Abstract
Summary: Congenital midline spinal hamartomas are relatively rare. Patients harboring this anomaly are generally asymptomatic, but present with an overlying skin anomaly. MR imaging depicts a mass that is isointense with the spinal cord on all sequences, and may show a dermal sinus tract that tethers the cord at the level of the lesion. We report the MR features of congenital midline spinal hamartoma in two children.
<<006>>The more common congenital spinal masses include lipomas, epidermoids, dermoids, and teratomas. A distinct yet relatively unknown entity is that of congenital midline spinal cord hamartoma (1). These lesions are not related to brain malformations or neurofibromatosis, and most patients have few or no symptoms related to them. We believe that MR imaging enables the physician to distinguish these lesions from other congenital spinal masses. We present the MR imaging findings in two cases of midline spinal cord hamartomas, one proved and one presumed, and discuss the possible embryologic errors responsible for their formation.
Case Reports
Case 1
A 16-month-old girl was referred to our institution because of a dimple in the skin of the lower, posterior portion of the neck. At physical examination, the skin surrounding the dimple appeared normal, and results of a neurologic examination were also normal. MR images showed a tract extending from the dimple through the subcutaneous tissues into the posterior vertebral elements (Fig 1A–C). At the C5 level, an abnormality was evident along the dorsal surface of the spinal cord. This lesion insinuated itself into the spinal cord and was isointense with the cord on T1-, proton density–, and T2-weighted images. The main preoperative diagnosis was that of a dermoid.
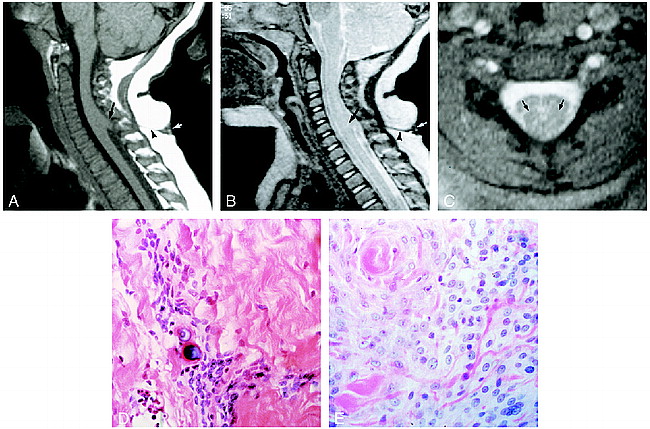
Case 1: 16-month-old girl.
A, Midline sagittal T1-weighted MR image shows a skin dimple (white arrow), a sinus tract (arrowhead) traversing the subcutaneous fat, and a midline spinal cord hamartoma (black arrow) slightly tethered posteriorly by the sinus tract. The lesion is isointense with the cord and merges imperceptibly with it.
B, Corresponding T2-weighted MR image shows the mass (black arrow) to be isointense with the spinal cord. The sinus tract (arrowhead) and skin dimple (white arrow) are well seen.
C, On an axial T2*-weighted MR image, the hamartoma (arrows) slightly splays the spinal cord and is isointense with it.
D, Microscopic examination shows aggregates of nonneoplastic meningothelial cells with occasional psammoma bodies surrounded by dense collagen (hematoxylin-eosin, original magnification ×250).
E, Higher-magnification view shows an overgrowth of meningothelial cells (hematoxylin-eosin, original magnification ×500).
At surgery, a fibrous tract extended into the dura and continued into the mass, which had insinuated itself into the spinal cord. The lesion was removed completely, together with the sinus tract. Microscopic examination of the dimple revealed skin containing unremarkable appendages and subcutaneous tissues displaying dense collagen and a nonneoplastic proliferation of meningothelial cells with scattered psammoma bodies (Fig 1D and E). Histologic examination of the spinal cord lesion showed neural tissue with reactive gliosis and portions of dense fibrous tissue admixed with a proliferation of meningothelial cells. The postoperative course was uneventful, and 3 years after surgery, the child was developing well. Findings on a follow-up MR study of the brain were normal.
Case 2
A 6-year-old girl was referred because of a skin dimple in the posterior and mid aspects of the neck. At physical examination, the skin surrounding the dimple was normal, and findings at neurologic examination were also normal. MR imaging showed a tract beginning at the dimple and extending into the posterior vertebral elements through the subcutaneous tissues. At the C4–C5 level, a small masslike lesion was noted on the dorsal surface of the spinal cord. The lesion blended with the spinal cord and was isointense with it on T1-, proton density–, and T2-weighted images (Fig 2).
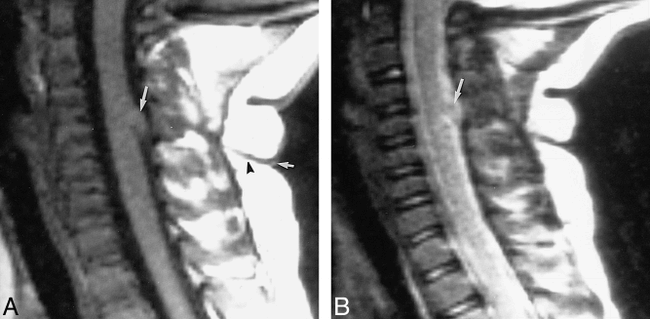
Case 2: 6-year-old girl.
A, Midsagittal T1-weighted MR image shows a skin dimple (short arrow), a sinus tract (arrowhead) traversing the subcutaneous fat, and a mass (long arrow) on the dorsal surface of the spinal cord. The lesion is isointense with the cord, inseparable from it, and appears to be tethered posteriorly (similar to case 1).
B, On midline sagittal T2-weighted MR image, the mass (arrow) is isointense with the spinal cord.
At surgery, the dimple was resected. It was connected to a well-formed fibrous stalk, which penetrated the cervical fascia and dura and was continuous with the mass. A lump of tissue, of similar appearance to the spinal cord, was present. The mass appeared densely adherent to the spinal cord and, because the patient was neurologically asymptomatic, no attempt at resection was made. The patient had an uneventful recovery and a follow-up MR study of the brain was normal. One year after surgery, the patient was developing well.
Discussion
Congenital midline spinal hamartomas are rare lesions composed of mature and well-differentiated elements in an abnormal location (1). These are distinct lesions and are different from those seen in patients with neurofibromatosis (NF). In patients with NF type 1, proliferations of glial cells, ganglion cells, disoriented axons, and vessels may be found within the spinal cord (2). These lesions have been termed vascular and neuroglial hamartomas, and they are different from the so-called midline spinal hamartomas reported here. Midline spinal hamartomas may present with a skin dimple, cutaneous angioma, subcutaneous mass, or normal overlying skin (1, 3). Hamartomas, which have a “meaty” appearance, may clinically simulate meningomyeloceles. Spinal hamartomas may be located in the thoracic, lumbar, and sacral regions or at the junctions between these segments. Most patients are neurologically intact. Midline spinal hamartomas are not associated with hydrocephalus, Chiari malformations, or open spinal dysraphisms (1, 3). Surgery is usually performed to prevent neurologic damage resulting from tethering of the cord (by dermal sinuses), to prevent infection, and/or to correct a cosmetic defect (1).
Histologically, spinal hamartomas have been found to contain nerves, fat, muscle, cartilage, bone, abnormal vessels, glands, synovial membranes, lymphoid tissues, and even urinary tract tissues (1). In our opinion, only those that contain local elements in a disorganized fashion can be considered pure hamartomas, while those that contain normal but ectopic tissues (such as glands, lymphoid tissue, urinary tract tissues) should be considered choristomas. The histologic examination in one of our patients showed dense connective tissues, meningothelial cell proliferation, and calcification, and we believe that the presence of these three tissues supports the diagnosis of hamartoma. The most important differential diagnosis is spinal teratoma. Teratomas are composed of elements from the three germinal layers, immature cells, and have a potential for malignant degeneration. Teratomas of the spinal canal are less common than the sacrococcygeal ones, and account for only 3% of all intraspinal masses in children (1, 3). They may be associated with spinal dysraphisms. Conversely, spinal hamartomas are composed of only ectodermal and mesodermal, mature and well-differentiated, elements, and do not contain elements from all three germ cell layers, as teratomas do. Epidermoids and dermoids are composed of only ectodermal derivatives. Unlike other spinal masses, spinal hamartomas do not carry a potential for excessive growth or malignancy, and the prognosis is excellent. A case of an unusual congenital mesenchymal proliferation (similar to a hamartoma) in the spine has been reported (4). In that patient, there was a large paraspinal subcutaneous mass that extended into the spinal canal. This lesion enhanced on MR imaging after contrast administration and, histologically, was found to be a giant cell angioblastoma with diffuse fibromatosis. Smooth muscle hamartomas may also be found in the skin and soft tissues adjacent to the spinal column (5). Additionally, skin hamartomas are usually not midline in location.
The mechanism for formation of spinal hamartomas is not known with certainty. The neural tube separates from the overlying ectoderm by the process of disjunction (6). After disjunction, the ectoderm over the neural tube undergoes midline fusion. At this stage, mesenchyme surrounds the neural tube (and the gap between it and the overlying ectoderm), eventually giving rise to muscles, bones, and dura. Because the neural tube closes early, the mesenchyme normally remains outside it. Incomplete disjunction of the neural tube from the overlying ectoderm may result in formation of the dermal sinus tract between skin and spinal cord. The presence of this tract prevents the mesenchyme from completely enclosing the dorsal aspect of the neural tube, resulting in formation of a spina bifida (a commonly reported finding in patients with midline spinal hamartoma). However, this mechanism alone does not explain the formation of spinal hamartomas, which contain well-differentiated mesenchymal elements. In cases of premature disjunction between the neural tube and ectoderm, the dorsal mesenchyme is allowed to enter the neural groove and come in contact with the inner aspect of the unfolded neural tube. The neural tube stimulates differentiation of these primitive mesenchymal precursors, inducing formation of lipomas. Therefore, it is possible that differentiation into other mature mesenchymal elements may also occur. We believe that the formation of spinal hamartomas probably represents an error of both incomplete and possibly premature disjunction. This observation may offer support for the embryogenesis of a spinal lipoma occurring in conjunction with a spinal neuromuscular hamartoma (7).
Although they are rare, midline spinal hamartomas seem to be a straightforward differential diagnosis from other masses on the basis of MR imaging findings. Teratomas are characterized by signal inhomogeneity and contrast enhancement. Most epidermoids have a signal intensity similar to fluid on both T1- and T2-weighted images. Most dermoids are bright on T1-weighted images and appear similar to intraspinal lipomas. In distinction to these, spinal hamartomas appear isointense with the spinal cord on all imaging sequences. We did not administer contrast material to these two patients and therefore do not know whether these lesions enhance. Although we have pathologic proof of the diagnosis in only one of our patients, the MR imaging findings were similar in both cases and characteristic enough that we believe the lesion in the second patient was also a midline spinal cord hamartoma. In addition, both patients were asymptomatic at the time of diagnosis and continue to be symptom-free.
Conclusion
Midline spinal cord hamartomas appear to be distinct entities that are being recognized with increasing frequency (1). Patients are usually asymptomatic or have only minimal neurologic deficits, and MR images show features that allow one to distinguish them from other intraspinal masses. Surgery may be indicated to avoid tethering of the spinal cord, to avoid infection, and/or to correct cosmetic lesions. The prognosis for these patients is excellent.
Footnotes
1 Address reprint requests to Mauricio Castillo, MD, Department of Radiology, University of North Carolina School of Medicine, CB# 7510, Chapel Hill, NC 27599.
References
- Received December 1, 1998.
- Accepted after revision February 16, 1999.
- Copyright © American Society of Neuroradiology