Abstract
SUMMARY: Vertebral artery recanalization in symptomatic stenosis/occlusion remains controversial, as no definite evidence exists regarding this topic. There are only a few reports regarding the feasibility and safety of recanalization in the first segment of the vertebral artery with atherosclerotic vertebral ostial occlusion. We report our experience treating first segment occlusion in 8 patients and present a balloon protection technique used to reduce the thromboembolic burden during the stent placement procedure. The outcome at 3 months showed an mRS ≤2 except for a patient with a poor initial status with basilar artery occlusion. Revascularization of a rather long first segment occlusion is technically feasible and can be safely performed by use of embolic protection methods.
ABBREVIATIONS:
- BA
- basilar artery
- DUS
- Doppler ultrasonography
- NIHSSo
- National Institutes of Health Stroke Scale on admission
- PcomA
- posterior communicating artery
- VA
- vertebral artery
- V1
- first segment of the vertebral artery.
Atherosclerotic lesions of the vertebral artery (VA) origin are a potential cause of posterior circulation ischemia, which has approximately a 20% to 50% risk for vertebrobasilar TIA or stroke.1 The VA ostium is known to be the most common site for atherosclerosis of the posterior circulation.2 Patients with symptomatic severe (> 70%) ostial VA stenosis face an 11% annual risk for recurrent stroke or TIA while receiving medical treatment.3
Early understanding of the stroke mechanism of VA ostial lesions was focused on their hemodynamic nature, which commonly presents with TIA including dizziness.4 After a few case series that described an embolic infarct originating from vertebral ostial occlusive disease,5 artery-to-artery embolism was also considered as an important cause of the stroke mechanism in lesions in the first segment of the VA (V1).6 A large series of patients revealed that half of the artery-to-artery embolic strokes in the posterior circulation were the result of VA ostial lesions.7 The presence of a clot in the vertebral ostial plaque provides the cause-and-effect relationship between vertebral arterial ostial lesions and posterior circulation stroke.8 Even a large ulcer in the vertebral arterial ostium has been described as adding to the possibility of the vertebral arterial ostium as a source of emboli.9
The optimal treatment of patients with symptomatic severe ostial VA stenosis is still unclear.3 The only randomized trial comparing endovascular with medical treatment is the Carotid and VA Transluminal Angioplasty Study.10 This study did not show a separate result for the small number of its patients (n=16) with VA stenosis, though it failed to demonstrate the benefit of angioplasty of carotid and vertebral stenosis.
Although several articles have dealt with revascularization of vertebral ostial lesions, they mostly have described revascularization of vertebral arterial stenosis.11 To our knowledge, only a few case reports have demonstrated anecdotal evidence of revascularization for vertebral ostial occlusion.12 Therefore, we present the revascularization procedure and the outcome of vertebral ostial occlusion in strokes related to artery-to-artery embolism.
Materials and Methods
Case Series
Eight consecutive patients (12%) were identified among 68 consecutive patients who were treated by angioplasty and/or stent placement for atherosclerotic stenosis or occlusion in the V1 segment between January 2002 and October 2012. There were 7 men and 1 woman with a median age of 67 years (age range, 54–75 years). Informed consent was obtained from each patient or their relative. This study was approved by our institutional review board.
The patients' baseline characteristics are listed in the On-line Table. Patients with acute stroke who were within 6 hours of symptom presentation (n=1) were treated according to our institution's inclusion criteria, which had been modified after other previously reported IV or intra-arterial thrombolytics.13 Patients with vertebrobasilar insufficiency or lacunar syndromes where vascular imaging results did not show any corresponding ischemic lesion were excluded from the analysis. All patients underwent a standard etiologic work-up, including blood tests (hemoglobin, white cell and platelet counts, prothrombin time, activated partial thromboplastin time, serum electrolytes, glucose, urea, creatinine, transaminases, and cholesterol levels); 12-lead electrocardiography; cranial CT and/or MR imaging (axial T2, intermediate-weighted images, fluid-attenuated inversion recovery, time-of-flight MR angiography, DWI, and contrast-enhanced MRA of the cervical vessels at 1.5T or 3T). The following ancillary investigations were carried out at the discretion of the neurologist treating the patient with stroke: neurovascular sonography, perfusion-weighted images, CTA, transthoracic echocardiography, and 24-hour electrocardiographic monitoring. The severity of the neurologic deficit was assessed on admission by a neurologist by use of the National Institutes of Health Stroke Scale on admission (NIHSSo) and is shown in the On-line Table. The study patients did not have cardiogenic problems suggesting the possibility of cardioembolism. The patient symptoms and signs varied at the time of presentation according to the time from the onset or the extent and degree of the acute ischemic lesion seen on DWI. Although there was only dizziness experienced by 2 patients at the time of hospital admission, both of these patients had a history of fluctuating neurologic deficits including dysarthria or sensory changes in the upper extremity.
Two patients (patients 5 and 6) had taken oral antiplatelet agents (ie, aspirin and/or clopidogrel) and a statin for 1 month when the severe VA ostial stenosis related to dizziness was detected. One patient (patient 8) had taken aspirin for 1 month for stroke prevention and without undergoing any vascular imaging. The remaining patients did not take any antiplatelet medication, probably because they did not have any symptoms until an acute stroke developed because of VA occlusion.
Cerebral Angiography and Revascularization Procedures
The lesion characteristics were then further studied by DSA. The contralateral VA, the posterior communicating artery (PcomA) from the anterior circulation, and the thyrocervical and costocervical trunks were evaluated to determine the collateral supply distal to the occluded vertebral segment. The PcomA size was designated as normal if it was visible on MRA without any hypoplasia of the ipsilateral P1 segment of the posterior cerebral artery.
The procedures were performed via the transfemoral route with the patient under local anesthesia. Cerebral angiography, which was performed with a 4F angiocatheter, included examination of both internal carotid arteries and both subclavian arteries, including both VAs. An optimal subclavian arteriogram was obtained to reveal the entire segment of the occluded VA by demonstration of collateral channels via the thyrocervical or costocervical trunks, as well as the presence of the anterior spinal artery and the status of the intracranial posterior circulation including the basilar artery (BA). The angiographic assessment of the external carotid artery-VA anastomosis via the occipital artery and the ascending pharyngeal artery, both of which are known to be effective collateral pathways in proximal VA stenosis, was also performed unless collateral filling of the VA was demonstrated via the ascending or deep cervical artery from the subclavian artery.14,15
After analysis of the complete angiogram, we inserted a 6–9F guiding catheter in the subclavian artery. After placement of the guiding catheter, a 4F catheter was then passed along a 0.035-inch guidewire through the occluded segment and a distal balloon (ie, PercuSurge; Medtronic, Minneapolis, Minnesota), was then introduced via the 4F angiocatheter, as shown in Figs 1 and 2. The location of the 4F catheter tip in the free lumen was identified by the suctioning of thrombotic debris in the distal segment beyond the occlusion and the regurgitation of blood. Once protective distal occlusion was achieved, suction of the proximal ICA segment was followed by angioplasty and/or stent placement.
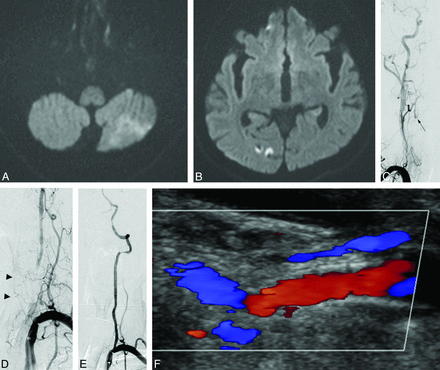
A 70-year-old man presented with severe dizziness and ataxia. DWI showed a wedge-shaped infarct in the left cerebellar hemisphere (A) and multifocal infarcts in the right occipital lobe (B). C, An angiogram of the right subclavian artery showed occlusion of the hypoplastic right VA at the C4 level (arrow). Both internal carotid arteriograms showed poor development of both PcomAs (not shown). D, An arteriogram of the left subclavian artery shows occlusion of the V1 segment. Note the filling of the anterior spinal artery arising from the costocervical trunk in the late phase of the angiogram (arrowheads). E, Final angiogram after stent placement shows good patency of the occluded VA and good filling of the posterior circulation. F, Good patency of the stented lumen was seen on follow-up DUS 4 months later and without any neurologic deficit (mRS=0).
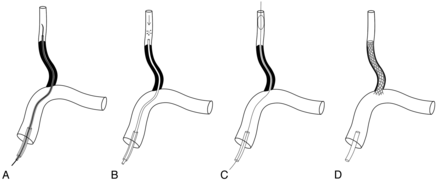
Diagram showing the recanalization process in patients with V1 segment occlusion. A, Smooth probing and traversing of the occluded segment by a 0.035-inch guidewire are followed by the introduction of a 4F angiocatheter (or a guiding catheter), which can also allow the passage of a PercuSurge protective balloon guidewire. B, Passage of the occluded segment can be identified by free regurgitation of blood as well as suction of debris beyond the occluded segment. Under distal balloon protection (C), angioplasty is followed by stent placement (D).
If the position of the guiding catheter was unstable, we inserted an additional sturdy guidewire as a supportive wire into the ipsilateral brachial artery. The occluded segment was probed by using a 0.014–0.035-inch guidewire.16⇓–18 In 3 patients, we used the exchange technique after passing the occluded V1 segment by using a combination of a 4F angiocatheter and a 0.035-inch guidewire (Figs 1 and 2). Once the guidewire was introduced, subsequent angioplasty by use of a 2–4-mm diameter angioplasty balloon was followed by stent placement. The degree and occlusion length were measured with clear delineation of the vertebral ostium with pixel shift on DSA. A self-expandable stent was placed with minimal protrusion to the subclavian artery to minimize distal migration of the stent and to properly cover the atherosclerotic lesion in the occluded segment. Poststenting angioplasty was performed if there was residual stenosis. In 2 patients with vertebral ostial occlusion with concomitant embolic occlusion of the basilar artery demonstrated on MRA and DSA, we revascularized the BA by using a self-expandable stent because mechanical thrombolysis or the introduction of urokinase to reduce the clot burden was not effective.
Preprocedural antiplatelet conditioning with 200 mg of aspirin and a loading dose of 300 mg of clopidogrel was given if the patients were not already taking these medications. The heparin was adjusted to achieve an activated clotting time of 2–2.5 times that of the baseline. After the revascularization procedure, maintenance doses of 100 mg of aspirin and 75 mg of clopidogrel were given. Also given were 75 mg of clopidogrel for at least 6 months and 100 mg of aspirin as a permanent medication.
Immediate results were evaluated according to the angiographic patency of the stented segment. Postprocedural precontrast CT or MR imaging scanning was then performed within 24 hours to identify any new infarction or hemorrhage. The NIHSSo and a 3-month mRS were compared to assess the clinical results of the procedures. Follow-up imaging was obtained in 4 patients with Doppler ultrasonography (DUS) (n=2) or CTA (n=2). For all patients treated with stent placement, routine angiographic follow-up was recommended 6–12 months after the procedure. However, patients who were at high risk for procedural complications during angiography and who had no symptoms or signs of ischemia, as well as patients who declined the recommendation, did not undergo routine follow-up angiography and, instead, underwent CTA or transcranial DUS which are noninvasive diagnostic tools used for follow-up in patients who have undergone V1 stent placement, as we already reported.19 We evaluate the mobile V1 segment with DUS because, in most patients, the stented segment can be seen without difficulty. The implanted stent is also a good anatomic landmark for clear visualization of the lumen. Restenosis on CTA was determined by reconstituted delineation of traced-stented vessel segments by Advanced Vessel Analysis (Siemens, Erlangen, Germany) along the stented vessel.
Results
Angiographic and Imaging Findings
Angiographic analysis revealed that all vertebral occlusions occurred in the dominant VA. The mean length of the occlusions was 43 mm (range, 10–69 mm). The contralateral VA was hypoplastic in all patients; it barely filled the BA (n=4), only filled the posteroinferior cerebellar artery (n=2), and was occluded in the proximal segment (n=2). The PcomA filled the distal aspect of the BA in 2 cases. Other patients showed no PcomA contribution to the BA flow. The PcomA was not well developed in any of these patients, thus suggesting that hypoperfusion-associated stump embolism was the primary stroke mechanism in these patients. The proximal stump of the V1 occlusion in the subclavian artery was barely able to be identified, thus suggesting that the occlusion was related to an atherosclerotic ostial plaque lesion leading to a tight occlusion. The deep cervical artery contribution was the primary angiographic collateral at the C3–4 levels of the VA distal to the occluded segment, as seen on DSA. The anterior spinal artery filling from the costocervical trunk was identified in 2 patients. There was no collateral development via the anterior spinal artery in our study patients.
Results revealed acute infarct in the cerebellum, brain stem, thalami, and occipital lobes in all 8 patients. The infarct lesions seen on DWI varied in size from focal or localized to territorial. Two patients had focal ischemic lesions in the middle cerebral artery territory, probably caused by concomitant perforator infarcts.
Recanalization Outcome
Endovascular revascularization was successful in 7 of 8 patients. Revascularization failed in 1 patient because of failed guidewire passage through the occluded vertebral ostium. Each patient's data are summarized in the On-line Table. Poststenting angioplasty was required in all patients because of elastic recoiling of the vertebral ostial lesion after angioplasty and stent placement. After postdilation, there was no significant residual stenosis. In 2 patients with concomitant BA occlusion that was identified as a thromboembolism on both MRA and DSA, we deployed a self-expanding stent in the BA in 1 patient and thrombolysis with 100,000 U of urokinase followed by a self-expanding stent in the BA in the other patient.
There were neither immediate (<24 hours) nor delayed (≥24 hours) complications except for 1 complication in a patient in whom a microguidewire remained in the BA and detached in the femoral artery because of inadvertent anchoring to the stent strut while advancing the microguidewire beyond the deployed stent in the mid-BA to the remaining basilar top occlusion. CT perfusion the next day showed good patency of the stented vessels, and DWI 5 days later showed localized infarcts in the cerebellum and the left temporal lobe, but without any symptom aggravation. This patient improved to NIHSSo 6 from NIHSSo 9, had an mRS score of 1 at the 3-month follow-up, and showed good patency of the stented lumen on CTA without any symptom recurrence during the 2.5-year follow-up period. Therefore, the rate of technical complications was 12.5% (95% CI, 2.24%–47.10%). However, there were no clinical complications in all 8 patients (95% CI, 0.00%–32.44%).
No hemorrhage or aggravation was seen in any patients, based on images taken during the following 24 hours after the procedure (n=5). Three patients did not undergo an imaging study because there was no clinical symptom aggravation and these patients had relief of their symptoms. Seven patients scored ≤2 in the mRS at 3 months. A patient with BA occlusion presented with a poor clinical condition (NIHSSo=17), resulting in a poor outcome at 3 months (mRS=5). There was good patency and no in-stent restenosis in 2 patients, as seen on DUS (n=2) after 4 months and 10 months, respectively. Another patient (patient 6) monitored with CTA at 5 months after the procedure showed 40% restenosis at the ostium but without symptom recurrence. Other study patients did not undergo imaging follow-up and had no symptom recurrence.
Discussion
Approximately 7.6% of patients who clinically manifest atherosclerotic disease have an asymptomatic VA stenosis or occlusion visible on DUS.20 However, to our knowledge the incidence of stroke caused by V1 occlusion has not been reported in the general population. Our study showed that stroke caused by V1 segmental occlusion was responsible for 12% of the recanalization procedures performed in the V1 segment. Our patients revealed dominant VA occlusion with insufficient or occluded flow through the contralateral VA.
Large-artery disease, especially in the V1 segment of the VA, is known to demonstrate topographic heterogeneity of the cerebellar infarcts, as seen in our study, which revealed a wide range of posterior circulation infarcts related to vertebral ostial occlusion strokes.21 Artery-to-artery embolism from the stump of the occluded V1 segment seems to be involved more often in large arterial embolic stroke than in hemodynamic stroke.22 The prevalence of the so-called “VA stump syndrome” is 1.4% in patients with posterior circulation stroke, the rate of recurrence of ischemic stroke is 25%, and it shows unfavorable outcomes in 25% of patients with posterior circulation stroke.23 A low-flow state in the stump might cause thrombus formation that can lead to distal embolism.24 Retrograde filling of the blood flow via the C3–4 collaterals through thyrocervical or costocervical trunks, or C1–2 anastomosis via the occipital artery or the ascending pharyngeal artery, slowly fills the occluded segment, and sluggish blood flow forms a clot as a source of emboli to the posterior circulation.25 Although the number and distribution of acute ischemic lesions in our patients were too varied to categorize them into a specific stroke pattern, such findings may suggest rather remote artery-to-artery embolism from the VA ostial stump in a relatively hypoperfused posterior circulation. However, the association of a perforator infarct cannot be excluded in this small number of patients.
Several studies have shown that thromboembolism caused by erosion of the plaque surface, or plaque rupture, can contribute to posterior circulation infarcts.23⇓–25 As VA-origin plaques are hard, smooth, and concentric and are less prone to ulceration or intramural hemorrhage, they are therefore known to have less risk for embolism than carotid bifurcation plaques.26 However, acute thrombus formation at the stump or due to plaque rupture can cause sudden occlusion of the V1 segment as well as a large thrombus burden.27,28
The endovascular treatment of VA atheromatous disease remains controversial, as its natural history is poorly understood and no randomized trials have assessed the effectiveness and long-term outcome of this therapy.29 There were a few anecdotal case reports regarding the protection technique in recanalization of a V1 occlusion by use of a flow-reversal technique30,31 or a distal filter protection device.32 Our clinical experience has shown that smooth passage of a 0.035-inch guidewire could be followed by the introduction of a 4F angiocatheter, which allows clot aspiration and also accepts a distal balloon system with a 0.014-inch outer diameter. A more rigid microguidewire can also be used for probing the occluded segment. We performed vertebral ostial stent placement by using a self-expandable stent19 because of its long lesion coverage, radial flexibility, and conformability to the tortuousness and longitudinal flexibility of a nitinol stent.33
Endovascular recanalization of the occluded V1 can be warranted for patients in whom stroke has developed despite medical treatment or for patients with tandem lesions. The timing of recanalization therapy must be at least in the subacute stage because recanalization 2 months after the onset of symptoms failed in one of our study patients. In addition to medical treatment of acute or subacute occlusion of the V1 segment, a recanalization procedure, if required, must be done in the acute or subacute stage of occlusion because successful recanalization may not be achieved in chronic occlusion or may not be necessary when there is good development of collaterals.16,17 In acute stroke with poor clinical status, recanalization should not be hesitated as a first-line of treatment. In patients with subacute stroke without alleviation of symptoms, recanalization could be an option to improve clinical status and to prevent further embolic stroke. Although surgical bypass can be performed for vertebral ostial occlusion, surgical morbidity seems to be significant and these procedures require shunt appliances.34 If a lesion is not accessible via the endovascular route, surgical treatment may be considered if there is no response to maximal medical therapy or if there is enough of a distal stump in the occluded V1 segment.35
Study Limitations
There were several limitations to our study. For example, the sample size of the study was relatively small, and the number of imaging follow-ups was limited in this patient cohort, thus making us unable to evaluate the rate of restenosis. Our series was based on a retrospective analysis, which can create bias for this recanalization procedure and which cannot predict the final outcome of patients with V1 ostial occlusion without performing a randomized controlled comparison. Therefore, recanalization should be used with caution in patients with symptomatic V1 occlusion. Other possibilities, such as dissection or vasculitis, could not be completely excluded. However, a small stump caused by tight stenotic atherosclerotic plaque differs from a normal-looking stump caliber, as shown in dissection.36 Intravascular DUS can be helpful in making the differential diagnosis.37 Lastly, because the follow-up period of our study was short and the long-term clinical benefits of recanalization treatment still remain unclear, such benefits should be more thoroughly evaluated in future studies.
Conclusions
The thrombus burden of an occluded V1 segment is variable and can cause a serious embolic infarct in the posterior circulation. Revascularization of a vertebral ostial occlusion can be achieved with good clinical results by use of strategic revascularization based on the V1 occlusion stroke mechanism. In some patients, passage of the occluded segment can be performed safely by gentle probing with use of a guidewire followed by introduction of an angiocatheter. Subsequent recanalization by angioplasty and stent placement can be successfully performed under balloon protection in some patients. Re-establishment of the antegrade vertebrobasilar blood flow via endovascular recanalization might be an option used to decrease stroke recurrence in selected patients with acute posterior circulation stroke caused by an ostial V1 segment occlusion.
References
- Received April 11, 2013.
- Accepted after revision May 31, 2013.
- © 2014 by American Journal of Neuroradiology