Abstract
Summary: A 64-year-old woman undergoing protected carotid artery stent placement developed acute stent thrombosis despite pretreatment with combined antiplatelet therapy. A reduced dose of recombinant tissue plasminogen activator and a half-dose bolus of abciximab were administered intra-arterially via superselective catherization followed by systemic intravenous infusion of abciximab for 12 hours. Control angiography showed complete restoration of blood flow paralleled by neurologic improvement.
In carotid stent placement, periprocedural complications include transient or permanent neurologic deficit due to distal thrombus embolization. Two measures are taken to reduce this risk: 1) a well-established antithrombotic regimen that combines antiplatelet premedication and periprocedural heparin to control thrombogenesis (1), and 2) routine use of distal embolic protection devices (2). Technical success with minimal residual stenosis is reported in most series, and acute stent thrombosis is an extremely rare event (2).
Herein we report one case of acute stent thrombosis during filter protected carotid stent placement despite combined antiplatelet therapy with aspirin and clopidogrel, as well as peri-interventional use of heparin. Combined thrombolysis and dethrombosis (3, 4)—so-called facilitated thrombolysis—led to rapid arterial recanalization with minor peri-interventional clinical symptoms.
Case Report
A 64-year-old woman presented with an asymptomatic, calcified, 90% stenosis with rapid progression of the right internal carotid artery (ICA) as judged by velocity criteria by using duplex sonography and a concomitant contralateral 70% ICA stenosis.
After placement of the vascular access sheath, 5000 IU of unfractionated heparin (UFH) were administered intra-arterially. The cerebral angiogram in two planes revealed 85% stenosis of the ICA according to NASCET criteria (5) with no major irregularities or visible appositional thrombus (Fig 1A). A FilterWire EX (Boston Scientific) was placed in the distal right ICA to provide distal embolic protection. A self-expandable 7 × 30 mm Wallstent (Boston Scientific, Natick, MA) was deployed by monorail technique across the stenosis and single poststent dilation was performed with an Agiltrac balloon catheter (5.5 × 30 mm, Guidant, Indianapolis, IN). The patient received 1 mg Atropin intravenously before stent deflation to control potential bradykardia and hypotension due to vasovagal stimulation by compression of the glomus caroticus. A control angiogram obtained immediately after balloon deflation with the protective device in place showed complete occlusion of the right carotid artery within the stent and thrombotic masses surrounded by contrast medium (Fig 1B). Peri-interventional neurologic deficit consisted of mild left facial paresis and dysarthria. Intracarotid injection of 5 mg rtPA was followed by 5 mg recombinant tissue plasminogen activator (rtPA) intra-arterially via the inguinal sheath and a half-dose bolus of abciximab (0.125 mg/kg) via the intracarotid guiding catheter within 9 minutes. Additional heparin (3000 IU) was administered to raise the activated clotting time from <250 to 276 seconds during the prolonged procedure. Carotid recanalization with completely restored blood flow was achieved within 15 minutes of visible onset of acute stent thrombosis (Fig 1C). Moderate vasospasm distal to the segment treated with a stent was observed without visible flow impairment; no further treatment was required. After complete restoration of blood flow, the filter device was removed. Intravenous abciximab therapy was continued for 12 hours at standard dose (0.125 μg.kg−1.min−1).
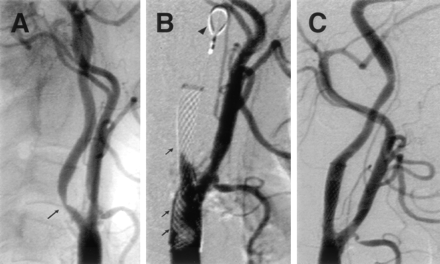
Resolution of acute carotid stent thrombosis by combined administration of rtPA and abciximab.
A, Digital subtraction angiogram obtained before intervention. Arrow indicates 85% stenosis of the right proximal ICA.
B, Small arrows indicate thrombus formation within the proximal stented segment and total thrombotic occlusion of the distal stented segment. Filter WilterEx protection device in situ in the distal ICA (arrowhead).
C, Restoration of blood flow with moderate residual vasospasm distal of the stented segment after successful combined thrombolysis.
Initial MR imaging showed small perfusion deficits within the right precentral gyrus. Control MR imaging after 48 hours confirmed reduced de novo ischemic lesions. Neurologic complications completely resolved within 24 hours, and the patient was discharged 48 hours after intervention with combined antiplatelet therapy for 4 weeks.
Discussion
Acute or subacute thrombosis in carotid stent placement is a rare complication previously described only in patients without combined antiplatelet therapy due to omission of an ADP-receptor antagonist (6, 7). In our patient, the reason for acute stent thrombosis remained obscure, because the standard antithrombotic regimen, including combined antiplatelet therapy, was administered and the intervention was performed in a routine fashion by using monorail technique and distal filter protection without repeat poststent dilation or any evidence of dissection. Routine laboratory parameters including prothrombin time, activated partial thromboplastin time, platelet count, and C-reactive protein at baseline were within the normal range; however, poorly controlled diabetes indicated by an elevated level of glycated hemoglobin (HbA1c 7.6 rel%) may have predisposed to a hypercoagulable state in this patient (8).
Peri-interventional carotid stent thrombosis requires (ideally) rapid intra-arterial–intracarotid initiation of reperfusion therapy to limit time of ischemia, reperfusion injury, and associated symptomatic intracranial hemorrhage (9). Thrombolysis with rtPA is the only thrombolytic therapy approved for the treatment of acute ischemic stroke (10). Its major limitations, however, consist of the high incidence of intracranial bleeding complications and the short time window for application within 3 hours from onset of symptoms. In addition, for the treatment of peri-interventional thrombembolic events in neuroendovascular procedures, rtPA did not significantly improve recanalization rates and clinical outcome (11).
By contrast, early thrombus disaggregation—termed “dethrombosis” (3)—by using the GP IIb/IIIa inhibitor abciximab as adjunct for carotid artery stent placement reportedly reduced adverse peri-interventional events (12, 13). Furthermore, abciximab for treatment of acute ischemic stroke proved safe and efficient with a positive trend toward a higher rate of minimal residual neurologic disability after 3 months (14).
A combined therapeutic regimen consisting of dethrombosis (abciximab) and fibrinolysis (by using alteplase)—termed “facilitated thrombolysis”—was studied in acute myocardial infarction (MI) (15) and was also reported in selected cases of acute basilar artery occlusion (16). Because of the increased bleeding risk observed with abciximab after full-dose thrombolysis in rescue or urgent angioplasty for acute MI, (17) study protocols for facilitated thrombolysis employing a lytic agent in combination with a GP IIb/IIIa antagonist were designed with half the usual dose of the fibrinolytic agent (15). Similarily, rtPA in a reduced dosage of only 10 mg/h (for a total of 2 hours) was given intra-arterially together with full-dose (bolus and infusion) intravenous abciximab for recanalization of acute basilar artery occlusion (16).
In our patient, both rtPA and the half-bolus abciximab were administered intra-arterially via superselective catheterization of the occluded carotid artery. This strategy ensures high local drug concentration associated with improved efficacy (18, 19). Therefore, a reduced dosage of both agents was used employing a careful “wait and watch” tactic; however, because fibrinolytic agents simultaneously exert clot-dissolving and procoagulant actions, which may in fact precipitate reocclusion of the infarct-related artery after thrombolysis (20), we decided to perform facilitated thrombolysis by using rtPA and abciximab simultaneously, rather than fibrinolysis alone.
Heparin was used to titrate the activated clotting time between 250 and 280 seconds, which is recommended for adjunct abciximab therapy. We doubt that UFH by itself had a prothrombotic effect in our patient, although it was previously shown to induce platelet P-selectin expression, to increase platelet surface bound fibrinogen and to significantly augment ADP-dependent platelet activation, when compared with low-molecular-weight heparin (21). These data were derived from whole blood flow cytometry and platelet aggregation assays. Whole blood flow cytometry performed in our patient showed postinterventional P-selectin expression on platelet-monocyte aggregates, a marker known to be more sensitive than direct platetelet P-selectin expression (22), that was comparable to the baseline level (pre 22.3% versus post 22.2% P-selectin-positive monocyte-platelet aggregates). This result, however, was not unexpected because of the suppressive effect of abciximab on the platelet load of individual monocytes previously described in in vitro (23) and in vivo (13) models.
The emergency situation of peri-interventional acute carotid stent thrombosis seems particularly suitable for facilitated thrombolysis (4), because the time window of ischemia is minimal (which is a determinant of reperfusion injury and associated bleeding risk), and the freshly formed thrombus may be prone to dethrombosis (3). Thus, coadministration of abciximab and rtPA may be beneficial with respect to both reduced time for arterial recanalization and suppression of recurrent thrombosis.
Conclusion
In our case, the bailout strategy by using the synergistic effect of thrombolysis and dethrombosis led to rapid revascularization after carotid stent thrombosis with minimal de novo ischemic lesion formation and transient neurologic deficit.
References
- Received October 13, 2003.
- Accepted after revision May 1, 2004.
- Copyright © American Society of Neuroradiology