Abstract
Summary: We present the case of a 19-month-old patient with tuberous sclerosis who developed a giant aneurysm of the midbasilar artery. Multiple Guglielmi detachable coils were used to fill and occlude the aneurysm and the involved segment of the basilar artery. After the procedure, the child had transient peripheral fifth cranial nerve palsy and no permanent neurologic deficits.
Tuberous sclerosis is a hereditary syndrome characterized by hamartomas in multiple organs. Non-CNS manifestations include angiomyolipomas in the kidneys, cystic lung disease that resembles lymphangioleiomyomatosis, cardiac rhabdomyomas, facial angiofibromas, and sclerotic bone islands in the vertebrae or long bone diaphyses (1, 2). Peripheral stenoses and aneurysms have been reported with some frequency in tuberous sclerosis, as have dysplasias of small vessels. These arterial lesions have been characterized by deficiency and fragmentation of elastic fibers and accumulation of mucopolysaccharides (3). Only recently have intracranial aneurysms been recognized as lesions associated with tuberous sclerosis. We present a case of a child with tuberous sclerosis that resulted in an aneurysm of the giant midbasilar artery that was treated with Guglielmi detachable coil (GDC) embolization. To our knowledge, this is the youngest patient treated with GDC for aneurysm occlusion. We also discuss the use of GDCs in the treatment of childhood aneurysms and the incidence of intracranial aneurysms in tuberous sclerosis.
Case Report
A 19-month-old female patient presented with three episodes of decreased movement and weakness of the left arm and leg. The maximum duration of these episodes was 2 minutes, with no change in mental status or overt convulsive activity. No cranial nerve deficits or ataxia was noted. The diagnosis of tuberous sclerosis had been confirmed when the patient was 4 months old, at which time she had clinical seizures consisting of fluttering of the eyes and deviation of the face and mouth to the left. MR imaging performed at that time showed numerous cortical and subcortical tubers and several subependymal nodules. Of note, the cerebral vasculature had a normal appearance on those MR images. The patient’s seizures had been well controlled with tiagabine for the 9 months preceding the episodes of left arm and leg weakness.
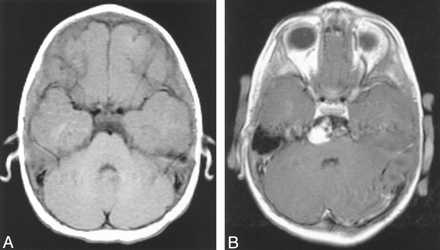
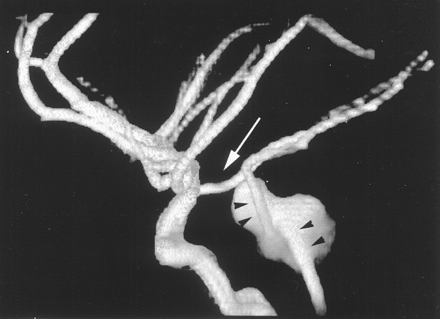
MR imaging of the brain performed when the patient was 19 months old revealed a large aneurysm of the midbasilar artery, projecting right of the midline and impinging on the adjacent pons (Fig 1). The aneurysm measured approximately 2.3 cm in largest oblique diameter, and the basilar artery between the anterior inferior cerebellar arteries and the superior cerebellar arteries was incorporated into the lumen of the aneurysm. MR angiography and subsequent conventional angiography both showed that the proximal basilar artery entered the aneurysm at a significantly more posterior and medial position than the distal basilar artery exited it. Flow from the proximal basilar artery was directed at the most dilated portion of the aneurysmal dome during systole. The circle of Willis was incomplete, with a single small posterior communicating artery on the left and a “fetal origin” configuration of the right posterior cerebral artery (Fig 2).
MR images obtained in a 19-month-old female patient who presented with three episodes of decreased movement and weakness of the left arm and leg.
A, Axial T1-weighted image, obtained through the level of the midpons when the patient was 4 months old.
B, Contrast-enhanced T1-weighted image, obtained through the same level when the patient was 19 months old. A large aneurysm has developed, projecting off of the basilar artery to the right.
Volume-rendered MR angiogram of the intracranial arterial circulation shows the large aneurysm of the basilar artery. Arrow, fetal origin posterior cerebral artery on right; arrowheads, acute angulation of basilar artery as it traverses aneurysm.
The rapid development of this aneurysm and the flow characteristics shown by conventional angiography both indicated a high likelihood of further aneurysmal growth and rupture. Because of her small size and the location and morphology of the aneurysm, direct surgical clipping and resection under cardiac arrest were thought to be extremely likely to result in a significant postoperative neurologic deficit or death. An endovascular approach was proposed. Aneurysmal occlusion with GDC embolization and basilar artery preservation was considered unlikely to be successful. Placement of a stent across the involved segment of the basilar artery and surrounding the stent with GDCs was considered as a method to preserve parent vessel flow. After careful measurement and analysis of the aneurysm with microcatheter angiography, it was concluded that the angle the stent would need to traverse was too great and that the risk of perforation or compromise of the proximal and distal basilar artery and adjacent branches was unpredictable. Accordingly, the decision was made to primarily occlude both the aneurysm and the involved segment of the basilar artery with multiple GDCs.
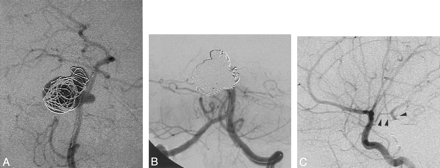
With the patient under general anesthesia and fully anticoagulated, a Rapid Transit (Cordis Endovascular, Miami Lakes, FL) microcatheter was passed through a 5F guide catheter positioned in the distal left vertebral artery, to rest with its tip in the distal basilar artery beyond the aneurysmal lumen. A second Rapid Transit microcatheter and 4F guide catheter system was placed in the distal right vertebral artery, with the microcatheter tip positioned in the dome of the aneurysm. A total of 19 GDCs were deployed into the aneurysm through this right-sided microcatheter, and three more GDCs were deployed at the distal junction of the aneurysmal lumen and the basilar artery through the left-sided system (Fig 3). The total length of deployed GDCs was 480 cm. After deployment, vertebral and carotid angiography revealed unimpeded flow into the anterior inferior cerebellar arteries through the proximal basilar artery and reconstitution of flow to the basilar tip and superior cerebellar arteries through the left posterior communicating artery.
Nineteen GDCs were deployed into the aneurysm through a right-sided microcatheter, and three more GDCs were deployed at the distal junction of the aneurysm lumen and the basilar artery through a left-sided system.
A, Digital subtraction angiogram of the left vertebral artery in the anteroposterior projection shows partial filling of the aneurysm with GDCs.
B, Repeat injection in the same projection after occlusion shows opacification of all proximal basilar artery branches and no distal flow.
C, Digital subtraction angiogram of the left internal carotid artery shows collateral flow to the basilar tip via the small left posterior communicating artery (arrowheads).
Sheaths were removed 1 hour after the procedure, and IV administered heparin infusion was instituted at a rate of 30 U/kg/h, which was the dose required to maintain full anticoagulation during the procedure. The infusion rate was decreased 8 hours after the procedure, when activated partial thromboplastin time exceeded 90 seconds. Significant bleeding developed at the groin puncture sites approximately 3 hours later, requiring transfusion. The heparin infusion was stopped, and bleeding was controlled with manual pressure. Blood pressure, oxygen saturation, and tissue perfusion remained normal, and a regimen of aspirin was begun the next day. No permanent sequelae or additional bleeding was noted. The child was irritable after the procedure, with episodes of clutching her head, suggesting headache. Clinically, it was suspected that she was experiencing pain from thrombosis of the aneurysm-coil mass, with resultant dural irritation and elaboration of inflammatory cytokines. A course of oral prednisone (2 mg/kg/day) was begun, resulting in improvement and eventual resolution of irritability and headache. The patient also developed a peripheral fifth cranial nerve palsy that gradually resolved 6 to 8 weeks after the procedure and did not require further specific therapy. A low-dose aspirin regimen was continued.
MR imaging performed 3 days after the procedure showed some abnormal signal intensity in the dorsal pons at the level of the aneurysm and multiple 1- to 2-mm foci of abnormal hyperintensity on the T2-weighted images obtained in the cerebellar hemispheres and occipital poles (Fig 4). The latter were presumed to reflect microemboli to the posterior circulation. Repeat MR imaging performed 6 weeks after the procedure showed resolution of these multiple small foci of abnormal signal intensity and a marked decrease in size of the abnormal signal intensity in the dorsal pons. MR angiography showed continued patency of the left posterior communicating artery, anterior inferior cerebellar arteries, and superior cerebellar arteries and absence of flow signal intensity into the aneurysm lumen. Approximately 2 months after the procedure, the patient had another episode of transient left hemiparesis, which completely resolved after 3 to 4 minutes. Emergent MR imaging showed stable findings compared with those noted 3 days after embolization, although the presumed microinfarcts were less prominent. At 12 months after the procedure, the patient remained otherwise healthy and without residual neurologic deficit.
MR images obtained 3 days after the procedure.
A, Axial T2-weighted image, obtained through the same level as the image shown in Figure 1, shows small foci of hyperintensity in the cerebellum (arrows) and hypointensity from the platinum coils filling the aneurysm.
B, Lateral volume-rendered MR angiogram shows the flow to the basilar tip (arrowheads) via the left posterior communicating artery (arrow), as well as flow into the proximal basilar artery from the vertebral arteries.
Discussion
Tuberous sclerosis is a hereditary syndrome characterized by hamartomas in multiple organs. It has been linked to genes on chromosomes 9 and 16, and the incidence of spontaneous mutation is very high. It is one of the phakomatoses, a group of inherited conditions characterized by CNS and skin lesions. The word phakoma was initially used to describe a characteristic retinal lesion in tuberous sclerosis (1). Intracranial lesions in tuberous sclerosis include cortical and subcortical tubers (hamartomas), linear or radial bands of abnormal signal intensity in the white matter on T2-weighted images, and subependymal tubers and giant cell astrocytomas. Non-CNS manifestations include angiomyolipomas in the kidneys, cystic lung disease that resembles lymphangioleiomyomatosis, cardiac rhabdomyomas, facial angiofibromas, and sclerotic bone islands in the vertebrae or long bone diaphyses (1, 2).
Peripheral stenoses and aneurysms have been reported with some frequency in tuberous sclerosis, as have dysplasias of small vessels. These arterial lesions have been characterized by deficiency and fragmentation of elastic fibers and accumulation of mucopolysaccharides (3). Only recently have intracranial aneurysms been recognized as lesions associated with tuberous sclerosis. A recent review (3) identified 15 cases of intracranial aneurysms in tuberous sclerosis reported in the literature. The authors noted that these aneurysms most commonly affected the internal carotid arteries and were frequently giant in size. Our experience has identified one other case of aneurysmal dilatation of the internal carotid artery in a child with tuberous sclerosis. Vascular lesions are a known manifestation of other phakomatoses, including moyamoya vascularity in cases of neurofibromatosis type 1 (4).
GDCs were developed for the endovascular treatment of intracranial aneurysms and have been used extensively for that purpose in adults. Few reports exist of their use in children, likely a consequence of the low incidence of intracranial aneurysms in this population. Lustgarten et al (5) reported their experience in treating a 12-year-old patient who had an aneurysm of the middle cerebral artery. They were unable to find reliable documentation of GDC embolization of aneurysms in pediatric patients in their review of the literature. Firat et al (6) reported their experience with GDC embolization of a giant aneurysm of the basilar artery in an 11-year-old patient. That aneurysm was partially thrombosed and dramatically decreased in size after coil occlusion of the residual lumen. To our knowledge, our patient is the youngest to have been treated with GDCs for aneurysm occlusion.
Arterial stents can be used as a scaffold through which to place embolic coils into an aneurysm. This technique holds promise for cases in which the aneurysmal neck is wide or not clearly defined, and patency of the parent artery is of paramount importance. In this case, there was very little normal vessel lumen proximal to or beyond the aneurysm on which to anchor a stent. Accordingly, the length of the stent would have to be ideal to provide the scaffolding for coil embolization without encroaching on the anterior inferior, superior cerebellar, or perforating arteries. Placement of a stent would limit circumferential growth of the basilar artery; the basilar artery diameter measured approximately 3 mm on cross-sectional and angiographic studies of this child, compared with a normal adult basilar artery diameter of just over 4 mm. Nevertheless, a stent would not likely limit longitudinal growth. Longitudinal growth of the basilar artery could result in uncovering of the aneurysmal neck and could become a source of recanalization of the aneurysm. All these factors contributed to the decision to primarily occlude the aneurysm and the involved segment of the basilar artery.
Two microcatheters were used in the deployment of coils in this procedure. One catheter was secured at the distal aspect of the aneurysm, ensuring that coils could be placed at this crucial location after aneurysm lumen occlusion. By placing coils through this distal catheter at the end of the procedure, we were able to prevent retrograde flow into the aneurysm while keeping the pressure within the lumen at normal to diminished levels during embolization of the lumen. The more distal catheter would have been very difficult to place after deployment of coils in the aneurysmal lumen, and attempting to do so would have risked disrupting the coil mass and causing distal embolization of previously detached coils.
During the first week after the procedure, our patient exhibited behavior that suggested headache: clutching her head and acting irritable. The exact cause of these symptoms was impossible to discern, but it is not unreasonable that the placement of almost 5 m of coils into the basilar artery might have caused some inflammatory reaction or dural irritation. The dramatic alteration in directional blood flow could also have contributed to some discomfort or disorientation, as could the presence of abducens nerve palsy. The mass of coils would be expected to have some direct mechanical effect on the adjacent pons as well, and the postprocedural MR imaging examination showed evidence of multiple microemboli. All these factors are suspected to have played a role in causing the patient’s headache and fussiness. Resolution of these symptoms with time and anti-platelet and anti-inflammatory treatment supports this theory. Pretreatment with steroids may be of value in attenuating these difficulties, as it is in limiting the fever, malaise, and irritability noted after embolization of solid lesions (7).
This child had potentially life-threatening bleeding from the groin puncture sites on the first day after the embolization, secondary to overaggressive anticoagulation therapy. This is a potential complication of any endovascular procedure performed in children for whom medications have to be adjusted for weight. Weight-equivalent doses may not be effective because of different rates of metabolism in children. The maintenance and adjustment of a heparin infusion is a routine task in adult intensive care units but is one not frequently encountered in pediatric intensive care units. A pediatric intensive care unit is without question a more ideal setting than an adult intensive care unit for the postprocedural care of a toddler treated with endovascular techniques. However, the interventionalist must make efforts to ensure that the unique features and requirements of these patients are clearly understood by all personnel providing care during the perioperative period.
Conclusion
This report describes the successful treatment of a giant aneurysm of the midbasilar artery in a 19-month-old patient with tuberous sclerosis. Aneurysms should be seen as an infrequent but important component of the tuberous sclerosis complex. The age of the patient and size of the aneurysm were unique aspects of this case, and both created difficulties in the treatment and periprocedural care of this child.
- Received June 11, 2001.
- Accepted after revision August 6, 2001.
- Copyright © American Society of Neuroradiology