We read with considerable interest the article by Schaefer et al regarding dynamic CT perfusion (CTP) in evaluation of the salvageable ischemic penumbra.1 The authors of that study, as well as other recent studies,1–3 refer to the usage of various thresholds and ratios relative to the contralateral side, obtained via dynamic CTP, to evaluate acutely which lesions are likely to progress to infarction in comparison to those that are potentially reversible ischemic lesions. The authors suggest that below a threshold for the cerebral blood volume (CBV) of 2.2 mL/100 g, a threshold for the cerebral blood flow (CBF) of 12.7 mL/100 g/min, a CBV ratio to the opposite side of <0.68, or CBF ratio of <0.32 leads to irreversible infarction. They state that no patients in whom CBV and/or CBF dropped below these thresholds had normal values and findings seen on follow-up examinations.
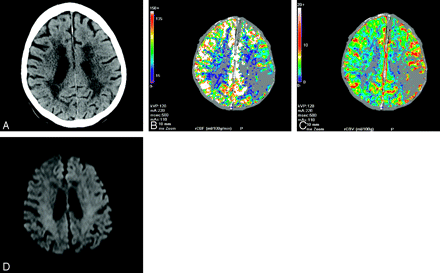
We respectfully submit that, despite the experience of these investigators, we have in fact encountered patients whose CTP studies clearly demonstrate focally or regionally absent CBF or CBV—ie, nearly 0—but have a normal appearance on diffusion-weighted imaging (DWI) performed in the acute phase. One such patient is illustrated here, a 75-year-old man presenting with acute onset of right upper extremity weakness, right facial weakness, and aphasia. Unenhanced CT, dynamic CTP, and CT angiography of the brain were performed as part of a 3-step protocol immediately in the same sitting within 2 hours of the onset of symptoms. Neither hemorrhage nor significant low attenuation was noted on the unenhanced CT scan (Fig 1A). The dynamic CTP examination was performed with a multisection scanner during 2 sequential 40-second dynamic scans (5-minute delay between the 2). This demonstrated a moderate-sized perfusion defect with no detectable CBV in the left posterior frontal and precentral and postcentral regions, consistent with the patient’s symptoms (Fig 1B, CBF images; Fig 1C, CBV). CT angiography (not shown) demonstrated a possible small M3 branch occlusion but no evidence of internal carotid or M1 branch occlusion. Immediately after completion of the stroke protocol and interpretation of the images, intravenous tissue plasminogen activator (tPA) was administered in the emergency department. It was interesting that the patient’s symptoms resolved within 2–3 minutes of administration of the tPA bolus. Follow-up MR images obtained just less than 24 hours after onset of symptoms demonstrated the typical findings of chronic small vessel ischemic disease on fluid-attenuated inversion recovery (FLAIR) imaging, with no abnormality on DWI imaging (Fig 1D) in the region of the CBV/CBF abnormality seen on CT perfusion. A tiny, punctuate, potential lesion on DWI (not shown) was noted in the contralateral, right parietal lobe, too small to visualize on the ADC maps, and presumed to be a tiny infarct superimposed on a large amount of leukoariosis. The patient’s neurologic status remained baseline (ie, normal) without neurologic sequelae on sequential clinical visits.
Although the etiology is unclear, but presumed to be thromboembolic, this may be related to a complete block of perfusion with no detectable circulating blood volume in the region of abnormality but not long enough to result in infarction. Artifacts can also simulate this abnormality, but in this case no such artifact or motion was noted. Moreover, re-evaluation of the CTP study confirmed that it was performed appropriately and that the contrast bolus appeared adequate. We note that an area of gray depicts an unmeasurable number on our software (Vitrea, Vital Images, Plymouth, Minn), either 0 or infinity (ie, either 0 CBF/CBV or infinity), whereas it seems there may be much variability among different software related to the color scheme used by the software implemented.1–6
An earlier article, by Wintermark et al, described false-positive cases of CT perfusion without abnormalities on follow-up imaging4 but stated that these cases had an element of ischemia with regard to CBF, with elevated mean transit time, but actually had a slightly elevated CBV (in contrast to our case), and symptom resolution was considered to be consistent with transient ischemic attacks. In our case, surrounding the complete (or near-complete) perfusion block and near-zero CBV was an area of mildly elevated CBV (Fig 1).
The significance of this observation is that, although the article by Schaefer et al illustrates the utility of CBV and CBF and ratios of these parameters in evaluating the extent of what is likely to infarct, the findings should always be carefully placed in perspective when affecting emergent clinical decision making, in particular with regard to administration of lytic therapy. This study and other recent studies comparing CT perfusion with MR DWI or MR perfusion2,3,5 are well designed but based on relatively small numbers (<20 with initial CTP and follow-up MR imaging) in light of the overall incidence of acute stroke. Another series by Mayer et al6 does postulate that this circumstance may occur, where flow rates less than approximately 5 mL/100 g/min might not be discerned from truly zero-flow states in CT-CBF maps, because the noise of the time-attenuation curve may obscure a very low level of flow. Presumably in our case a very minimal amount of CBF and very low CBV was present, but not detectable. Mayer et al urge less emphasis on exact thresholds, stating that these measurements are a marker of a point in time and do not reflect the state of perfusion before and after the perfusion examination. Those authors describe that a decrease of CBV down to 0 is likely not because of true lack of blood-containing capillary attenuation in that region, but rather a failure of contrast enhancement in the region of no detectable flow. Hence, the severity of the CBV defect may not be as relevant as the temporal extension and delay before the tissue is reperfused.6,7 Therefore, it is plausible that the rare case of a complete discrepancy between CT perfusion CBV and MR DW may exist, particularly if a hyperacute defect is immediately alleviated, whether spontaneously or from lytic therapy. Hence, in light of the occasional “near-zero-flow” situation described here, CBF or CBV values below the threshold should not necessarily drive whether to administer tPA. In a case of a moderate-sized or smaller “complete” perfusion deficit (in other words, no detectable cerebral perfusion or blood volume), intravenous tPA may still be a primary consideration as long as there is no evidence of hemorrhage or significant low attenuation is present on the noncontrast CT scan, and symptoms are clearly within the appropriate time window for intravenous tPA. These patients may occasionally completely reperfuse, with dramatic improvement, and no significant longstanding neurologic sequelae.
A 75-year-old man underwent a 3-part stroke protocol within 2 hours of the onset of aphasia and right-sided facial and upper extremity weakness. Typical unenhanced CT (A) findings of chronic small vessel ischemic disease revealed neither hemorrhage nor typical findings of acute infarction. CTP showed a wedge-shaped area of nondetectable flow on CBF (B) and CBV (C) maps in the posterior frontal and anterior parietal lobes (gray color represents an unmeasurable number such as 0 or infinity), which would typically suggest infarction with a small penumbra anteriorly. Lytics were given promptly with complete resolution of the patient’s symptoms within minutes. MR imaging DWIs obtained <24 hours later demonstrated no corresponding abnormality (D), and turbo FLAIR images (not shown) confirmed the chronic deep white matter findings.
References
Reply:
We thank Drs. McKinney et al for their interest in our manuscript1 and for their thoughtful comments regarding the potential role of CT perfusion imaging (CTP) in the triage of acute stroke patients. Their case, of a patient imaged within 3 hours of stroke onset, with an apparent ischemic lesion on both CT-cerebral blood volume (CBV) and CT-cerebral blood flow (CBF) maps, but with normal diffusion-weighted MR imaging (DWI) at 24 hours, raises a number of important issues. Explanations for this discordance between the suggestion of infarct on the admission CBV maps and the absence of infarct on the follow-up DWI scan could be either physiologic or technical.
First, we emphasize that an implicit goal of “advanced” stroke imaging, whether using DWI/perfusion-weighted MR imaging (PWI) or CT CBV/CBF mismatch, is to extend the time window for treatment so that it can be applied to patients beyond a narrow 3-hour period. We are not aware of any paper, including our own, that advocates exclusion from intravenous thrombolysis based only on early (less than 3 hours) MR imaging or CTP findings. Indeed, there are rare but well-documented examples in the literature of true DWI reversibility in the setting of early, complete reperfusion of ischemic regions.2–4 We have occasionally observed a similar phenomenon with CT angiography source images (which are blood volume weighted), though no CBV reversibility occurred in our study cohort (none of whom recanalized within 3 hours of stroke onset).1
Perfusion thresholds for tissue viability likely depend on the timing of reperfusion and are likely significantly lower at very early time points following vascular occlusion. For example, Jones et al5 demonstrated in a primate model that the CBF threshold for viability associated with only 2–3 hours of middle cerebral artery (MCA) occlusion was 10–12 mL/100 g/min, whereas this threshold rose to 17–18 mL/100 g/min when the MCA was permanently occluded. The case presented by McKinney et al might be explained by recanalization of the occluded MCA branch just after the CT examination. Such early complete reperfusion has the potential to reverse the acute CT-CBV lesion, with subsequent normalization of the apparent diffusion coefficient (ADC) at 24 hours. This ADC normalization could reflect either truly normal or “pseudonormal” tissue, with the latter being brain parenchyma that remains destined to infarct, despite a transient restoration of energy metabolism and hence normal proton diffusion at early follow-up. In fact, most patients in Dr Kidwell’s paper2 who had reversal of restricted diffusion immediately following intraarterial thrombolysis, showed encephalomalacic change in the initially ischemic regions at 1-week follow-up.
The discrepancy between the initial CBV and the subsequent DWI images could also have occurred for technical reasons. A neck CTA was not obtained in this case. If there had been a proximal internal carotid artery occlusion or severe stenosis, contrast filling of the ischemic territory could have been delayed. This, in combination with the authors’ short CTP acquisition time of 40 seconds and a possible M3 occlusion, could have resulted in a falsely low CT-CBV measurement, overestimating the degree of potentially infarcted tissue. We therefore recommend a CTP acquisition time of at least 50–60 seconds to reduce this problem of “perfusion weighting” of the blood volume maps. Even without an internal carotid artery stenosis or occlusion, the matched CBV-CBF lesion shown in this case could have resulted in part from poor filling distal to the M3 lesion described on the CTA (not shown). Had the acquisition time been longer, additional contrast may have reached the territory of the CBV lesion via collateral flow.
Poor signal intensity-to-noise ratio on the CTP source images could also lead to false-positive perfusion maps. We recommend that at least 45–50 mL of contrast with 300 mg iodine/mL (or its equivalent) be administered when performing CTP to achieve adequate signal intensity. In addition, we have found that thicker CTP map sections (10 mm rather than 5 mm) have an improved signal intensity-to-noise ratio.6 Also, accurate quantification of both CBF and mean transit time is optimized with a software package capable of deconvolution. Finally, other factors, such as streak and motion artifact, could result in false-positive CBV images. Careful review of the CTP source images, as well as the arterial and tissue time-course curves, is mandatory.
In summary, there are a number of possible explanations, both physiologic and technical, for the discrepancy between the CBV and DWI findings in the case presented. We again are grateful to Drs. McKinney et al for calling these potential pitfalls of CTP acquisition and interpretation to the attention of AJNR readers.
- Copyright © American Society of Neuroradiology