Abstract
BACKGROUND AND PURPOSE: A new option in the treatment of recurrent malignant glioma is surgical placement of chemotherapy-laden biodegradable wafers. We describe the CT and MR appearance of chemotherapy wafers in patients after surgery for recurrent malignant glioma.
METHODS: Eighteen patients had carmustine (BCNU) wafers implanted during reoperation for malignant glioma; three patients had empty, placebo wafers placed. The 21 patients had a total of 22 CT and 57 MR imaging studies. Repeat CT studies were conducted for up to 6 months, the MR studies for up to 1 year. Examinations were evaluated for attenuation on CT scans, signal abnormalities on MR images, and changing appearance during the follow-up period. Enhancement characteristics were also assessed.
RESULTS: On CT scans, 13 of 16 acute (<7 days) cases showed linear high-attenuation wafers, with three showing low attenuation. On MR images, all T1 and T2 studies performed in the acute stage showed decreased signal of the wafers. Eight of 15 studies showed a transient increase in T1 only at about 2 months. Wafers decreased in conspicuity on both CT and MR studies after 2 months. The wafers did not enhance. One postoperative tumor showed a transient increase in edema and increased enhancement at 5 weeks. The presence or absence of BCNU within the wafers did not change their appearance.
CONCLUSION: BCNU wafers have a characteristic appearance: in the first 7 days after implantation they are linear, usually of increased attenuation on CT scans, and always show decreased signal on MR images; they do not enhance, and become less conspicuous after 2 months.
Controlled-release polymers are designed to deliver chemotherapeutic agents directly into resection sites after gross total removal of recurrent malignant gliomas. The system allows prolonged local exposure with limited systemic exposure. A recent study of this technique evaluating 222 patients showed increased survival in patients treated with carmustine (BCNU) wafers versus those who received only placebo wafers (1). Since BCNU wafers are now approved by the FDA and will probably be encountered more frequently, radiologists should be familiar with their imaging features.
The purpose of this retrospective study was to describe the MR and CT characteristics of biodegradable wafer polymers implanted at the time of reoperation for recurrent malignant gliomas.
Methods
Postoperative CT and MR imaging studies from 21 patients (13 men and eight women; average age, 51 years) who had undergone repeat surgery for malignant glioma were reviewed retrospectively. In all subjects, malignant glioma was diagnosed by histopathologic findings at the time of initial surgery.
Eight patients were enrolled in a multicenter, double-blind, randomized clinical trial to determine the effectiveness of BCNU-impregnated wafers. Five patients received wafers with BCNU and three patients received placebos. Postoperative imaging was not controlled. The ordering physicians preferred MR imaging, but CT was used when the patient was unstable or unable to tolerate the longer MR examination. One patient had CT only and two patients had MR only. Five patients had both MR and CT. Among the patients who were given placebos, two had CT and MR, and one had MR only. The results of this trial are reported elsewhere (1).
Another 13 patients received wafers with BCNU after the original trial was completed. The wafers are composed of a copolymer of polycarboxyphenoxypropane and sebacic acid in a 20/80 ratio. BCNU is incorporated into a hydrophobic matrix and is thereby protected from hydrolysis.
To be a candidate for insertion of wafers, a patient had to have had a previous resection of either a histologically proved anaplastic glioma or glioblastoma multiforme followed by external-beam radiation therapy. Each patient had to have a recurrent tumor that was a unilateral enhancing mass and a Karnovsky performance score of 60 or above (a Karnovsky score of 60 indicates the ability to function independently). In each case, the surgeons made an independent determination that another tumor resection was indicated. Wafers were not placed in patients who had not had previous resection. At surgery, frozen-section histopathologic examination confirmed the malignant nature of each tumor and excluded radiation necrosis. In each case, gross-total or near gross-total resection was achieved, as determined by microsurgical inspection of the tumor bed.
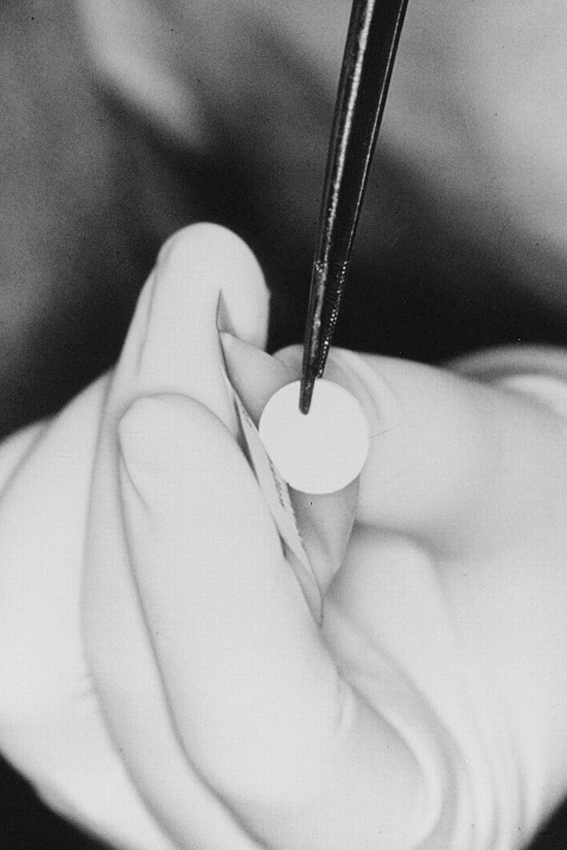
After hemostasis was achieved, the wafers were placed in the tumor bed. Each wafer was 1.4 cm in diameter and 1 mm thick (Fig 1). In each case eight wafers were placed in the tumor bed and spaced evenly. In some cases the wafer edges were separated by as much as 5 mm, and in other cases the wafer edges were in contact with one another, depending on the size of the tumor resection cavity. In no cases were the wafers overlapping. The wafers were placed so as to cover the maximum amount of surface area in the walls of the resection cavity. In most cases the wafers were covered with sheets of oxidized regenerated cellulose (Surgicel, Johnson & Johnson, New Brunswick, NJ) to promote adhesion to the wall of the tumor resection cavity. Wafer contact with large vascular structures and openings into ventricles was avoided.
Biodegradable wafer: diameter, 1.4 cm; thickness, 1 mm
CT or MR examinations or both were performed in the first 24 hours after surgery when the patients were stable. Subsequent studies were usually obtained at least once every 2 months. The studies were then evaluated in concert by three neuroradiologists. CT attenuation and MR signal characteristics of the wafers, the operative bed, and the surrounding parenchyma were determined along with enhancement characteristics and any changes from prior studies. In particular, we evaluated the subjective conspicuity of the wafers. Evaluation was from film only. Evaluators were able to agree on the findings. MR studies were all performed on a 1.5-T system. Imaging parameters for T1-weighted studies were 700/11/2 (TR/TE/excitations), 5-mm-thick sections with a 1-mm gap, and a 512 × 192 matrix; for T2-weighted studies, parameters were 4500/17,119/2 (TR/TE/TEeff/excitations), 7-mm-thick sections with a 1.5-mm gap, and a 512 × 192 matrix. Routine studies included sagittal and axial T1-weighted images, axial T2-weighted images, and contrast-enhanced axial and coronal T1-weighted images. For patients weighing less than 150 lbs, 15 mL of gadopentetate dimeglumine was used; for those over 150 lbs, 20 mL was used. CT examinations were all done with 7- or 10-mm-thick sections. When used, contrast material was 100 mL of 300 mg I/mL.
Results
On CT and MR studies the BCNU wafers had a characteristic appearance acutely (<7 days) as a linear object just greater than 1 cm in length and 1 to 2 mm in thickness. No enhancement of the wafers was seen in this early stage. In the three patients who received placebos, the empty wafers had a similar appearance to those with BCNU. There was no gross hemorrhage or infection associated with any of the surgical procedures.
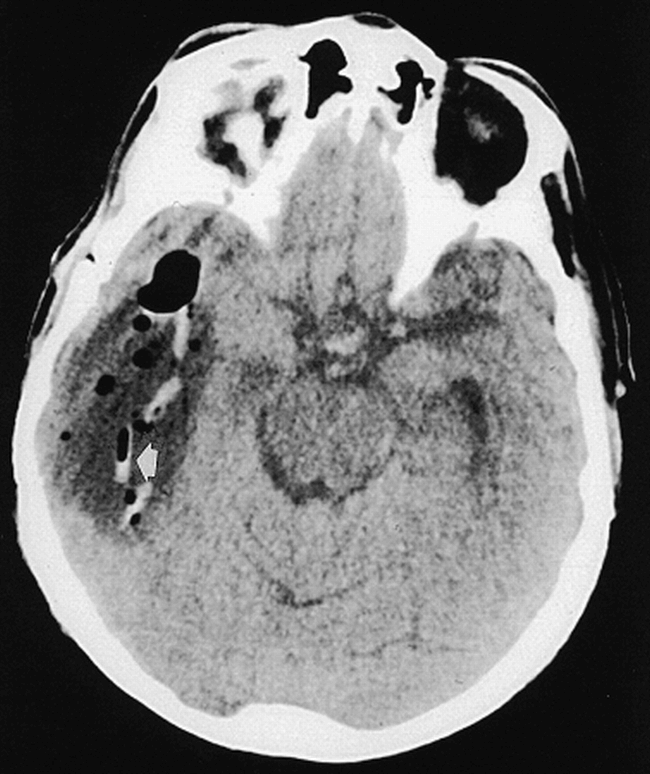
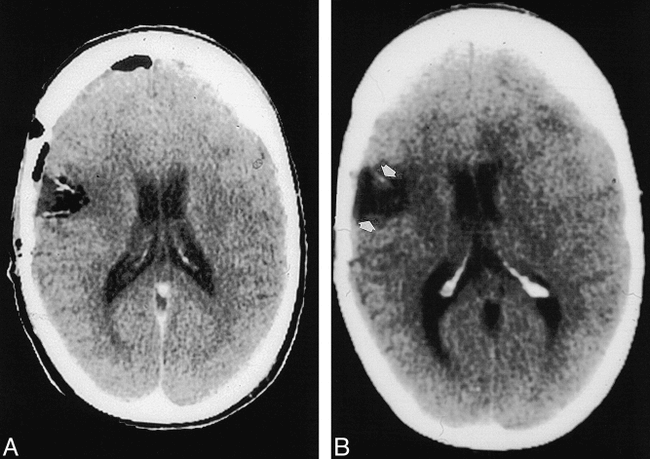
Sixteen of the 21 patients had CT, for a total of 22 CT studies. The maximum follow-up time was 6 months. On the acute scan (<7 days), 13 (81%) of the 16 cases showed high attenuation (Fig 2); three (19%) showed low attenuation. (If any portion of a wafer had high attenuation it was regarded as a high-attenuation wafer only.) All the wafers showed decreased visibility and were poorly seen after 3 months (Fig 3).
CT scan obtained less than 24 hours after surgery shows four clearly defined wafers. One wafer has an unusual appearance, half-increased attenuation and half-decreased attenuation (arrow)
CT study.
A, Less than 24 hours after surgery, two well-defined wafers are seen.
B, Follow-up study at 2 months shows faint wafer remnants (arrows).
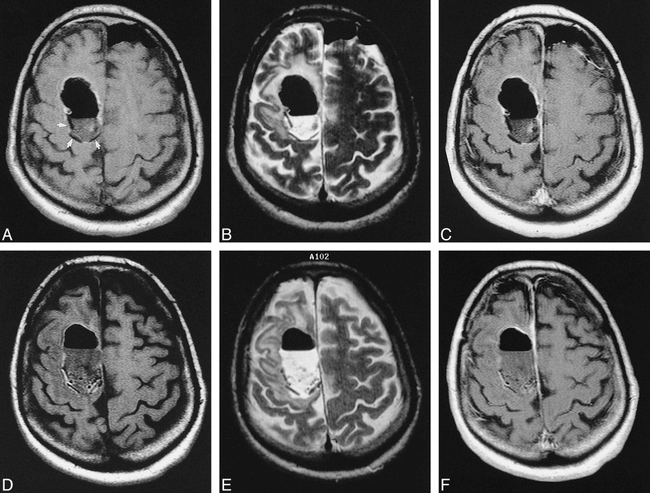
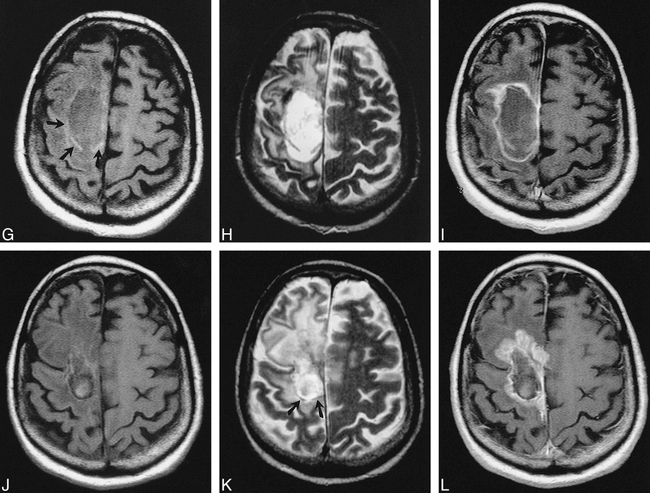
Nineteen of the 21 patients had MR examinations, for a total of 57 MR studies. All subjects had T1-weighted images and 30 had T2-weighted sequences in addition (limited studies requested by clinical service). Acute MR studies showed low signal on all T1-weighted (19 cases) and T2-weighted (eight cases) sequences (Fig 4A-F). A transient increase in T1 signal was seen in eight of 15 cases at around 2 months (Fig 4G-I). T2 sequences showed gradual reduction in conspicuity and no evidence of increased signal. Wafers were poorly seen on both T1- and T2-weighted sequences after 2 months (Fig 4J-L). Maximum MR imaging follow-up was 1 year.
←MR study.
A–C, T1-weighted (A), T2-weighted (B), and contrast-enhanced T1-weighted (C) images less than 24 hours after surgery show three well-defined, decreased-signal wafers (arrows, A). There is no enhancement; subtle increased T1 signal is visible in the operative bed.
D–F, T1-weighted (D), T2-weighted (E), and contrast-enhanced T1-weighted (F) images 1 week after surgery show resolving postoperative changes with decreased edema, mass effect, and intracranial gas. The wafers are still easily seen but are somewhat less distinct.
G–I, T1-weighted (G), T2-weighted (H), and contrast-enhanced T1-weighted (I) images 2 months after surgery. The wafers have increased signal on T1-weighted image (arrows, G) and less prominent decreased signal on T2-weighted image. There is enhancement of the operative bed and in the parenchyma.
J–L, T1-weighted (J), T2-weighted (K), and contrast-enhanced T1-weighted (L) images 5 months after surgery show increased T2 signal abnormality in the brain. Incomplete wafer remnants are seen on T2-weighted image only (arrows, K). After contrast administration, marked increased enhancement is seen in operative bed and parenchyma (L).
In one case, a transient increase in edema and enhancement was seen on MR images at 5 weeks. Follow-up at 8 months showed no enhancement or mass effect at the site of surgery; however, enhancing nodules were present on the contralateral side. All other studies were essentially stable or showed tumor progression on follow-up studies.
Discussion
Malignant gliomas have a notoriously poor response to therapy. The blood-brain barrier restricts delivery of chemotherapeutic agents and there are complications from systemic exposure to these agents. Most tumors recur locally, and the most effective chemotherapeutic agent for malignant glioma is BCNU (1). A biodegradable polymer (Gliadel) has been developed to deliver BCNU directly to the operative bed at surgery. Gliadel is now FDA approved, and radiologists should become familiar with its MR and CT appearance.
Brem et al (2) reported the first study involving BCNU wafers in patients with recurrent tumors. After tumor removal, up to eight wafers were placed on the resection surface. Three different doses of BCNU were used. Twenty of 21 cases were followed up by CT and one patient was followed up by MR imaging. Studies were obtained at 1 to 2 days, 14 days, and 49 days. No systemic toxicity was reported. The top median survival time from implantation was 65 weeks. At reoperation (for increasing mass effect and symptoms) or autopsy, disk remnants were present but without BCNU or polyanhydride bonds. On CT scans, 13 of their 21 cases had perioperative areas of enhancement, which mostly resolved in 7 weeks. This finding is frequently seen after neurosurgical neocortical resection with or without neoplasms (3). There was minimal mass effect. The wafers were seen as “bright white lines” along the operative bed.
Grossman et al (4), in a separate study with biodegradable wafers, showed that radiolabeled compound was extensively distributed in the brain at 3 days, but only directly adjacent to the polymer 1 to 3 weeks after insertion. Brem et al (5), in a study comparing imaging and histologic findings in monkey brains, found no difference in the CT/MR appearance in 10 brains between wafers with and without BCNU: there was always increased attenuation on the CT scans and decreased signal on the MR images. Microscopic analysis showed a 1- to 2-mm rim of increased necrosis in the BCNU-wafer group and greater inflammatory response with gliosis and neovascularization. There was less phagocytic activity and more intimal hyperplasia in the BCNU group. By 10 weeks there was a chronic inflammatory response.
A large, double-blind, randomized, controlled study involving 222 patients has also been reported (1). The 110 patients treated with BCNU-impregnated wafers had a median survival of 31 weeks, compared with 23 weeks for patients treated with empty wafers at reoperation. Each treated wafer contained 7.7 mg of BCNU, with patients receiving a maximum of eight wafers, or 62 mg. Compared with systemic delivery, this represents an increase of 113-fold in brain exposure to BCNU adjacent to the tumor resection bed. No significant adverse reactions were encountered. Imaging findings were not reported.
In our study, 19 patients had marked decreased signal on T1- and T2-weighted images in the acute phase, presumably due to the hydrophobic wafers. The transient increase in T1 signal seen in half the cases at 2 months was associated with hypo- to isointense T2 signal. Thus, the increased T1 signal may represent proteinaceous material or the interval development of blood in the operative bed. The wafers never enhanced with administration of contrast material. In a single case, a transient local increase in enhancement and mass effect was seen at 5 weeks; however, an 8-month follow-up study showed resolution of the enhancement and mass effect in the operative region. This case probably represents a more serious local reaction to the toxic effects of BCNU. A similar flare response has been described in patients receiving intralesional immunotherapy (6). Unfortunately, the 8-month follow-up study also showed contralateral enhancing nodules.
Three of the acute cases showed marked attenuation of the wafer on CT scans, presumably because of gas trapped within the wafer of underneath surgicel.
The major limitations of our study are related to its retrospective nature and to the absence of control of the type and timing of follow-up studies. Also, because studies were not always done in three planes, partial volume effects may have hindered visualization of the wafers.
Conclusion
Biodegradable polymers with BCNU have a characteristic appearance on CT and MR studies. Acutely, they appear as wafers on edge or as linear objects, usually bright on CT scans and always dark on MR images. They become less conspicuous over time, although there is often a transient increase in T1 signal seen subacutely. A transient local reaction with increase in enhancement and mass effect is rare.
Footnotes
1 Presented in part at the annual meeting of the American Society of Neuroradiology, Toronto, May 1997.
2 Address reprint requests to Jordan M. Prager, MD, Department of Radiology, Evanston Northwestern Healthcare, Evanston Hospital, 2650 Ridge Ave, Evanston, IL 60201.
References
- Received January 4, 1999.
- Copyright © American Society of Neuroradiology