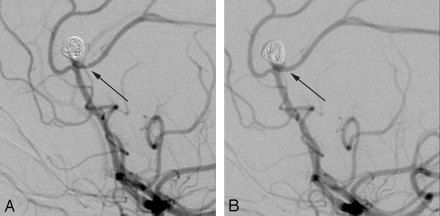
Local thrombosis controlled by tirofiban. This 56-year-old female patient presented with a subarachnoidal hemorrhage and focal deficits (WFNS 3). Multiple aneurysms were found on diagnostic angiography; however, the pericallosal aneurysm was determined to be the symptomatic one. During the interventional procedure, a local thrombus was detected in the left pericallosal artery (arrow, A). Tirofiban was administered and patency of the vessel was restored (B). Outcome of the patient was excellent; there were no focal or generalized deficits (mRS 0).
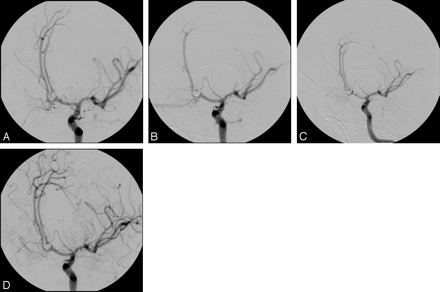
Six patients had a pretreatment WFNS grade of 5; 2 died and 4 survived with an mRS of 4–5. The 2 patients who died had increased intracranial pressure despite intensified therapy, and 1 of them also had a subsequent myocardial infarction. One of these patients (1, who initially had a WFNS of 1, had secondary bleeding on arrival at the hospital and worsened to a comatose state [WFNS 5]) had had a bi-lobar aneurysm of the anterior communicating artery. The aneurysm was treated with 3D GDC coils, and with the use of a balloon remodelling technique, a good angiographic result was achieved. However, a small thrombus was seen at the coil-vessel interface. The heparin dose was elevated, and aspirin (500 mg) was given intravenously. In the subsequent monitoring series, the left pericallosal artery was occluded. Tirofiban was given, and the thrombus was observed for 30 minutes. Because spontaneous recanalization did not occur, the pericallosal artery was mechanically recanalized approximately 50 minutes later (see the “Materials and Methods” section for details) (Fig 3). The patient developed a right hemiparesis that improved during subsequent rehabilitation.
Extensive thrombosis and thromboembolism in part controlled by tirofiban. This 64-year-old male patient (10) presented with an extensive subarachnoidal hemorrhage (Fisher IV) and suffered a severe rebleeding during transfer from an outside hospital (WFNS V). The bilobar aneurysm of the anterior communicating branch had broad-based contact to the parent vessel (A). Because of the poor clinical status, a surgical approach was excluded. During the interventional procedure, an occlusion of the left pericallosal artery was detected (B). Tirofiban and aspirin were administered; however, patency of the vessel was not restored after 30 minutes. After mechanical assistance with the use of a microwire and various microcatheters (C), the pericallosal artery was partially recanalized (D). However, the patient had focal deficits (mRS 4).
Thrombus developed at the coil-parent artery interface in all patients despite administration of therapeutic doses of heparin. Aspirin was given at the operator’s discretion in 11 of 16 patients intraoperatively before the use of tirofiban (Table 1). In all of these cases, the administered aspirin solution did not prevent the detected thrombus from growing.
Three small (<1 cm) subclinical intracranial bleedings along the puncture track were detected by follow-up CT in the 8 patients who received shunt surgery ventriculostomy. Extracranial bleeding such as retroperitoneal or gastrointestinal were not detected during the intervention, the immediate peri-interventional follow-up, or the later course of hospitalization.
Discussion
The data presented here describe a case collection in which thrombus formation was detected at the coil-vessel interface during endovascular treatment of intracranial cerebral aneurysms. In all of these cases, the generally accepted therapy of heparin alone failed to hinder thrombus formation. Therefore, the operators decided individually to use a glycoprotein IIb-IIIa drug, that was considered necessary to prevent further thrombus formation and embolism. Our retrospective data thus document an alternative approach to the dilemma of how to treat a thromboembolic complication during endovascular treatment of a ruptured intracranial aneurysm. As a result of intravenous tirofiban, all of the parent vessels improved, and the vessels were completely recanalized in all but 2 patients. In other words, angiographically detectable thrombi were successfully treated with intravenous tirofiban, resulting in 88% angiographic resolution of the thrombi. There were no major hemorrhagic complications.
These results are interesting in the light of previously published findings. In the series of Cronqvist et al10 for example, 19 patients received urokinase for thrombolysis. Nine achieved complete recanalization (47%) and 5 achieved partial recanalization (26%). Fourteen patients had a good clinical recovery. One patient was moderately disabled and 2 were severely disabled. Two patients died, one as a consequence of the pre-existing subarachnoid hemorrhage and the other because of a large intracerebral hematoma that developed after fibrinolysis.
The use of other glycoprotein IIb-IIIa inhibitors such as abciximab (ReoPro) was also described earlier. Workman et al5 used abciximab in 4 patients with unruptured aneurysms when thrombus was forming at the coil-parent artery interface and in 2 patients with a small residual thrombus. However, they did not use abciximab in combination with heparin in patients with ruptured aneurysms. Song et al11 used abciximab intra-arterially in the same situation for 4 ruptured and 3 unruptured aneurysms; recanalization was complete in 5 of 7 patients, incomplete in 1 patient, and not recanalized in 1 patient. Other reports have been published on the use of abciximab as an adjunct to standard heparin in the same setting—ie, the treatment dilemma of detecting a thromboembolic complication in an attempt to treat an intracranial aneurysm.12,13 Some reported extravasation of contrast material during the procedure and while giving the drug intra-arterially,11 whereas others did not report abciximab-related bleeding complications.5,14
Tirofiban hydrochloride monohydrate is a nonpeptide tyrosine derivative that acts as a reversible antagonist of fibrinogen binding to the glycoprotein IIb/IIIa receptor of platelets and thus highly effectively inhibits platelet aggregation after a 30-minute loading dose. Platelet function is known to almost normalize within approximately 4 hours after discontinuation,15 and the plasma half-life of tirofiban is 2 hours.16,17 Its known half-life is thus considerably shorter than that of abciximab, which has a reported time interval of up to 48 hours to wear off.5,15 The short half-life of tirofiban is believed to be advantageous if, for example, a ventriculostomy or another revision procedure becomes necessary.
The outcome of the patients was also influenced by various other factors. An analysis of the cofactors, such as the grade of bleeding rated with the Fisher score, the presence or absence of vasospasm or intracranial hypertension, and general factors such as age and sex, however, did not reveal clear trends for the 2 main parameters, patency of the parent vessel and outcome mRS.
Close observation of the access route and the aneurysm site was performed to detect thrombus formation as soon as possible to allow early medical management and avert distal thromboembolism. However, thrombus at the coil-parent artery interface can be subtle and is probably generally underdetected. Although the angiographic sensitivity for the detection of thrombus is not known, it may be much less than 100%, probably depending on the technique used and the size of the thrombus. A retrospective analysis in a cardiologic series has shown that even relatively large thrombi can be missed during the procedure.18 Thus, thrombi may be present at the coil-vessel interface and remain undetected despite a suitable angiographic technique. We therefore performed frequent monitoring angiographies and closely observed the coil-parent artery interface to ensure earliest possible recognition.
The most likely reasons for thrombus formation during the coil treatment process are flow disturbance and flow stagnation within the aneurysm,5 as well as procedure-related injury of the endothelium. It has also been suggested that the process of thrombus formation at the anode (coil) during electrolysis in aneurysms and during GDC detachment plays a role.19 However, other groups questioned the concept of electrothrombosis.20 In our series, most of the coils could be detached electrically. We observed no clear trend toward a higher incidence of thromboembolism in our series in coils detached hydrostatically or electrically.
A higher incidence of thromboembolism had been reported for aneurysms with a wide neck.1,6 In our series, the dome/neck ratio was, on average, 2.0 (range, 1.3 to 3.0). In our institution, we also consider patients with wide-neck aneurysms to be candidates for procedures with elevated risk; however, our data show that the patients with a relatively poor outcome (Table 1) had neck ratios between 1.7 and 2.0 and therefore were not representative of the cases with the poorest neck definition.
Alternative treatment decisions were also considered, such as the removal of the thrombogenic coil material. However, if the thrombus is detected before coil detachment, it is probably not advisable to remove the coil, in that this would risk shearing the thrombus from the coil surface or out of the aneurysm sac, resulting in distal embolization of the thrombus. Therefore, it seems advantageous to leave the coil in place and “hold” the thrombus to the coil while attempting to dissolve the thrombus formation.
Mechanical devices have been used in 2 selected cases. Special care was taken to avoid movement of the coils in these specific situations. In one instance, both pericallosal arteries of patient 1 were not recanalized 30 minutes after the tirofiban infusion. Therefore, the thrombosed vessel was carefully catheterized by a flexible microwire and microcatheter to assist the drug-related effect. In the other case, coils of a large aneurysm of the basilar tip induced a relative compression of a P1 segment (patient 14). Here, a HyperGlide balloon was used to carefully dilate a seemingly compressed segment and fragment the thrombotic material to assist the drug effect (see “Materials and Methods”).
The limitations of the study are (1) the retrospective nature of the reported observations, and (2) the small number of patients included. The durations of the administration of the tirofiban infusion were not yet systematically evaluated; there are no data on long-term administration. Further studies are necessary here before we can give definite treatment recommendations. These studies might be useful to address a comparison of abciximab and tirofiban, and to address dose, route, and length of medication.
In light of our results, however, the intravenous administration of tirofiban, even in off-label use, seems to provide several advantages, such as the short half-life, which makes the drug easy to monitor (and to stop, when patient handling necessitates another procedure, for example, to deliver or change a ventriculostomy), the “controlled” venous administration, and its relative safety regarding the low rebleeding rate.
Conclusion
In conclusion, the prevention of platelet aggregation with the use of tirofiban in the setting of endovascular treatment of ruptured intracranial aneurysms seems to be a safe and efficient therapy that facilitates a high rate of recanalization in thromboembolic events while posing relatively little risk concerning bleeding complications.
Acknowledgments
We thank Mrs. J. Benson for editing the manuscript.
References
- Received July 20, 2005.
- Accepted after revision October 17, 2005.
- Copyright © American Society of Neuroradiology