Abstract
BACKGROUND AND PURPOSE: Thromboembolic events are a common source of complications during Guglielmi detachable coil (GDC) treatment of intracranial aneurysms. Thrombus formation at the coil-parent artery interface is not commonly reported but is an important potential source of emboli. We describe nine cases in which thrombus propagated from GDCs into the parent artery during coil therapy of cerebral aneurysms and subsequent treatment of the thrombus.
METHODS: A retrospective review of a procedural database was performed to identify cases in which thrombus occurred during GDC treatment of cerebral aneurysms during a 30-month period. All images were reviewed at the time of the procedure. Nine cases of thrombus forming at the coil-parent artery interface and five cases of distal emboli were identified among 210 cases. All patients underwent anticoagulation with heparin during GDC treatment procedures.
RESULTS: Thrombus was identified at the coil-parent artery interface during GDC treatment in nine (4.3%) of 210 cases. In each case, the thrombus was recognized before distal embolic complication occurred and was successfully treated with heparin alone (five patients) or with heparin plus a glycoprotein IIb-IIIa inhibitor (four patients).
CONCLUSION: Potential clinical complications can be avoided by early recognition of thrombus at the coil-parent artery interface and by administering appropriate medical therapy.
Thromboembolic events are a common source of complications during Guglielmi detachable coil (GDC) treatment of intracranial aneurysms. The incidence of reported thromboembolic events in large series ranges from 2.5% to 11%, with permanent deficits occurring in 2.5% to 5.5% of cases (1–3). Meticulous technique during embolization procedures is used to limit potential thromboemboli. Continuous flushing of catheters with heparinized saline eliminates static blood, which can clot and embolize. In general, patients are systemically anticoagulated with heparin during the procedure. Despite these measures, thromboemboli continue to occur with the potential to cause morbidity and mortality. Sources of emboli include chronic intra-aneurysmal thrombus displacement during coil placement, thrombus forming on the coil surface, stenosis of an adjacent artery due to the coil mass or coil herniation, or thrombus forming on the catheters during the procedure. Thrombus formation at the coil-parent artery interface is not commonly reported but is an important potential source of emboli.
We report a series of nine cases in which thrombus formed at the coil-parent artery interface during GDC treatment procedures. The thrombus was treated with additional IV administered heparin alone or in combination with a platelet glycoprotein IIb-IIIa receptor inhibitor. Thrombus formation at the coil-parent artery interface is an important phenomenon to recognize, because early treatment is likely to result in a good clinical outcome.
Methods
All cases of aneurysms treated by GDC placement during a 30-month period were reviewed for the presence of thrombus at the coil-parent artery interface. The parent artery is the artery that is in contiguity with the ostium of the aneurysmal sac. The coil-parent artery interface is the surface of the coil pack that interacts with in-flowing blood and is located at the aneurysmal neck in a completely coiled aneurysm. A retrospective review of our patient database was performed to identify cases in which thrombus occurred. All angiographic images were reviewed at the time of the procedure. Procedural data, information regarding postprocedural therapy, and results of the discharge neurologic examination were obtained from the patient’s hospital chart. There were multiple operators performing the GDC treatment procedures who used similar techniques. The technique for GDC therapy has been previously described (4). All patients received IV administered heparin after placement of a vascular sheath in the common femoral artery. A 6F guiding catheter was placed in either the internal carotid artery or the vertebral artery, and the aneurysmal sac was accessed with a microcatheter. All cerebral aneurysms were treated by using GDC T-10 or T-18 coils (Target Therapeutics, Natick, MA). The common femoral arteriotomy was managed with a 6F Perclose device (Abbott Laboratories, Redwood, CA) in patients 1, 3, 6, 7, and 9 and with manual compression in patients 2, 4, 5, and 8. General anesthesia was administered to patients 1, 3, 7, 8, and 9, and conscious sedation was achieved with IV administered fentanyl and midazolam to patients 2, 4, 5, and 6.
Therapy for coil surface thrombus was administered at the operator’s discretion. Abciximab (ReoPro; Eli Lilly, Indianapolis, IN) was IV administered as a bolus (0.25 mg/kg during 3 min) and then by 12-hour infusion (10 mcg/min). Integrilin (Eptifibatide; COR Therapeutics, South San Francisco, CA) was IV administered as a bolus (180 ì/kg during 3 min) and then by 20-hour infusion (2 mcg/kg/min). Activated clotting time (ACT) was measured in seconds by using a Hemochron 801 ACT machine (ITC, Edison, NJ). Postprocedural anticoagulation consisted of aspirin (325 mg/day) or aspirin and Plavix (75 mg/day) (Clopidogrel; Bristol-Myers Squibb, New York, NY).
Results
During a 30-month period, 210 patients underwent GDC therapy of cerebral aneurysms. Fourteen (6.7%) patients experienced thrombus-related complications. Distal thromboemboli were present in five of 14 patients, but none of these had demonstrable thrombus at the coil surface during the procedure. Thrombus was shown at the coil-parent artery interface in nine patients (Table 1). At the time of coil therapy, four aneurysms were ruptured and five were unruptured. Although all patients were systemically heparinized during the procedure, no patient with subarachnoid hemorrhage received a glycoprotein IIb-IIIa inhibitor. After thrombus was recognized at the coil-parent artery interface, additional heparin was administered in seven of nine cases (Table 2). In two cases of unruptured aneurysms, because the ACT measured near 300 seconds at the time of thrombus formation, it was thought that additional heparin may not treat the thrombus. Our previous experience with this problem led the operator to choose a glycoprotein IIb-IIIa agent as the initial therapy. Glycoprotein IIb-IIIa inhibitors (abciximab or Integrilin) were administered in five of nine cases. In four of five cases treated with a glycoprotein IIb-IIIa agent, thrombus at the coil surface had progressed or there was no diminution of thrombus despite increased heparin dosing. No bleeding complications occurred in the nine cases with thrombus at the coil-parent artery interface. All patients received orally administered anti-platelet therapy (aspirin, nine of nice patients; aspirin and Plavix three of nine patients) for 6 weeks after undergoing GDC therapy. Aspirin was to be continued indefinitely for all patients. Patient 7 experienced transient pronator drift secondary to vasospasm in the cerebral vessels contralateral to the treated aneurysm. However, no patient had evidence of stroke at the time of discharge or 30 days after treatment.
Clinical data of GDC treatment cases with thrombus forming at coil-parent artery interface
Case summary of intra-procedural anti-coagulation therapy
Representative Case Reports
Case 1.—
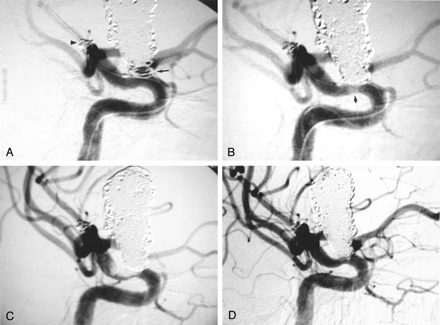
Two years previously, a 54-year-old female patient experienced Hunt and Hess grade IV subarachnoid hemorrhage with Terson syndrome in the right eye from a giant right ophthalmic artery aneurysm (14 × 30 mm) rupture. At the time of the subarachnoid hemorrhage, the aneurysm was treated with GDCs. The patient fully recovered except for a minor visual field deficit in the right eye. During the 2-year interval, enlargement of the small residual aneurysm occurred, measuring 6 × 3 mm (width by height). GDC treatment of the ophthalmic aneurysm remnant was performed. After the vascular sheath was placed, a 6000-U heparin bolus was administered and then 1000 U/hr heparin was administered. The residual aneurysm was packed with GDCs by using a balloon remodeling technique. When the last coil was placed and the balloon removed, control angiography revealed nonocclusive thrombus at the coil-parent artery interface (Fig 1). ACT measured 307 seconds. Repeat angiograms obtained 15 and 25 minutes later revealed an increase in thrombus burden. Abciximab (0.25 mg/kg) was IV administered and was then infused (10 μg/min). Serial angiograms showed diminished thrombus burden after 10 minutes and only small residual thrombus 40 minutes after abciximab dosing. No distal artery emboli were observed. Abciximab infusion was continued for 12 hours. After the patient recovered from general anesthesia, a 300-mg loading dose of Plavix and 325 mg of aspirin were administered orally. A regimen of aspirin and Plavix (75 mg/day) was initiated and was continued for 6 weeks. The patient had no neurologic sequelae.
Images from the case of a 54-year-old female patient with giant ophthalmic artery aneurysm remnant.
A, Right internal carotid artery injection, oblique projection. Mask artifact outlines the margin of the dense coil pack filling the giant ophthalmic artery aneurysm. The first coil has been detached within the remnant at the aneurysm base (black arrow). Note GDCs within a previously embolized middle cerebral artery bifurcation aneurysm (arrowhead).
B, After completion of the embolization procedure, control angiogram shows a small, poorly marginated, hazy opacity at the coil-parent artery interface, suggesting early thrombus formation (arrow).
C, Repeat angiogram obtained 25 minutes later shows marked increase in thrombus burden at the coil surface and in the parent artery.
D, Forty minutes after the administration of the abciximab bolus, near-complete resolution of thrombus at the coil surface can be seen.
Case 2.—
A 67-year-old female patient presented for GDC treatment of an unruptured basilar tip aneurysm (5-mm diameter, 3.5-mm neck). A bolus of 5000 U of heparin was IV administered after placement of a femoral artery vascular sheath, after which the ACT measured 238 seconds. A 6F guiding catheter was placed in the left vertebral artery, and a microcatheter was advanced into the aneurysmal sac. Using high-resolution, biplane road map angiography, GDCs were deployed and electrolytically detached. After placement of the third coil into the aneurysm, control angiography showed thrombus on the coil loops at the aneurysm neck. An additional 3000 U of heparin was immediately IV administered, which increased the ACT to 318 seconds. After 30 minutes of observation, the appearance of the thrombus remained unchanged. In an attempt to allow fibrinolysis of the intraluminal thrombus and prevent further platelet aggregation, abciximab (0.25 mg/kg) was IV administered during 3 minutes and then an infusion of 10 μg/min was maintained for 12 hours. Angiography performed 20 minutes after the administration of the abciximab bolus showed near-complete resolution of the thrombus, with complete resolution at 30 minutes. Cerebral angiography showed no evidence of distal embolism. Coil treatment of the aneurysm continued until a tight coil mass was achieved. A regimen of aspirin (325 mg) was started on postprocedural day 1. The results of the neurologic examination remained unchanged, with no deficits at the time of discharge.
Case 5.—
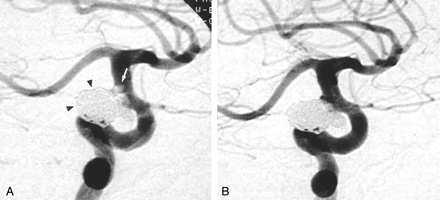
A 49-year-old male patient with subarachnoid hemorrhage was referred for GDC treatment of a basilar tip aneurysm. After placement of a vascular sheath, 5000 U of heparin was IV administered. Diagnostic angiography revealed a 6-mm-diameter basilar tip aneurysm with a 5-mm neck. A catheter was placed in the left vertebral artery with a guide catheter, and a microcatheter was used to access the aneurysmal sac. ACT measured 200 seconds, and a 2000-U bolus of heparin was administered. A T-10 6 mm × 8 cm 3D coil was placed and detached successfully in the aneurysmal sac. During placement of the second coil (T-10 2D, 4 mm × 6 cm), 1 to 2 mm of the leading edge of the initially placed 3D coil was displaced into the basilar tip. Control angiography showed no thrombus or distal emboli. The aneurysm was subsequently tightly packed with GDCs. After completion of coil placement, control angiography showed that nonocclusive thrombus had formed on the coil and extended into the right and left P1 segments of the posterior cerebral artery (Fig 2). Additional heparin was administered to increase the ACT to 300 seconds. Repeat angiography showed a decrease in the thrombus burden without distal emboli. The remainder of the aneurysmal lumen was packed with GDCs. At the termination of the case, there was a small, nonocclusive thrombus remaining in the right P1 segment and along the left, inferior coil-parent vessel interface. Heparin infusion was maintained to keep the activated partial thromboplastin time at 50 to 70 seconds. A regimen of aspirin (325 mg) was started after the procedure. Repeat angiography performed on post-treatment day 1 showed resolution of the thrombus without distal emboli (Fig 2). However, there was partial recanalization of the aneurysm centrally. Follow-up angiograms obtained at 6 months showed no opacification of the central portion of the aneurysm (Fig 2B) but no change in the residual contrast opacification at the right base of the aneurysm, consistent with a small remnant. On post-treatment day 2, the patient had a generalized seizure. CT of the head showed hydrocephalus, which was treated with lumbar puncture and drainage. A right sixth nerve palsy was detected on post-treatment day 3, which resolved within 24 hours. The neurologic examination performed at the time of discharge showed no deficit.
Images from the case of a 49-year-old male patient with a basilar tip aneurysm.
A, Angiogram of the left vertebral artery, transfacial projection. The basilar tip aneurysm is packed with GDCs (short arrows). A linear filling defect with hazy margins, consistent with thrombus, is present in the distal basilar artery and extends into the right P1 segment (long arrows).
B, Angiogram of the left vertebral artery, transfacial projection. After 24 hours of heparin infusion, follow-up angiogram obtained after GDC treatment shows resolution of thrombus. However, there is partial recanalization of the aneurysm centrally.
Case 8.—
A 57-year-old female patient with an unruptured 10-mm-diameter, lobulated right superior hypophyseal aneurysm with a 6-mm neck was referred for GDC treatment. The aneurysm was treated with GDCs. However, a 4 × 3 mm anterior-inferior lobulation near the aneurysm base could not be accessed and resulted in a remnant. Follow-up angiography performed at 3 months showed interval recanalization of the aneurysm with coil compaction at the neck and no change in the uncoiled aneurysm remnant. A guide catheter was placed in the right internal carotid artery, and 5000 U of heparin was IV administered. The aneurysm remnant was accessed with a microcatheter and treated with a single GDC. Control angiography performed before coil detachment revealed thrombus formation at the coil-parent artery interface (Fig 3). Cerebral angiography showed no distal emboli. An additional 5000 U of heparin was administered, with subsequent ACT measuring 270 seconds. Repeat angiography showed persistent thrombus that remained unchanged for 30 minutes. Seventeen milligrams of Integrilin (180 μg/kg) was administered as an IV bolus during 3 minutes. Repeat angiography performed approximately 10 minutes later showed no thrombus on the coil surface or distal embolization. A maintenance dose of Integrilin (2.0 μg/kg/min) was continued for the next 20 hours. A regimen of aspirin (325 mg) was started after the patient recovered from general anesthesia. The results of the neurologic examination performed at the time of discharge were normal.
Images from the case of a 57-year-old female patient with a superior hypophyseal artery aneurysm.
A, Control angiogram of the right internal carotid artery, contralateral transorbital oblique projection. The superior hypophyseal artery aneurysm is tightly packed with GDCs (arrowheads). Thrombus has formed at the coil-parent artery interface, manifesting as a filling defect with a lobular contour with hazy margins (white arrow).
B, Control angiogram obtained 10 minutes after the administration of the Integrilin bolus shows resolution of thrombus at the coil-parent artery interface.
Discussion
We describe nine cases of thrombus forming at the GDC-parent artery interface during coil treatment of cerebral saccular aneurysms. We consider these cases to be instructive in that they highlight the importance of early recognition and treatment of such thrombus formation. Thrombus formation on coils, both inside and outside the aneurysmal lumen, may be the source of most distal thromboembolic complications during GDC therapy. Early recognition of thrombus formation allows medical management to avert distal thromboembolism. In five of nine cases, early treatment with additional heparin alone resulted in resolution of the thrombus. In four of nine cases, heparin failed to reverse the thrombus formation and treatment with a glycoprotein IIb-IIIa drug was needed. We urge frequent control angiography and close scrutiny of the coil-parent artery interface to improve recognition and allow treatment of parent artery thrombus formation.
Thrombus formation at the coil-parent artery interface has rarely been previously reported. Lempert et al (5) reported a case of thrombus forming at the coil-parent artery interface in a patient with a ruptured basilar tip aneurysm during GDC treatment. Cronqvist et al (6) described a case in which thrombus was present at the neck of a middle cerebral artery aneurysm 2 hours after GDC treatment. Tateshima et al (7) described a case in which thrombus formed on a GDC protruding into the P1 segment and occluding the posterior cerebral artery during embolization of a basilar tip aneurysm. However, most previously reported cases of thrombus formation during GDC treatment of cerebral aneurysms manifest as distal emboli causing vessel occlusions or filling defects (1, 2, 6, 8).
Thrombus at the coil-parent artery interface can be a subtle finding and is probably under-recognized. Detection of thrombus by angiography requires that it be large enough to displace contrast material and appear as a filling defect. Angioscopic studies have shown thrombus to be consistently underestimated by angiography (9). In intracoronary vessels, angiography was 75% sensitive for large thrombi (occupying >25% of the luminal diameter) and was only 30% sensitive to small thrombi (10). During GDC therapy, frequent control angiography with high-spatial-resolution imaging and a high index of suspicion are necessary for diagnosis. However, even under optimal conditions, thrombus may be present but not detected.
Thrombus probably forms during the coil treatment process, primarily from flow disturbance and stasis within the aneurysm and perhaps from electrothrombosis. The process of thrombus formation at the anode (coil) during electrolysis in aneurysms and during GDC detachment has been well described (11). Theoretically, short detachment times may limit the quantity of thrombus forming on the coil during the detachment process (11). However, others have questioned the relevance of electrothrombosis of aneurysms during detachment of GDCs (12). Incidence of ischemic events after GDC treatment has been reported to occur with greater frequency in wide neck aneurysms (1, 8). Interestingly, seven of our nine cases had aneurysmal necks ≥5 mm. A wide aneurysmal neck may promote propagation of thrombi by providing a larger coil surface area on which to form or greater access of thrombus formed in the aneurysmal sac to the parent artery.
Slow flow due to vasospasm or presence of the guide catheter in the carotid or vertebral artery can promote thrombus formation in the parent artery. Our standard practice is to use a 6F guide catheter. Four patients in our case series had basilar tip aneurysms with placement of 6F guide catheters in the vertebral artery during treatment. After placement, catheter position and flow within the parent artery is checked with the injection of contrast material; the catheter is repositioned if flow within the parent artery is diminished or local vasospasm is shown. We do not use 5F guide catheters except in pediatric cases, because they offer inferior control angiography, which can impair visualization of relevant anatomy and diagnosis of thrombus formation. Diminished flow from placement of a 6F instead of a 5F guide catheter is unlikely to have resulted in thrombus formation at the coil-parent artery interface, because any marginal decrease in flow from the ipsilateral vertebral to the basilar artery should be maintained by the contralateral vertebral artery.
If thrombus is recognized before coil detachment, it is probably not advisable to remove the coil. This action would risk shearing thrombus from the coil surface, resulting in distal embolization of the thrombus. Rather, by leaving the coil in place, the thrombus is adherent to the coil surface and is relatively “controlled.” After resolution of visible thrombus, coil therapy of the aneurysm can be completed. The decision to treat was predicated on the idea that thrombus forming at the coil-parent artery interface despite concurrent use of heparin is a potential source of thromboemboli or parent vessel occlusion. Although in none of our cases did thrombus at the coil-parent artery interface embolize to a distal vessel, we cannot assume that it would not have done so in time.
A variety of options are available to treat thromboemboli recognized during GDC procedures, including thrombolytic agents, platelet glycoprotein IIb-IIIa inhibitor, and systemic heparin. Cronqvist et al (6) describe 19 cases of thromboemboli occurring during GDC embolization of aneurysms that were treated with urokinase. Complete recanalization occurred in 10 cases, with good clinical recovery being achieved in 14. Fibrinolysis with urokinase or tissue plasminogen activator in the presence of an unruptured aneurysm probably adds little risk of intraprocedural rupture. However, the use of thrombolytics in patients with ruptured aneurysms is controversial and does not have an insignificant rate of intracranial hemorrhage. In the series reported by Cronqvist et al (6), devastating intracranial hemorrhage occurred in three of six patients with ruptured aneurysms, and distal emboli were treated with thrombolytic agents. We chose to use heparin alone or heparin in combination with a glycoprotein IIb-IIIa agent because of their safety profile and ability to inhibit further thrombus formation (13).
Anticoagulation with heparin during GDC treatment for both ruptured and unruptured aneurysms is a standard measure to prevent thromboembolic complications. Continuous flushing of the catheters with heparinized saline eliminates static blood, which can clot and embolize. ACT is used to gauge the degree of systemic heparinization by measuring the degree of ex vivo prolongation of blood clotting due to heparin. Some interventionalists use empiric heparin dosing without evaluation of ACT, whereas others dose heparin to keep the ACT at a predetermined level, ranging from two to six times normal (2, 3, 7). Heparin was used in all our cases, and ACT was used to monitor the degree of anticoagulation. However, there were multiple operators and there was no standard procedure regarding monitoring or the degree of anticoagulation. Data regarding intraprocedural heparin were derived primarily from evaluating patients undergoing percutaneous coronary interventions. Although there is no consensus, in patients undergoing percutaneous coronary interventions who do not receive a glycoprotein IIb-IIIa inhibitor, heparin dosing to achieve an ACT of 300 to 350 seconds by using the Hemochron device has been recommended (14). With the addition of a glycoprotein IIb-IIIa agent, there is no longer a benefit to a high ACT in lowering the ischemic complication rate and it significantly increases the bleeding complication rate (13). When a glycoprotein IIb-IIIa agent is used, low doses of heparin are administered, with a target ACT of 200 seconds (14). Heparin dosing resulted in an ACT less than 250 seconds in four cases and may have contributed to thrombus forming at the coil surface. However, thrombus formed and progressed by serial angiography in two cases, with ACT measuring 299 and 307 seconds. Heparin dosing was increased in seven cases, once thrombus at the coil-parent artery interface was shown. In five of seven cases, resolution or a decrease in thrombus burden was observed, and in two cases, no effect was observed. The anticoagulant effect of heparin is due to its high affinity for antithrombin III, which accelerates binding and inactivation of thrombin, factor Xa, and factor IXa (15). Although not fibrinolytic, high dose heparin therapy alone has been shown to promote coronary reperfusion and to promote fibrinolysis in experimental thrombi (16). In patients with stroke who have been treated with heparin alone, an 18% vessel recanalization rate has been observed (17).
Several factors could have led to inadequate anticoagulation. In all our cases, heparin was intermittently administered as a bolus by using a fixed dose instead of an initial weight-adjusted bolus. A fixed-dose bolus of heparin can result in increased bleeding complications in patients undergoing percutaneous coronary procedures and receiving a glycoprotein IIb-IIIa agent, especially in persons with smaller body mass (18). However, a recent randomized trial comparing fixed-dose heparin versus weight-adjusted heparin dose during coronary angioplasty did not show a difference in ischemic or bleeding complications between groups but did result in earlier sheath removal in the weight-adjusted group (19). Continuous infusion of heparin has been reported to maintain a steady level of anticoagulation, as measured by partial thromboplastin time, and is associated with fewer bleeding complications than intermittent boluses in patients being treated for deep vein thrombosis over a period of days (20). This observation probably does not translate to the time constraints of neurointerventional procedures, because they are completed within hours. Dose response to unfractionated heparin is heterogeneous, often with an initial sensitivity or resistance and then an independent and variable rate of consumption (21). This suggests that frequent monitoring is necessary to maintain adequate anticoagulation in a chosen therapeutic range for individual patients regardless of the chosen heparin dosing scheme. However, the level of anticoagulation with heparin needed to prevent thrombus formation in neurovascular procedures is unknown. Importantly, despite aggressive intra- and postprocedural anticoagulation with heparin to prevent thrombus formation, thromboembolic complications continue to occur (2, 8). Combined anticoagulation strategies with heparin and antiplatelet agents may be necessary to further decrease thromboembolic complication rates during GDC treatment of aneurysms.
Glycoprotein IIb-IIIa agents have been extensively studied in patients undergoing percutaneous coronary interventions and acute coronary syndromes (13). However, experience with glycoprotein IIb-IIIa inhibitors in the cerebral vessels is limited. Wallace et al (22) used abciximab to prevent rethrombosis in the basilar artery after balloon angioplasty. Lempert et al (5) described the use of IV administered abciximab as rescue therapy for treatment of acute parent vessel thrombosis during GDC treatment of an intracranial aneurysm. Qureshi et al (23) recently described their experience with abciximab for high risk angioplasty of the carotid, vertebral origin, and basilar arteries. In the Abciximab in Acute Ischemic Stoke Trial, abciximab was administered to 54 patients within 24 hours of onset of acute ischemic stroke (24). This study found no increase in hemorrhagic complications and a trend toward better functional outcomes for patients treated with abciximab. Whether this low intracranial hemorrhage rate will be similar in patients with unruptured and ruptured aneurysms is unknown. Recently, Ng et al (25) reported successfully treating a thrombotic occlusion of the anterior communicating and anterior cerebral artery during GDC treatment with abciximab in the setting of an acutely ruptured aneurysm.
Glycoprotein IIb-IIIa inhibitors are potentially useful in facilitating thrombolysis in the setting of thromboembolic complications during endovascular procedures. Rapid fibrinolysis was observed in four of our cases in which a glycoprotein IIb-IIIa inhibitor was used as a “rescue” agent. This observation is consistent with observed rates in the coronary arteries in which fibrinolysis occurred within 10 minutes of patients receiving abciximab (26). Glycoprotein IIb-IIIa inhibitors are potent inhibitors of platelet aggregation; however, mechanisms by which glycoprotein IIb-IIIa inhibitors may promote fibrinolysis have not been completely elucidated. There is evidence that by blocking platelet aggregation, acceleration of thrombin generation is significantly slowed by five to six orders of magnitude, resulting in diminished fibrin formation (27). Plasminogen activator inhibitor-1, a potent inhibitor of tissue plasminogen activator, is released by activated platelets and found in high concentrations in platelet-rich thrombi (28). Glycoprotein IIb-IIIa inhibitors do not directly inhibit plasminogen activator inhibitor-1 secretion, but by decreasing platelet concentration within thrombi, it may decrease plasminogen activator inhibitor-1 concentrations (29). Abciximab has been shown to cause increased urokinase and decreased secretion of plasminogen activator inhibitor-1 from endothelial cells (30).
Our case series is limited by the small number of cases in which thrombi were shown at the coil-parent vessel interface. Although no definite treatment recommendations can be made, we suggest close monitoring of the ACT, with heparin dosing to keep the ACT at least greater than 250 seconds with a range to 350 seconds when using the Hemochron device. We use glycoprotein IIb-IIIa inhibitors as rescue agents to treat thromboembolic complications during coil therapy of unruptured aneurysms. On the basis of our experience with these complications, we have started to dose all unruptured aneurysm cases with aspirin before coil therapy. We conclude that thrombus does form at the coil-parent artery interface during GDC treatment of intracranial aneurysms, and recognition and early treatment may avert this potential source of morbidity.
Footnotes
This work was supported in part by an educational grant from Phillips Medical Systems.
References
- Received November 21, 2001.
- Accepted after revision April 29, 2002.
- Copyright © American Society of Neuroradiology