Abstract
Summary: Congenital abnormalities of the pituitary gland are rare and may be associated with midline cranial, orbital, and facial anomalies and with hormonal insufficiency. Here we report a case of asymptomatic, abnormal migration of the adenohypophysis. The normally developed adenohypophysis was located in the sphenoid bone and developed on the intersphenoidal septum, extending from the superior pharyngeal wall to the floor of the sella turcica (craniopharyngeal canal). The abnormal migration of the pituitary gland was isolated without hormonal deficit, brain, or facial developmental anomalies.
Migration disorders of the pituitary gland are a rare phenomenon, almost always reported for the neurohypophysis in patients with micropituitarism (1). Migration arrest of the adenohypophysis in the sphenoid bone is extremely rare, probably because it is usually asymptomatic. The exact etiology of this anomaly is not clear but it probably results from vascular, toxic, or genetic insults occurring during the period of Rathke’s pouch obliteration (42–45 days of gestational life). As it migrates with the pharyngohypophyseal pouch, the adenohypophysis may develop within the sphenoid bone in front of the sphenooccipital synchondrosis. Moreover, in case of excessive sphenoid pneumatization (eg, sphenoid pneumatization over the clivus) the ectopic adenohypophysis may develop within the sphenoid sinus, attached to the sphenoid septum, presenting as an intrasinus mass.
Case Report
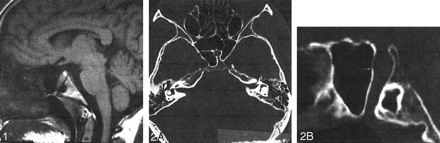
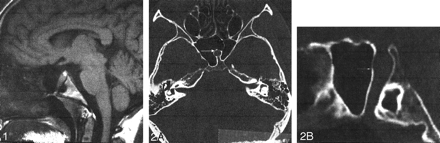
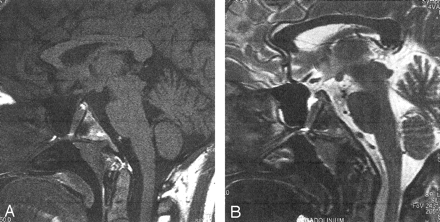
A 20-year-old woman was referred to our department for MR imaging of the brain following a new onset of seizures. Physical examination was normal. She had no significant medical history. Review of systems was negative, and she took no medications. Brain MR documented normal signal intensity and morphology of the brain parenchyma except for an empty sella turcica. A linear structure going from the floor of the sella turcica to the inferior wall of the sphenoid sinus was identified (Fig 1). A CT examination of the skull base demonstrated an intrasphenoidal bony canal 8 mm in diameter, extending from the sella turcica to the nasopharynx and adjacent to the right lateral border of the sphenoid septum (Fig 2A and B). Pituitary MR imaging demonstrated normal, intrasellar high signal intensity of the posterior pituitary gland and a well-developed pituitary infundibulum inferiorly (Fig 3A and B). No anterior pituitary gland was seen within the sella turcica. The craniopharyngeal canal was filled by tissue of intermediate signal intensity on both T1- and T2-weighted images (sagittal sections), which demonstrated intense enhancement similar to nasal mucosa on dynamic contrast-enhanced sequences. An associated clival anomaly was present.
Sagittal T1-weighted image (528/16/2 [TR/TE/NEX]). Note the canal extending from the floor of the sella to the nasopharynx (arrows).
Axial (A) and sagittal (B) reconstructed CT images through the sphenoid bone. Note the intrashenoidal bony canal (arrows).
Midsagittal T1- (A, 450/14/2) and B) T2-weighted (B, 550/117/3) weighted MR images. Note the midline canal filled with intermediate signal intensity material on both T1- (A) and T2-weighted (B) images extending from the sella to the nasopharynx. Note the normal location of the pituitary infundibulum (A, arrow) and a small amount of CSF with the upper portion of the canal in (B).
Because the patient had no hormonal abnormalities, the imaging studies were thought to be most consistent with an ectopic adenohypophysis due to arrest of normal gland migration. The patient had no stigmata of any other developmental anomalies, and inquiry into possible insults occurring in utero did not yield clues. The patient was informed of this developmental anomaly and of the risk it might pose in the event of sphenoid surgery.
Discussion
The hypophysis develops from two different origins: one from an ectodermal outpocketing of the stomodeum, known as Rathke’s pouch, and the other from a downward extension of the diencephalon, called the infundibulum (1). At 3 weeks of gestation, Rathke’s pouch grows dorsally as the infundibulum grows ventrally. During the second month of gestational development, the Rathke’s pouch loses its connection with the stomodeum, and its anterior part forms the adenohypophysis. The obliteration of Rathke’s pouch occurs between the 42 and 45 days of gestational life (2).
The pituitary gland lies within the boundary between the neural crest cell (NCC)-dependant and NCC-independent portions present the composite sella turcica. At the end of cephalic rotation, both the adenohypophysis and neurohypophysis are located within the sella turcica, separated by the Rathke’s pouch. The embryonic pharyngohypophyseal transsphenoidal (craniopharyngeal) canal is located beneath to the sella turcica, extending between the superior wall of the nasopharynx to the floor of the sella turcica in an oblique fashion (3). It is located within the central sphenoid ossification center, in front of the sphenobasilar synchondrosis (4). This inconstant persistent remnant is termed the craniopharyngeal canal and, in cases of larger defects, “persistent hypophyseal canal” (4). It is observed in approximately 0.42% of the asymptomatic population (3). It may be incomplete, ending blindly without communication with the sella turcica and may contain air if a communication with the pharyngeal lumen exists (3). Its imaging features depend on 1) status of sphenoid bone aeration; 2) status of canal obliteration; and 3) communication with the pharynx (3).
On axial CT sections, the craniopharyngeal canal is located anterior to the lucency of the sphenobasilar synchondrosis (5). It is a midline round foramen with a sclerotic rim. Some air may be present within the canal. On MR images, the low signal intensity of the canal contrasts against the high signal intensity of fatty clival bone marrow (4).
In excessive sphenoid sinus aeration (approximately 5–10% of patients), the craniopharyngeal canal follows the intersphenoidal septum and may, therefore, be located within the sphenoidal sinus, surrounded by the aerated sphenoid cells, sometimes laterally. Moreover, the presence of pituitary tissue within the canal may mimic an intrasphenoidal cystic mass as in the patient. This is correlated to the date of aeration of the sphenoid sinus, which occurs after the pharyngo-hypophyseal canal closure.
Migration and development of the adenohypophysis and neurohypophysis follow two different pathways. It is widely accepted that the epithelial hypophysis has its developmental origin from the extraneural portion of the embryo (ie, the stomodeal ectoderm). But development of adenohypophysal cells requires a good differentiation of the neural crest. Recent works have shown that, in mammalian embryos, the origin of adenohypophyseal cells is before the formation of Rathke’s pouch and adenohypophysis becomes visible at the end of the first month of gestational development (1, 6). Then, the developing pituitary establishes a link to the hypothalamus by the progressive elaboration of the hypophyseal stalk, the median eminence, and the venous portal circulation. By contrast, the posterior hypophysis pinches off from the diencephalon remaining attached by a bridge that will develop into the hypophyseal stalk. All these were normally developed in the reported case.
Migration anomalies are congenital anomalies caused by genetic (neurofibromatosis type 1, trisomy 13), vascular, or environmental insults to migrating neuroblasts (7). The major cell migrational activity lasts approximately 2 months, occurring from 7 to 16 weeks of embryonic life (7, 8). Exposure to ionizing radiation, excessive levels of alcohol (fetal alcohol syndrome), anticonvulsants (phenytoin, barbiturates), or toxic materials (methyl mercury poisoning) may be associated with abnormal neural cell migration (9). Because of the different origins of adenohypophisis and neurohypophysis, an abnormal migration of the pituitary gland involves only the anterior portion of the gland. Arrested migration of the anterior pituitary gland is due to an NCC disorder occurring in utero and probably results from an absence of signal to the epithelial cells during the 2nd month of embryonic life (10). Its occurrence after emergence of neural crest cells explains concomitant normal development of the posterior pituitary gland; the pituitary stalk; the orbital globe; the pituitary vascular system resulting from mesenchymal differentiation; the sphenoid bone, in which the posterior part came from the cephalic mesoderm while the anterior part derived from NCC.
Congenital anomalies of the adenohypophysis may be associated with other midline anomalies such as Chiari type 1 malformation and medial deviation of the internal carotid canal. Associated cortical brain dysplasia was recently reported (2). The association between congenital anomalies of the pituitary gland and breech deliveries is probably related to fetal pituitary hormonal deficiency.
Occasionally, as in our patient, a portion or all of the adenohypophysis may persist in the pathway of the pharyngeal pouch, either in the nasopharyngeal wall or within the sphenoid bone, termed “ectopic intrasphenoidal hypophysis.” In this anomaly of adenohypophyseal migration, a well-developed normal neurohypophysis may be located in its normal position (10). Persistence of the craniopharyngeal canal allows nasopharyngeal development of the adenohypophysis. Panhypopituitarism in these instances is probably related to inadvertent hypophysectomy (4, 10, 11).
Moreover, our case demonstrates that large persistent craniopharyngeal canals are not always related to transphenoidal meningocele as some authors postulated (12). Intrasphenoidal ectopic normal functioning pituitary gland was recently reported by Ekinci et al (Table) (4, 11, 13).
Analysis of the literature
If the etiology of the persistence of the craniopharyngeal canal is still not completely clarified, recent works on embryology and presence in normal location of the posterior pituirary lobe and stalk argue against a remnant of a vascular channel. Moreover, trappage of CSF inside this canal without leak as in our reported case may coexist as the pituitary gland is completely enclosed by a dura mater fold except for the aperture of the diaphragma sellae. The craniopharyngeal canal is probably the path of spontaneous CSF leaks and midline meningocele through the sella turcica.
Conclusion
A rare anomaly of the adenohypophysis is described with arrested migration occurring within the sphenoid bone surrounded by sphenoid cells. The main imaging features are 1) presence of a large craniopharyngeal canal filled with normal glandular tissue and CSF; 2) dehiscence of the sella turcica floor; and 3) normal development of pituitary stalk, portal circulation, and neurohypophysis. A concomitant, well-developed pituitary vascular network allows the normal function of ectopic adenohypophysis.
A relationship between the intersphenoidal septum and the path of the adenohypophysis migration is reported. The superimposition in gestational time of the first migration wave and duplicate Rathke’s pouch obliteration supports the contention that the insult causing this anomaly occurs between 42 and 45 days of gestational life. This anomaly testifies that the migration of the neurohypophysis and adenohypophysis are independent.
References
- Received February 24, 2003.
- Accepted after revision April 29, 2003.
- Copyright © American Society of Neuroradiology