Abstract
Summary: Although variations of the anterior cerebral artery (ACA)–anterior communicating artery complex are commonly identified on imaging studies, an infraoptic course of the ACA is exceedingly rare. What is believed to be the first case of an infraoptic course of the ACA discovered with MR angiography and further characterized with conventional angiography is presented. The high prevalence of associated aneurysms and the implications for surgical planning make preoperative recognition of this anomaly critical.
Anomalies of the anterior cerebral artery (ACA)–anterior communicating artery (AcomA) complex are commonly discovered with imaging and are usually of little clinical significance. However, an infraoptic course of the ACA is an exceedingly rare anomaly with important implications. Approximately 30 cases of this anomaly have been identified during autopsy or surgery or with conventional angiography (1). We present what is believed to be the first case identified with MR angiography and further evaluated with conventional arteriography.
Case Report
A 63-year-old woman with symptoms of transient ischemic attacks was referred from an outside facility for further evaluation of abnormalities discovered with MR imaging. An MR angiogram revealed aneurysms of the left middle cerebral artery bifurcation and basilar tip (Fig 1A) and an unusually low bifurcation of the right internal carotid artery (ICA) (Fig 1A and B). The A1 segment of the right ACA was noted to ascend between the optic nerves (Fig 1C). Conventional arteriography revealed an anomalous origin of the ACA proximal to the ophthalmic artery origin, possibly in an extradural location (Fig 2). There was an initial horizontal-medial course of the A1 segment of the right ACA, followed by an ascent to join the normally positioned AcomA (Fig 2C). The origin and course of the A1 segment were consistent with an infraoptic course of the ACA. The patient returned to the referring facility and was lost to follow-up.
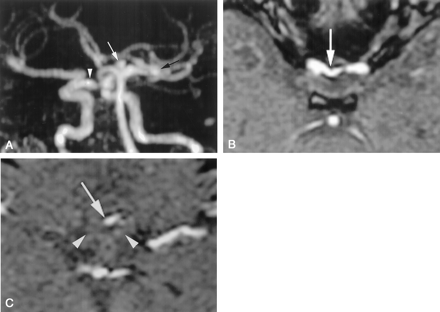
MR angiograms.
A, Posterior oblique reformation of a 3D time-of-flight MR arteriographic data set reveals low bifurcation of the right ICA (arrowhead). Basilar tip (white arrow) and left middle cerebral artery bifurcation (black arrow) aneurysms are demonstrated.
B and C, Source images from a 3D time-of-flight MR angiographic study at the level of the cavernous carotid (B) and optic chiasm (C). The right ACA (arrow) has a low origin from the ICA and ascends in a medial location between the optic nerves (arrowheads in C).
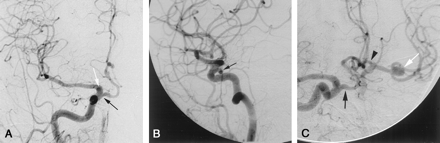
Arteriograms.
A–C, Frontal (A), lateral (B), and right anterior oblique (C) views from a conventional right common carotid arteriographic study demonstrate a low origin of the right ACA (black arrow), proximal to the ophthalmic artery. A small infundibulum (white arrow in A) demarcates the origin of the ophthalmic artery. The A1 segment of the ACA has a characteristic horizontal-medial course (as it passes under the ipsilateral optic nerve) before ascending to join a normally positioned AcomA. Reflux of contrast material across the patent AcomA illustrates the normal appearance of the contralateral A1 segment (arrowhead in C). The left middle cerebral artery bifurcation aneurysm (white arrow in C) is demonstrated.
Discussion
The A1 segment of the ACA usually arises from the ICA bifurcation and extends anteromedially above the ipsilateral optic nerve or chiasm to join the AcomA. Variation within the ACA-AcomA complex is extremely common, with some degree of asymmetry between the two A1 segments identifiable in approximately 80% of patients (2). An infraoptic course of the A1 segment is exceedingly rare, with approximately 30 previously reported cases (1). The infraoptic ACA usually arises from the intradural ICA at (or near) the origin of the ophthalmic artery. To our knowledge, this is the first case documenting the origin of this anomalous vessel proximal to that of the ophthalmic artery (Fig 2A), possibly in an extradural location.
The infraoptic ACA has a characteristic appearance at conventional angiography: apparent low bifurcation of the ICA and a horizontal-medial course of the proximal ACA as it passes under the ipsilateral optic nerve before turning superiorly to join the normally positioned AcomA (Figs 1A and 2C). MR angiography mirrors the findings seen with conventional angiography, but reviewing the source images or obtaining high-spatial-resolution images of the suprasellar region provides additional information not afforded with conventional angiography, such as the relationship of the ACA to the optic chiasm (Fig 1C). There are numerous variations of the ascent of the infraoptic ACA in relation to the optic apparatus; most frequently, the infraoptic ACA ascends anterior to the chiasm between the optic nerves (Fig 1C) or might pierce the ipsilateral optic nerve chiasm (2). That routine MR imaging of the brain will enable identification of this variation without concurrent MR angiographic sequences, except in the most fortuitous of cases, is doubtful, as shown in our case.
There is a slight right-sided predilection in cases of unilateral infraoptic ACA (3), but rarer cases of bilateral involvement have been reported (4). Although this anomalous vessel is functionally equivalent to the A1 segment of the ACA, in several reported cases a normally positioned (albeit extremely hypoplastic) A1 segment has been identified during autopsy or surgery or with conventional angiography (5). As a result, some authors proclaim that this anomalous vessel is more fittingly termed a “carotid–anterior cerebral artery anastomosis” (5, 6). Several theories regarding the origin of this anomalous vessel have been proposed. Early bifurcation of the ICA (7), enlargement of the prechiasmal anastomosis (5, 8, 9), persistent in utero communication between the primitive dorsal and ventral ophthalmic arteries (5, 6), and anastomosis between branches of the primitive olfactory and primitive maxillary arteries (8, 5, 9) have all been postulated as embryologic explanations.
Similar to other variations in the circle of Willis, the prevalence of cerebral aneurysms associated with an infraoptic course of the ACA is much higher (1, 6). The most common site of aneurysm formation is at the ACA-AcomA complex (3, 6). However, aneurysm formation may occur anywhere within the circle of Willis, as shown by our case. Other reported associations include ICA agenesis (5), anomalous origin of the ipsilateral ophthalmic artery from the external circulation (5), fused pericallosal arteries (3), moyamoya disease (1), and symptoms related to optic nerve or optic chiasma compression (9). Recognition of this anomaly will be vitally important in planning surgery for ACA-AcomA complex aneurysms, because an alternative surgical approach might be required to gain proximal control of the aneurysm (9). Failure to account for this anomaly might result in unnecessary dissection along and possible damage to the optic apparatus during aneurysm repair (9).
- Received June 18, 2001.
- Accepted after revision August 6, 2001.
- Copyright © American Society of Neuroradiology