Abstract
SUMMARY: As we defeat infectious diseases and cancer, one of the greatest medical challenges facing us in the mid-21st century will be the increasing prevalence of degenerative disease. Those diseases, which affect movement and cognition, can be the most debilitating. Dysfunction of the extrapyramidal system results in increasing motor disability often manifest as tremor, bradykinesia, and rigidity. The common pathologic pathway of these diseases, collectively described as parkinsonian syndromes, such as Parkinson disease, multiple system atrophy, progressive supranuclear palsy, corticobasal degeneration, and dementia with Lewy bodies, is degeneration of the presynaptic dopaminergic pathways in the basal ganglia. Conventional MR imaging is insensitive, especially in early disease, so functional imaging has become the primary method used to differentiate a true parkinsonian syndrome from vascular parkinsonism, drug-induced changes, or essential tremor. Unusually for a modern functional imaging technique, the method most widely used in European clinics depends on SPECT and not PET. This SPECT technique (described in the first of 2 parts) commonly reports dopamine-transporter function, with decreasing striatal uptake demonstrating increasingly severe disease.
ABBREVIATIONS:
- DaT
- dopamine transporters
- 18F
- fluorine 18
- 18F-DOPA
- 6-[18F]fluoro-L-3,4-dihydroxyphenylalanine
- 123I
- iodine 123
- 123I-FP-CIT
- 123I-ioflupane (N-ω-fluoropropyl-β CIT)
- PD
- idiopathic Parkinson disease
- SWEDD
- scans without evidence of dopaminergic deficit
Parkinsonian syndromes are a group of movement disorders characterized by tremor, bradykinesia, and rigidity. They are most frequently due to primary neurodegenerative disease, resulting in loss of dopaminergic nerve terminals along the nigrostriatal pathway, as occurs in idiopathic Parkinson disease (PD), multiple system atrophy, progressive supranuclear palsy, corticobasal degeneration, and dementia with Lewy bodies. Other causes of parkinsonism that do not involve nigrostriatal degeneration include drug-induced, vascular, or toxic insult and psychogenic disease. Patients with nigrostriatal degeneration may benefit from dopaminergic medication, whereas nondegenerative or postsynaptic etiologies do not.
The clinical diagnosis of PD, based on clinical signs, assessment of inclusion and exclusion criteria, and good response to levodopa,1 can be straightforward. However, mild, atypical, or ambiguous signs with unconvincing responses to levodopa can make the diagnosis inconclusive.2 Lack of diagnosis may have adverse prognostic implications in the management of parkinsonism. Where there is clinical doubt, functional imaging may be decisive.
Pathophysiology of Parkinson Disease
PD accounts for approximately 75% of all cases of parkinsonism.3 Clinicopathologic studies have established the underlying cause of the dominant motor features of PD as dysfunction of the dopaminergic neurotransmitter system.4
Dopamine is a neurotransmitter that plays a central role in the regulation and control of movement, motivation, and cognition. Dopaminergic neurons are predominantly found in the midbrain and project from the substantia nigra pars compacta to the striatum (caudate nucleus and putamen). This is the presynaptic nigrostriatal dopaminergic pathway. The postsynaptic neurons lie in the striatum.
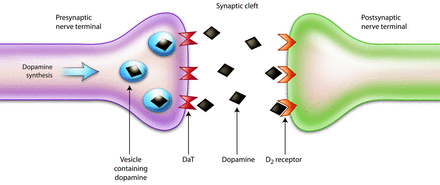
Dopamine is produced by the dopaminergic neurons and is stored in vesicles within the neuron, which protect it from oxidation by monoamine oxidase. Once the appropriate signal is received by the presynaptic neuron, dopamine is released into the synaptic cleft, where it interacts with dopamine receptors on the postsynaptic striatal neuron (Fig 1). To regulate dopamine levels and enable a response to the next signal, extracellular dopamine is actively pumped back into the presynaptic neuron by dopamine transporters (DaT) on the cell surface, where dopamine may either be re-stored in vesicles or catabolized into breakdown products.
Schematic illustration of the dopaminergic pathway within the striatum. Dopamine is synthesized and stored in vesicles until released into the synaptic cleft in response to an action potential. After interacting with the postsynaptic dopamine receptors, dopamine is released back into the synaptic cleft, where it is actively taken up by dopamine transporters and carried into the presynaptic neuron.
PD is characterized neuropathologically by degeneration of the substantia nigra pars compacta within the midbrain with subsequent deposition of neuronal Lewy bodies. Nigrostriatal degeneration, with consequent DaT depletion and downregulation, has the effect of reducing the levels of dopamine within the striatum, manifesting clinically as resting tremor, rigidity, and bradykinesia. Nigrostriatal degeneration is asymmetric, with almost all patients diagnosed with PD presenting initially with unilateral signs.
An Overview of Imaging in Neurodegenerative Parkinsonian Syndromes
Conventional MR imaging alone is not sufficiently sensitive in diagnosing these disorders.5 Volumetric, spectroscopic, and diffusion MR imaging techniques and transcranial sonography show promise but are seldom used in routine clinical practice. Much of the evidence surrounding these methods derives from retrospective studies or small series. Moreover, differentiation among disease types is commonly demonstrated in group comparisons, rather than allowing a specific diagnosis in an individual patient. The most sensitive imaging techniques for an early diagnosis of parkinsonian syndromes are SPECT and PET, by using ligands that report nigrostriatal dopaminergic function.6 SPECT has proved useful in differentiating parkinsonism due to nigrostriatal degeneration and nondegenerative or postsynaptic causes. SPECT is, therefore, a valuable tool for the clinician in determining the correct management algorithm for the patient. However, other imaging modalities may exclude treatable nondegenerative etiologies and play a role in differentiating neurodegenerative parkinsonian disorders.
Imaging the Dopamine Transporter
DaT is a sodium chloride–dependent transmembrane protein localized to the presynaptic nigrostriatal cell surface,7 whose expression is not fixed but can be up- or downregulated by certain factors, by using membrane trafficking as the key mechanism.8 For example, insulin can increase DaT expression on the cell surface, while amphetamines or levodopa can decrease expression.9,10 In vivo SPECT imaging of DaT with specific tracers provides a guide to the attenuation of functional dopaminergic neurons.
Postmortem studies have shown a severe reduction of presynaptic cocaine-binding dopamine receptors associated with DaT in patients with PD.7 Consequently, DaT selective radiotracers have been developed by using analogues of cocaine, of which iodine 123 (123I)- β CIT (2β-carbomethoxy-3β-(4-iodophenyl) tropane) and 123I-ioflupane (N-ω-fluoropropyl-β CIT; or FP-CIT) are the most widely used11,12 and are used in the DaT-SPECT clinical studies described below. Other ligand tracers include 99mTc-TRODAT [2-[[2-[[[3-(4-chlorophenyl)-8-methyl-8-azabicyclo[3.2.1]oct-2-yl]methyl](2-mercaptoethyl)amino]ethy]-amino]ethanethiolato-(3-)-N2,N2′,S2,S2′]oxo-[1R-(exo-exo)], which has a lower striatal uptake compared with background than 123I probes13 and 123I-IPT [N-(3-iodopropen-2-yl)-2β-carbomethoxy-3β-(4-chlorophenyl) tropane].14 123I-FP-CIT (trade name DaTSCAN; GE Healthcare, Buckinghamshire, United Kingdom) has been the preferred radiotracer due to the shorter interval (3–6 hours) between injection and image acquisition.15 In addition, its uptake is not competitive with dopamine products, so treatment with levodopa can be continued, though other drugs such as monoamine oxidase inhibitors can decrease uptake. 123I-FP-CIT has been approved by the European Medicines Agency since 2000 and the US Food and Drug Administration since 2011.
The Role of DaT-SPECT Imaging in Parkinsonian Syndromes
DaT-SPECT imaging enables differentiation of neurodegenerative causes of parkinsonism, where typically patients will have abnormal scans, from other movement or tremor disorders where typically the DaT-SPECT study will be normal. Indications for DaT-SPECT imaging are given in the Table.
Clinical indications for DaT-SPECT imaging
Before the use of functional imaging, the diagnosis of a neurodegenerative parkinsonian disorder depended on clinical evaluation with confirmation, when applicable, based on postmortem neuropathology. Even in specialized centers for movement disorders, where the positive predictive value of the final clinical diagnosis for all patients with parkinsonism was 85% (99% for PD alone), 36% of patients were reclassified within a mean interval of 3.4 years from initial diagnosis.16 Since then, an industry-sponsored, prospective, 3-year longitudinal, multicenter study compared clinical scoring methods and DaT-SPECT findings in patients with clinical diagnostic uncertainty.17 Clinical scoring methods led to an overdiagnosis of PD, and it was suggested that DaT-SPECT should be implemented to reduce overdiagnosis in this population. In another recent, industry-sponsored, multicenter, randomized controlled trial, patients with a clinically uncertain parkinsonian syndrome or a monosymptomatic, atypical, or incomplete presentation of parkinsonian clinical features were randomized to DaT-SPECT imaging to see whether it led to a change in clinical diagnosis during 1 year.18 At 1 year, 54% of the patients randomized to DaT-SPECT imaging had a change in diagnosis, compared with 23% of the controls, and the imaging led to an increase in confidence in the clinicians' diagnoses. The most common change in management was initiation of medication, including dopaminergic therapy, confirming results from a previous multicenter trial.2 Seventy-five percent of patients randomized to DaT-SPECT imaging treated by general neurologists had a change in diagnosis, as did 47% of those treated by a movement-disorder specialist, suggesting utility in both a general and specialist setting. The diagnostic stability of DaT-SPECT with time, together with a high interobserver agreement rate (κ = 0.97), demonstrates the reproducibility and reliability of the technique and highlights its value in adding objectivity to clinical scoring methods.17
DaT-SPECT imaging may be indicated at baseline or after a period of monitoring. The radiation dose is approximately equivalent to a head CT when using 123I-FP-CIT.2 Central nervous stimulants, such as amphetamines and other sympathomimetics, have a high affinity for DaT protein. Serotonin-reuptake inhibitors may upregulate or downregulate DaT. Therefore, these drugs can potentially confound DaT SPECT image interpretation and are usually stopped temporarily before imaging. Images are acquired by using standard gamma camera systems 3–4 hours after injection of 185-MBq 123I-FP-CIT. The quality of the scan is dependent on optimization of the acquisition; it is essential that patient movement be minimized and that the rotational radius of the gamma camera detector head be as small as possible. Following reconstruction by using an iterative technique, images are usually displayed as axial sections parallel to a line drawn from a point where the anterior part of the frontal lobe is at its maximum distance from the posterior part of the occipital lobe. For diagnostic purposes, visual assessment of DaT binding appears to be comparable with semiquantitative analysis of ROIs normalized to reference brain (where there is absent or low background DaT expression).19 Detailed 123I-FP-CIT practice guidelines have recently been published by the Society of Nuclear Medicine.20
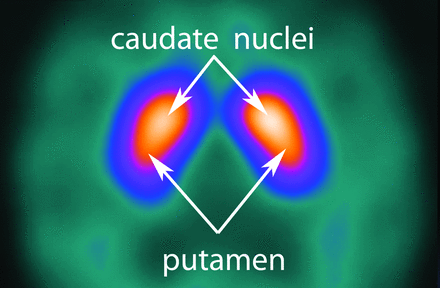
Because most dopaminergic transmission occurs in the striatum, this area will show the maximum uptake of DaT radiotracers, with minimal background activity in the remainder of the brain. In scans with normal findings, the striata appear as symmetric “comma” shapes (Fig 2). Any asymmetry or distortion of this shape, in the absence of patient motion, implies an abnormal scan finding.
Normal study findings. The axial 123I-FP-CIT DaT-SPECT image demonstrates symmetric tracer uptake in the caudate nuclei and putamina, with very low-grade, almost absent, background activity.
Parkinson Disease
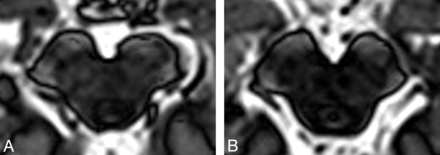
Structural abnormalities on MR imaging in PD are subtle and, other than nonspecific putaminal atrophy, are found in <20% of patients with PD.21 There may be nonspecific abnormalities of the substantia nigra, including “smudging” of the hypointensity in the substantia nigra toward the red nucleus on T2-weighted sequences (Fig 3)22 or increased signal with gray matter–suppressed T1-weighted inversion recovery sequences (Fig 4).23 Voxel-based morphometry might reveal nonspecific gray matter loss of limbic, paralimbic, and prefrontal cortices.24 Pathologic diffusion imaging findings in patients with PD are very rare5; however, elevated ADC may be seen in the region of the olfactory tracts in patients with PD compared with healthy controls.25 1H-MRS studies are nonspecific, showing reduced NAA/Cr and NAA/choline ratios in the basal ganglia, reflecting neuronal loss.26
Axial T2-weighted MR imaging sequence in a patient with PD (A) demonstrating “smudging” of the hypointensity in the substantia nigra toward the red nucleus in the midbrain (right subtlely more marked than the left, concordant with the contralateral clinical features). It is more easily visualized in the corresponding susceptibility-weighted image (B). Axial T2*-weighted MR imaging sequence also demonstrates the “smudging” sign as shown in another patient with PD (C) (left more than right concordant with worse contralateral clinical features). The “smudging” sign is nonspecific and seen in progressive supranuclear palsy, multiple system atrophy, and corticobasal degeneration. It is a subtle sign and often not used in routine clinical practice.
On these inversion-recovery T1-weighted images in which deep gray matter signal is suppressed, the substantia nigra in a patient with severe PD (A) appears both substantially shrunk and with altered contrast in comparison with a healthy control (B). There is a correlation between the substantia nigra area with the Unified Parkinson Disease Rating Scale score.23 There is also a group difference between those with PD and controls; however, this metric has not been proved to be useful for individuals. Images courtesy of Dr Ludovico Minati, Scientific Department, Istituto Di Ricovero e Cura a Carattere Scientifico Foundation Neurologic Institute, Carlo Besta, Milan, Italy.
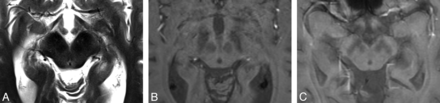
Transcranial sonography reveals hyperechogenicity of the substantia nigra (Fig 5), which, together with normal echogenicity of the basal ganglia, has a positive predictive value of 93% for a prospective PD diagnosis.27 An advantage of transcranial sonography is that these findings may also be premotor biomarkers.28 However, a disadvantage is that up to 20% of white and 60% of Asians cannot be studied due to an inadequate temporal acoustic bone window.
T2-weighted fast spin-echo MR image (A) and corresponding transcranial sonography images (B and C) of midbrain axial sections at the orbitomeatal line. B, Transcranial sonography image of the axial midbrain section in a healthy person depicting the hypoechoic midbrain (outlined) surrounded by the hyperechogenic basal cisterns. In the anatomic area of the ipsilateral substantia nigra, only small hyperechoic patches are visible (small arrows); the planimetrically measured area is <0.20 cm2. The raphe is detected as a highly echogenic, continuous line; the aqueduct is shown at the dorsal part of the midbrain. C, Transcranial sonography of a patient with Parkinson disease showing a bilateral hyperechogenic substantia nigra (area ≥0.20 cm2). Small arrows indicate the substantia nigra. Images courtesy of Dr Rita CL Fernandes, Department of Neurodegeneration at Tübingen University Hospital and Hertie Institute of Clinical Brain Research, Tübingen, Germany.
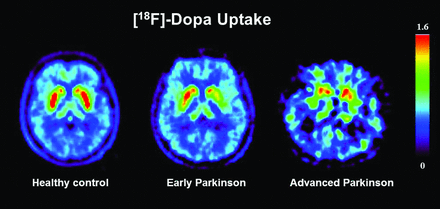
6-[18F]fluoro-L-3,4-dihydroxyphenylalanine (fluorine 18 [18F]-DOPA) PET is a valuable functional technique that provides an indirect measure of nigrostriatal neuron attenuation through evaluation of presynaptic dopa decarboxylase activity and intravesicular dopamine storage. With progressive nigrostriatal degeneration, there will be a consequent reduction in dopa decarboxylase activity and presynaptic dopamine storage. This is reflected on 18F-DOPA PET imaging as a reduction in uptake in the basal ganglia (Fig 6), similar to that in DaT SPECT imaging. With PD, there is a more pronounced reduction of radiotracer striatal binding in the putamen than in the caudate nucleus, which is asymmetric and correlated with disease severity.29,30 Most important, subclinical disease may also be identified on imaging.31 Other PET techniques are less informative.32,33 18F-FDG PET measures regional metabolism and shows normal or increased uptake in the striatum contralateral to the initial clinical signs. Postsynaptic striatal neuron density can also be measured with 11C-[((-)-(S)-3,5-dichloro-N-((1-ethyl-2-pyrrolidinyl) methyl)-6-methoxy-salicylamide tartrate; FLA 870(-); A40664] (raclopride) PET showing normal or increased dopamine receptor binding.
Axial 18F-DOPA PET images through the striatum. The patient with early Parkinson disease shows an asymmetric reduction in putaminal radiotracer uptake. With further disease progression, both putamina show a substantial reduction in radiotracer uptake. Images courtesy of Professor Philippe Remy, l'Hôpital Henri Mondor, Creteil, France.
DaT-SPECT in Parkinson Disease
Like PET, SPECT reflects the typical asymmetric nigrostriatal degeneration with a reduction in striatal DaT binding contralateral to the initial signs. In keeping with PET and neuropathologic evidence, SPECT typically demonstrates preferential DaT loss in the putamen compared with the caudate nucleus,34,35 though symmetric and somewhat uniform striatal loss of DaT may occur in genetic forms of parkinsonism such as those due to parkin (PARK2) mutations (the most commonly known cause of young-onset, autosomal recessive parkinsonism).36
To categorize DaT-SPECT abnormality, the imaging findings from 3 prospective multicenter studies were visually classified into normal and abnormal, of which the latter was further subdivided into 3 different patterns.2,17,37 Studies with normal DaT findings demonstrated symmetric intense tracer uptake in both caudate nuclei and putamina. Abnormal patterns (Fig 7) are classified by Catafau and Tolosa2 as the following: asymmetric uptake with reduced putaminal activity in 1 hemisphere (type 1); clear symmetric reduction of putaminal uptake in both hemispheres (type 2); virtual absence of uptake in both the putamina and caudate nuclei in both hemispheres, resulting in a significant reduction in contrast, and the visualization of background activity throughout the rest of the image (type 3). Some centers use this classification as a routine descriptive system, but there is no clinical or outcome-measure correlation.
Axial 123I-FP-CIT DaT-SPECT sections depicting the different patterns of abnormality seen in PD as described by Catafau and Tolosa2—type 1: asymmetric activity with reduced putaminal uptake in 1 hemisphere (A); type 2: symmetric reduction in putaminal uptake in both hemispheres (B); and type 3: virtual absence of uptake in the putamina and caudate nuclei despite high gain as demonstrated by ample background activity (C).
All studies in a meta-analysis on the diagnostic accuracy of DaT-SPECT demonstrated that patients with PD and healthy patients could be differentiated with 100% specificity.38 However, the sensitivity varied from 38% to 100% among centers. This is reflected in 2 large multicenter clinical trials that used 123I-β-CIT SPECT, which found that up to 15% of patients diagnosed as having PD had normal DaT-SPECT study findings classified as “scans without evidence of dopaminergic deficit” (SWEDD).10,39 It has been suggested that the discrepancy between clinical diagnosis and imaging findings is caused by reductions in striatal DaT availability in early PD that are below the detection threshold.1 However, it is improbable that DaT imaging would have normal findings in initial early PD as the following combined evidence suggests: first, by the time PD clinically manifests, approximately 80% of striatal dopamine has been lost; this change is correlated to presynaptic nigrostriatal cell loss with a parallel reduction in DaT of 80%40,41; second, studies suggest that DaT-SPECT is highly sensitive in detecting dopaminergic degeneration because there is a correlation between the extent of degeneration measured with DaT-SPECT and clinical severity.42 Furthermore, in asymptomatic but hyposmic first-degree relatives of patients with PD, DaT-SPECT predicts subsequent PD.43 Additionally, studies in patients with hemiparkinsonian disorders demonstrate a bilateral loss of striatal DaT.35,42 Follow-up of SWEDD cases has since shown that these patients neither deteriorate nor respond to levodopa and that their DaT-SPECT study findings remain normal.10,17 Some have since been diagnosed with dystonic tremor. In summary, it is thought highly likely that in the SWEDD cases, the initial clinical diagnosis of PD was incorrect and that DaT-SPECT may be more sensitive in diagnosing PD than some studies have shown.
Another potential role for DaT-SPECT imaging is in monitoring disease progression in patients with treated and untreated PD.39,44 Nigrostriatal loss in patients with PD is estimated to be at least 5% per year, which is substantially higher than the age-associated physiologic loss of nigrostriatal neurons, estimated to be 8% per decade.29,44 With this in mind, extending the role of DaT-SPECT to assess the efficacy of neuroprotective therapies in trials has been debated.35 However, because there is no proved correlation between progressive decline in DaT and clinical outcome, its usefulness as a surrogate end point is questionable.45 Furthermore, there is uncertainty about the pharmacodynamic interaction between neuroprotective therapies and radio-labeled ligands binding to DaT. In summary, while DaT-SPECT imaging could have a role in monitoring disease progression in PD, its role as a surrogate biomarker in neuroprotective therapy trials is less clear-cut.
Finally, although we have mentioned that PD may be detected subclinically, we emphasize that there remains no agreed-upon and clearly defined at-risk population to be screened.1
In conclusion, conventional MR imaging alone is not sufficiently sensitive to differentiate neurodegenerative parkinsonian disorders from non-neurodegenerative parkinsonian disorders (disorders other than PD are covered in Part 2). Volumetric, spectroscopic, and diffusion MR imaging techniques and transcranial sonography are very promising but are seldom used in routine clinical practice.
As a screening tool for those at risk of PD and as a biomarker for monitoring disease progression, in particular in neuroprotective therapy trials, the role of DaT-SPECT has yet to be realized. What appears to be the strength of DaT-SPECT, like 18F-DOPA PET, is that nigrostriatal degeneration is observed in both clinically inconclusive parkinsonism and early, even premotor, disease. DaT-SPECT images are easy to interpret compared with advanced MR imaging techniques; the cost is low compared with PET; all compliant patients can be imaged, unlike in transcranial sonography; and at least in Europe, SPECT is widely available compared with PET, advanced MR imaging, and transcranial sonography. DaT-SPECT is of unequivocal clinical value in carefully selected patients, aiding diagnosis and prognosis, as well as therapy. Nevertheless, any enthusiasm to use this technique should be tempered, as with most imaging tests, by the paucity of literature determining how DaT-SPECT actually affects patient outcomes.
Footnotes
Disclosures: Ann-Marie Quigley—RELATED: Other: Travel, hotel, and conference fees were paid by Novartis to attend the European Neuroendocrine Tumor Society 2013 meeting; travel and hotel paid by Nordion to attend the TheraSphere User Group in 2013; No fees were paid to me or my institution. Adam D. Waldman—UNRELATED: Consultancy: Bayer Healthcare, Payment for Lectures (including service on Speakers Bureaus): Bayer Healthcare, Siemens Healthcare, Comments: fees for lectures on education courses, Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: Bayer Healthcare, Comments: expenses for attendance at meetings for discussion groups and consultancy. John Buscombe—UNRELATED: Consultancy: Navidea, Comments: for work on Medicines and Healthcare Products Regulatory Agency submission, Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: BTG, Comments: to learn about TheraSpheres, Germany, February 2014. Anthony H. Schapira—RELATED: receives Medical Research Council and Wellcome Trust funding.
Indicates open access to non-subscribers at www.ajnr.org
REFERENCES
- © 2015 by American Journal of Neuroradiology