Abstract
BACKGROUND AND PURPOSE: Although there have been several reports on postoperative MR imaging of the sella, immediate postoperative changes (usually within 3 days) have not been extensively analyzed. The purpose of this study was to establish the value of early postoperative MR imaging in differentiating residual tumor from postoperative surgical changes in the sella after transsphenoidal resection of pituitary adenomas.
METHODS: Eighty-three patients with surgically proven pituitary adenomas (32 nonfunctioning, 24 prolactin-secreting, 22 growth hormone–secreting, and five prolactin- and growth hormone–secreting tumors) were studied prospectively. All patients underwent dynamic MR imaging within 7 days after surgery. We analyzed the postoperative MR images by focusing on changes in the pituitary gland, signal intensity, resorption of implanted material, and visibility of residual tumor. The patients were divided into four groups according to enhancement pattern of the postoperative pituitary mass: no enhancement, nodular enhancement, peripheral rim enhancement, and a combination of nodular and peripheral rim enhancement.
RESULTS: Postoperative changes included resorption of implanted material and reexpansion of the pituitary gland. In 22 patients, residual tumors were found, and all patients showed nodular or combined enhancement. The residual tumors were confirmed by immediate reoperation in three patients, by hormonal assay and follow-up MR images in 11 patients with functioning adenomas, and by growth of the tumor on follow-up MR images in eight patients with nonfunctioning adenomas. Forty-eight patients showed no enhancement and 13 patients showed peripheral rim enhancement.
CONCLUSION: Early postoperative dynamic MR imaging after transsphenoidal resection in pituitary adenoma is very effective in differentiating residual tumor from postoperative surgical changes.
MR imaging with dynamic enhanced study is accepted as the most sensitive imaging method for the evaluation of pituitary adenomas and the normal pituitary gland, because time of peak enhancement of adenomas is slightly later than that for the normal pituitary gland. MR imaging is used frequently in the postoperative follow-up of a pituitary adenoma, particularly a nonfunctioning adenoma (1–4). In most studies, however, the period for postoperative follow-up MR imaging has been several months after surgery (1). Therefore, it is sometimes difficult to differentiate residual tumor from postoperative fibrosis, surgical packing material, or even a normal pituitary gland. There have been several reports about immediate postoperative MR, which may establish the baseline of postoperative status and immediately detect postoperative complications (2, 3). These reports, however, have dealt primarily with postoperative physiological changes of the sella, such as changes of normal pituitary gland, pituitary stalk and optic chiasm, and implanted materials.
Most residual pituitary tumors are located in areas where surgery is difficult, such as the cavernous sinus, the suprasellar cistern in very firm tumors, and the posterior clivus. However, some residual tumors are not in these areas. Early detection of these residual tumors, for which surgery is not difficult, can provide the opportunity for an immediate second operation (via the same transsphenoidal approach) before the development of adhesion at the operation site. Although intraoperative MR imaging can be helpful in resection of high-grade gliomas, it is not yet popular (5). The purpose of this study was to evaluate MR findings of usual postoperative changes of the sella in the early period after transsphenoidal resection and, therefore, to detect residual tumor, allowing for an immediate second operation, and to establish a postoperative baseline in late postoperative MR imaging.
Methods
We prospectively studied 83 patients with pituitary adenomas that were surgically proven (52 women and 31 men, aged 15 to 71 years [mean age 41.1 years]). Of the 83 patients studied, 16 had microadenomas and 67 had macroadenomas. Thirty-two patients had nonfunctioning pituitary adenomas and 51 patients had functioning adenomas. Among the 51 patients, 24 had prolactin-secreting adenomas, 22 patients had growth hormone–secreting adenomas, and the remaining five patients had a combination of prolactin- and growth hormone–secreting tumors.
All these patients had transsphenoidal resection of the tumor. The resection cavity was covered with autologous fat encased by oxygenated cellulose and fibrin glue in 69 patients. In 14 patients, nothing was used as a packing material to fill the resection cavity. The sellar floor defect was reconstructed by using small fragments taken from the osseous nasal septum.
All patients underwent preoperative MR studies and early postoperative MR imaging, including dynamic enhanced coronal T1-weighted images. Preoperative and postoperative MR imaging studies were performed using a 1.5-T superconducting unit with a circular polarized head coil. The initial imaging sequence included sagittal T1-weighted images with the following parameters: 5-mm-thick slices, no skip, 400–700/14–16 (TR/TE), 256 × 192-pixel matrix, 18-cm field of view (FOV). This was followed by coronal long-TR imaging using fast spin-echo technique: 3-mm-thick slices, 1-mm skip, 3500–4000/95–100, 256 × 256-pixel matrix, and 18-cm FOV.
Dynamic MR imaging was performed using a T1-weighted sequence: 400/14–16/1 (TR/TE/excitation), 192 × 256 rectangular matrix. It took 88–90 seconds to obtain three contiguous images in a data acquisition. The FOV was 16 cm, and three contiguous sections with 3-mm thickness and no interslice gap were obtained with a multisection technique. Before the administration of gadopentetate dimeglumine, T1-weighted spin-echo images were obtained in the coronal planes. After a rapid injection (2 mL/s) of gadopentetate dimeglumine (0.1 mmol/kg body weight), dynamic MR images were obtained every 80–90 seconds in the coronal plane for 3–4 minutes. After dynamic imaging, routine T1-weighted spin-echo images were obtained.
The early postoperative MR imaging examinations were performed within 3 days after resection of the pituitary adenoma in 68 patients. In 15 patients, initial postoperative MR studies were obtained between the fourth and seventh days. In all patients, late postoperative follow-up examinations (1 to 4) were performed during the following 6–48 months. Hormonal assay was performed with postoperative MR imaging for those who had functioning pituitary adenomas.
The early postoperative pituitary masses were considered to represent implant materials, reexpanded normal pituitary gland, and/or postoperative hemorrhage when the tumor had been surgically removed and the resection cavity was filled with fat only. This information was obtained from correlation with operative reports. Signal intensity and time course of resorption of the implants were analyzed.
We analyzed the postoperative MR images, placing special emphasis on the enhancement pattern of the postoperative pituitary mass, signal intensity and resorption of implanted material, and residual tumor on dynamic enhanced study. We divided the patients according to the enhancement pattern of the postoperative pituitary mass: 1) no enhancement, 2) peripheral rim enhancement, 3) nodular enhancement, and 4) combined enhancement. No enhancement pattern meant that there was no enhancement in the postoperative pituitary mass where the pituitary tumor was located on the preoperative image after intravenous administration of contrast material. Peripheral rim enhancement pattern meant that there was contrast enhancement in the periphery of the postoperative pituitary mass. Nodular enhancement pattern meant that nodular enhancement was demonstrated in the postoperative pituitary mass. Finally, combined enhancement pattern meant that there was coexistence of peripheral rim and nodular enhancement.
We confirmed a residual tumor by an immediate second operation, hormonal assay, and follow-up MR imaging in functioning tumors. Follow-up MR imaging only was used to confirm residual nonfunctioning tumors.
Results
On the early postoperative MR scans, 69 patients who underwent transsphenoidal resection and intrasellar packing with fat only showed high signal intensity on the precontrast T1-weighted image where the tumor was located (Figs 1 and 2). Follow-up (6 months) MR imaging showed this implanted fat remained with partial resorption (Fig 1). There was no intrasellar packing in 14 patients. Sixteen patients showed reexpanded normal pituitary gland. Ten of the 16 patients with reexpanded pituitary gland underwent early postoperative MR scan within 4 to 7 days after operation.
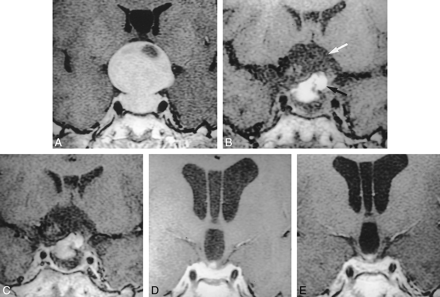
No enhancement of early postoperative sella in a 29-year-old woman with nonfunctioning tumor.
A, Preoperative image of the sella (TR/TE/excitations = 550/12/2) shows pituitary tumor with suprasellar extension. Normal pituitary gland cannot be seen because of compression by the tumor.
B, Immediate 1-day postoperative image (400/16/1) before contrast material infusion shows pituitary mass composed of fat (black arrow) and hemorrhage (white arrow) at the operative site.
C, After contrast infusion, there is no abnormal enhancement in the pituitary mass.
D, After 6 months, the normal pituitary gland is reexpanded (550/12/2).
E, There is no change on 30-month follow-up MR image (400/12/2).
In 13 of 67 macroadenomas, there was isointensity, except for fat signal intensity in the tumor area. In these patients, this isointensity was seen as low signal intensity on T2-weighted image, suggestive of intracellular deoxyhemoglobin (Fig 2). In some patients, the follow-up (a few days postoperatively) T1-weighted image showed the isointense signal had changed to high signal intensity, suggesting a methemoglobin component of the blood. The fluid-fluid level, which was suggestive of hemorrhage, was seen on T2-weighted axial images in some of these patients. Two of these 13 patients had an immediate second operation because of the severe mass effect, and hemorrhage was confirmed.
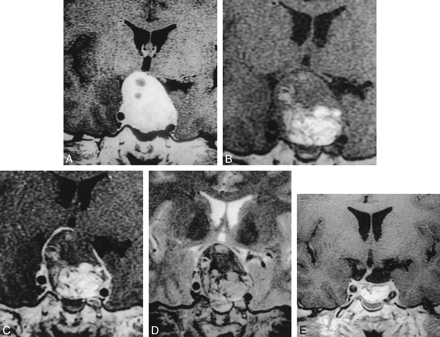
Peripheral enhancement of an early postoperative sella in a 35-year-old man with nonfunctioning tumor.
A, Preoperative image of the sella (TR/TE/excitations = 700/12/2) shows pituitary tumor with suprasellar extension. Normal pituitary gland cannot be seen because of compression by the tumor.
B, Immediate 2-day postoperative image before contrast infusion (400/16/1) shows pituitary mass composed of fat and hemorrhage at the operative site.
C, After contrast infusion, there is a peripheral enhancing rim around the postoperative pituitary mass.
D, Immediate postoperative T2-weighted image (3500/95/2) shows hemorrhage as low signal intensity suggesting intracellular deoxyhemoglobin.
E, After 6 months, the hemorrhage is totally absorbed (500/9/2).
We divided the patients according to the enhancement pattern on early postoperative dynamic enhanced MR imaging (Table). Forty-eight patients showed no enhancement on dynamic MR imaging (Fig 1). Peripheral rim enhancement was seen in 13 patients (Fig 2), and 18 patients showed nodular enhancement (Fig 3). A combination of nodular and peripheral rim enhancement was seen in four patients (Fig 4). Residual tumor was confirmed in 22 patients (18 with nodular enhancement and four with combined pattern of enhancement). In 14 patients with functioning adenomas, the residual tumor was confirmed by an immediate second operation in three patients, and by follow-up hormonal assay and MR images in the other 11 patients. In eight patients with nonfunctioning adenomas, residual tumor was confirmed by follow-up MR imaging only. In the three patients who had an immediate second operation, the residual tumor corresponded to the site that showed nodular enhancement on the immediate postoperative MR images (Fig 5). In 13 patients with peripheral rim enhancement, there were no residual tumors. This was confirmed by follow-up hormonal assay and MR imaging in seven patients with functioning tumors and by follow-up MR imaging in six patients with non-functioning tumors. Forty-eight patients with no enhancement had no evidence of residual tumor on follow-up MR images and hormonal assay.
Enhancement of early postoperative sella
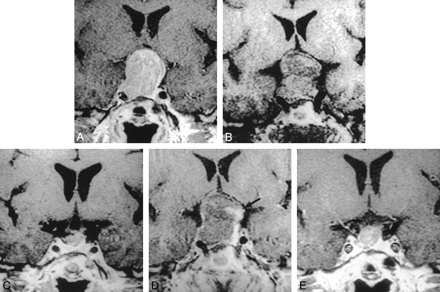
Nodular enhancement of early postoperative sella in a 62-year-old woman with nonfunctioning tumor.
A, Preoperative image of the sella (TR/TE/excitations = 550/12/2) shows pituitary tumor with suprasellar extension.
B, Immediate 1-day postoperative image before contrast infusion (400/14/1) shows pituitary mass composed of hemorrhage at the operative site. Intrasellar packing material (fat) was not used in this patient.
C, After contrast infusion, there is nodular enhancing region at the left periphery of the pituitary mass (arrow).
D, After 6 months, a small residual tumor is seen (700/12/2).
E, This residual tumor is slowly growing on 24-month follow-up MR images (433/10/2). This case was diagnosed by growth of the tumor on follow-up MR imaging.
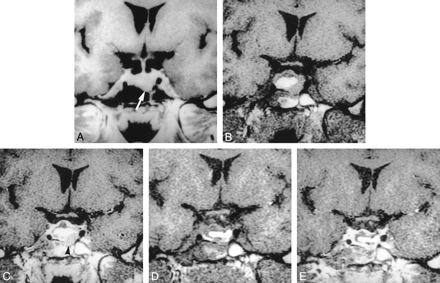
Combined enhancement of early postoperative sella in a 60-year-old man with nonfunctioning tumor.
A, Preoperative image of the sella (TR/TE/excitations = 600/16/1) shows pituitary tumor with suprasellar extension.
B, Immediate 1-day postoperative image before contrast infusion (400/14/1) shows pituitary mass composed of fat (white arrow) and hemorrhage (black arrow) at the operative site.
C, After contrast infusion, there are nodular enhancing regions (arrows) at both lateral portions of the postoperative pituitary mass with peripheral enhancing rim.
D, After 6 months, residual tumor was seen with increase in tumor size (550/12/2).
Immediate MR scan following surgery confirmed residual tumor in a 27-year-old woman with growth hormone-secreting tumor.
A, Preoperative image of the sella (TR/TE/excitations = 550/12/2) shows pituitary tumor in the inferior portion of the pituitary gland (white arrow).
B and C, Immediate 1-day postoperative image before and after contrast medium infusion (400/14/1) shows pituitary mass composed of fat and nodular enhancing tissue (arrowhead).
D, A second operation was performed to remove the residual tumor, and this scan (400/14/1) was performed 2 days following surgery.
E, After contrast medium infusion, there is no abnormal enhancing lesion in this area (550/12/2).
Discussion
Pituitary adenomas are common lesions, accounting for approximately 10% to 15% of all primary intracranial neoplasms and between one third and one half of all sellar/juxtasellar masses (6). Endocrinologically active adenomas account for 75% of cases (6). Transsphenoidal microsurgery has been the most commonly used procedure for pituitary adenoma because of its safety and effectiveness (7). Since the incidence of invasive pituitary adenoma is not uncommon, complete surgical removal is not possible in all cases. In hormonally active pituitary adenomas, persistent or recurrent hypersecretion of hormone indicates residual or recurrent tumors and follow-up examination is required. Follow-up imaging study may be necessary in instances of nonfunctioning adenomas or suspected residual tumor at surgery.
To detect a residual tumor after surgery, one needs to know the usual postoperative changes in the sella after transsphenoidal pituitary resection. There have been several reports on postoperative changes of the sella after transsphenoidal resection of a pituitary adenoma (1–3). These reports primarily mentioned the normal postoperative changes of the postoperative sella and intrasellar packing material. They did not emphasize detectability of a residual tumor on follow-up MR imaging because it was difficult to differentiate a residual tumor from a normal gland, implanted material, or postsurgical granulation tissue on follow-up MR images more than 6 months after surgery. Dina et al (2) mentioned a residual tumor on early postoperative MR images, although the number of residual tumors were few. Rodriguez et al (3) also reported early postoperative changes of the sella, but since they did not deal with residual tumors, they did not determine how a residual tumor could be detected on early postoperative MR imaging. In our study, a relatively large population of patients underwent early postoperative MR imaging within 7 days after surgery, and follow-up MR imaging every 6 months.
The usual postoperative changes of the postoperative sella were well described in several reports (1–3). These changes included the reexpansion of normal pituitary gland, thickening of the pituitary stalk, swelling of the optic chiasm, and resorption of implanted material. In our study, reexpansion of the pituitary gland and resorption of the implanted material were studied. However, our findings were different than other reports, because our findings were based on early postoperative images.
Reexpansion of the normal gland was seen in 16 patients with four microadenomas and 12 macroadenomas. Steiner et al (1) reported that postoperative reexpansion of the gland was seen in 12 of 25 patients; however, the high incidence might be attributed to late postoperative imaging. In the early postoperative period, Dina et al (2) reported that two pituitary masses were unchanged, and three increased in height compared with the preoperative scans. The remaining five masses were decreased in height, but only minimally compared with the height of the preoperative mass (5%–33% decrease in height), although they did not mention reexpansion of the gland. Several reports on CT studies in the early postoperative period demonstrated a lack of change in overall size of the pituitary mass (8–10). Follow-up CT studies have shown a decrease in size of the pituitary mass during the 3 or 4 months following surgery. Several explanations have been offered to explain this phenomenon, including resorption of the packing, overpacking of the tumor bed, postoperative hemorrhage, persistent tumor or tumor capsule, and adhesions between the diaphragma sellae or tumor and brain tissue (11). In our patients, we only used fat as packing material because we could easily detect fat with high signal intensity on precontrast T1-weighted image. There was obviously another material besides fat within the postoperative pituitary mass. This material was isointense on precontrast T1-weighted image and hypointense on T2-weighted image. This signal intensity corresponded to intracellular doxyhemoglobin, and this acute stage of hemorrhage was well correlated with the time interval between surgery and the early postoperative MR imaging. In some of these patients, the fluid-fluid level suggestive of hemorrhage was seen on axial T2-weighted images. In addition, follow-up MR imaging was available in some patients and showed hyperintense signal on precontrast T1-weighted images, which was suggestive of methemoglobin in the subacute stage of hemorrhage. In two patients, immediate reoperation was performed because of severe mass effect from this pituitary mass, and hemorrhage was confirmed. Therefore, we believe that the isointense signal in the postoperative pituitary mass was mainly composed of postoperative hemorrhage, even if small amounts of serosanguineous fluid were mixed.
In our study, the number of patients who showed reexpansion of the gland was less than in other studies, because our study was performed early in the postoperative period and there was insufficient time for the gland to reexpand. The postoperative pituitary mass had the same volume as the preoperative study due to the implanted material and postoperative hemorrhage. Nevertheless, 16 of our patients showed reexpansion of the gland. However, early postoperative MR imaging was performed in 10 patients between 4 and 7 days after surgery; the number of reexpanded glands might be reduced if these patients underwent surgery within 3 days after transsphenoidal resection. Only four patients with microadenoma showed reexpansion of the gland after surgery. This might be due to the mass effect of the implanted material.
When a pituitary adenoma is removed transsphenoidally, the resection cavity in the sella is packed with either gelatin foam or autologous fat to achieve hemostasis or to prevent leakage of cerebrospinal fluid. These packing materials may appear as endosellar masses in the postoperative follow-up examination (1, 2). We used autologous fat only as the implanted material and this can be readily differentiated from surrounding structures on precontrast T1-weighted images. On follow-up MR images 6 months after surgery, signal intensity of the implanted fat could be seen with little or no change in the volume. Much of the implanted fat was resorbed in the follow-up image taken 1 year after surgery.
We divided our patients into four categories based on the contrast enhancement pattern of the postoperative pituitary mass. No enhancement meant that the postoperative pituitary mass was composed of fat, retained fluid, or hemorrhage. The second category was peripheral rim enhancement, meaning there was marginal enhancement in the postoperative pituitary mass after contrast material administration. Steiner et al (1) reported that the periphery of the gelatin foam implant demonstrated a circular rim of contrast enhancement, which was seen in all cases except one. This patient underwent postoperative MR imaging 5 days after surgery. Steiner suggested this peripheral enhancement was very likely caused by granulation tissue, which was seen several months after surgery. Peripheral enhancement in our study focused on early postoperative findings. A thin enhancing rim, as seen on CT, has been referred to as a persistent tumor capsule (9, 12, 13). Dina et al (2) also reported this peripheral enhancing rim in three of 10 patients in the early postoperative period, and it could not be determined on a single imaging study whether this tissue represented a residual tumor, tumor capsule, or pituitary gland. Dina et al (2) suggested that on follow-up studies, this tissue assumed a more normal pituitary size and shape within the confines of the sella 4 to 9 months later. In our patients with peripheral rim enhancement, there was no case that showed increased size on follow-up MR images or elevation of hormone level on follow-up laboratory studies. In addition, on follow-up MR images, peripheral rim enhancement disappeared and normal gland reexpanded in the pituitary fossa. Therefore, we regarded this peripheral rim enhancement as compressed normal pituitary gland or pseudocapsule around the tumor, although tissue analysis was not available.
We believe that the nodular portion of an early postoperative pituitary mass that showed nodular and combined enhancement was residual tumor, because the normal gland did not fully reexpand in this early period, as described above, and this nodular portion showed the same signal intensity and contrast enhancement compared with that of the preoperative adenoma. We confirmed these residual tumors by an immediate second operation, hormone assays, and follow-up MR imaging in functioning tumors, and by follow-up MR imaging only in nonfunctioning tumors. We also concluded that the portions that showed nodular enhancement and no growth on follow-up MR images were also residual tumors. If this portion was not a tumor, it could be a normal gland or granulation tissue. In this early period, the pituitary gland had not yet reexpanded, so differentiation from a residual tumor was possible. Likewise, granulation tissue could not have developed yet, so differentiation from a residual tumor was also possible. Steiner et al (1) and Dina et al (2) also reported residual tumors in their studies. However, they did not mention differentiation from other structures or materials in detail because there was no imaging in this early period or because there were small numbers of residual tumors (1, 2). The high incidence of residual tumors could be attributed to the prevalence of large, infiltrating macroadenomas in our patients. In our study, most residual tumors were found in areas where surgery is difficult, such as the cavernous sinus, suprasellar cistern (in very firm tumors), and posterior clivus.
It is interesting to note that there was only one case of nodular enhancement seen in a microadenoma case, whereas there were 21 cases of nodular or combined enhancement in macroadenomas. This would imply that postoperative imaging is far more useful in evaluating postoperative macroadenoma cases rather than in cases of microadenoma. However, in cases of microadenoma, most surgeons do not leave a residual mass, which is why there was only one case of microadenoma with residual mass in our study. Most pituitary microadenomas are functioning tumors. Therefore, postoperative imaging for detection of a residual mass is also useful in microadenomas, because residual mass still acts as a functioning tumor.
Conclusion
Early postoperative MR imaging is useful in the detection of residual tumor. Since the early postoperative sella retains its preoperative volume, one can easily differentiate residual tumor from a normal gland, implanted materials, or postsurgical granulation tissue. In addition, early postoperative MR images can be an excellent baseline if radiation therapy is necessary for treatment of residual tumor or recurrent tumor suspected on follow-up MR images.
Footnotes
1 Address reprint requests to Dong-Ik Kim, MD, Department of Diagnostic Radiology, Yonsei University College of Medicine, 134 Shinchondong, Seodaemoonku, Seoul 120–752, Korea.
References
- Received July 17, 2000.
- Copyright © American Society of Neuroradiology