Abstract
BACKGROUND AND PURPOSE: Most neurologic events that complicate the carotid artery stent placement procedure are embolic. Strategies are being developed and evaluated for their ability to minimize the clinical embolic risk. The purpose of this study was to determine the feasibility, safety, and technical considerations of performing carotid artery stent placement with use of a distal balloon antiembolization system.
METHODS: Carotid artery stent placement with use of a commercially available distal balloon antiembolization system was performed in 303 patients (325 vessels). Clinical neurologic complications were adjudicated at the time of intervention and at 1 month.
RESULTS: Technical success was achieved in all but one case, with reduction of the mean percentage carotid artery stenosis from 79 ± 9% to 3 ± 8%. Intolerance to internal carotid artery occlusion occurred in 11 patients (3.6%); in one patient the procedure was aborted, and in 10 the procedures were expeditiously completed with no adverse neurologic events. The 30-day combined rate of all occurrences of stroke and death was 2%. These events included three (0.9%) retinal emboli, three (0.9%) minor strokes, and one (0.3%) fatal stroke from brain hemorrhage, all of which occurred during the procedure with no events occurring after discharge. In addition, two (0.6%) transient ischemic attacks and three (0.9%) reperfusion syndromes occurred, with the patients experiencing complete recovery.
CONCLUSION: Carotid artery stent placement with use of the distal balloon protection system is feasible and safe, and the short-term outcomes appear to be favorable. Optimal application of proper technique is important.
Carotid artery stent placement is being investigated as an endovascular alternative to carotid endarterectomy for treatment of obstructive atherosclerotic carotid artery disease (1–3). Although the goal of both procedures is identical (ie, stroke prevention), stent placement offers patients a less invasive means of achieving this goal. Despite reported favorable experiences, an appreciable risk exists for embolic neurologic events during this procedure (1–3). Obstructive carotid artery lesions are known to contain friable thrombotic and atherosclerotic components that can be released during intervention and are responsible for most of these neurologic events (4). Investigations are ongoing to evaluate a variety of antiembolization strategies for their efficacy in capturing embolic matter released during carotid artery stent placement (4–7). In our overall experience in 1397 carotid artery stent placement procedures (588 with an antiembolic system), the frequency of all strokes was decreased from 5.6% to 1.9% (J.J.V., unpublished data, 2004). The purpose of our current study was to perform a prospective evaluation of carotid artery stent placement with a distal balloon antiembolization system and to describe the technical considerations in the application of this strategy.
Methods
Between February 2000 and September 2002, 303 patients (325 carotid arteries) underwent carotid artery stent placement with a distal balloon antiembolization system (GuardWire Temporary Occlusion and Aspiration System; Medtronic AVA, Danvers, MA) (Fig 1). All patients were treated under a prospectively defined protocol that was approved by the institutional review board, and all gave written informed consent for the procedure. Patients were referred based on duplex ultrasonographic or MR angiographic findings. All patients underwent cervicocerebral angiography in our laboratory and were enrolled in this protocol if they were symptomatic (38%) or asymptomatic (62%) with extracranial internal carotid artery (ICA) stenosis of 50% or greater, or 70% or greater, respectively, based on North American Symptomatic Carotid Endarterectomy Trial criteria. Quantitative carotid angiography was performed before and after the procedure. Included in the protocol was a neurologic examination performed by an independent board-certified neurologist before and immediately after completion of the stent placement procedure, and at 30 days and 6 and 12 months after the procedure. Patients were excluded from the study for the following reasons: stroke within 7 days before the procedure; baseline neurologic deficit that was likely to confound determination of the study clinical end points (National Institutes of Health Stroke Scale [NIHSS] score ≥ 15); more than one ipsilateral carotid stenosis requiring treatment; a cardiac source of emboli such as atrial fibrillation; the presence of known intracranial tumor or vascular malformation; contralateral carotid artery occlusion, severe ipsilateral intracranial carotid stenosis, and insufficient or nonexistent collateral blood supply to the ipsilateral hemisphere; and renal insufficiency (serum creatinine level ≥ 2.5 mg/dL).
The GuardWire protection system.
Patients’ baseline clinical characteristics are summarized in the Table. The mean age was 68 ± 9 years (range, 57–89 years) with 48 patients (16%) being 80 years or older. One hundred fifteen patients (38%) had symptoms attributable to the treated carotid artery within 3 months preceding the intervention (13% had stroke, 19% had transient ischemic attack, and 6% had amaurosis fugax).
Demographic and clinical characteristics of the 303 patients
All patients were premedicated with clopidogrel 75 mg once per day and aspirin 100–325 mg twice per day for a minimum of 4 days before the procedure. Clopidogrel was continued for 30 days after stent placement, and aspirin was continued indefinitely. All stent placement procedures were performed by using a standard technique as previously described (8). A 6F, 90-cm-long guiding sheath (Shuttleā, Cook Inc, Indianapolis, IN) was used to access the carotid artery. A single bolus of intravenous heparin (4000 U) was administered through the sheath at the beginning of the procedure, with no heparin administered after the procedure. Only local anesthesia (groin) was used. Intraprocedural monitoring was accomplished by being in verbal contact with the patient and having the patient squeeze a toy placed in the contralateral hand after every step of the intervention. Glycoprotein IIb-IIIa antagonists were not used. All percutaneous transluminal angioplasty balloons used were low-profile, coronary-type balloons, such as Ranger (BSC), Maverick (BSC), or Adante (DSC) for predilation and Savvy (Cordis) or Gazelle (BSC) for stent postdilation. Only self-expanding stents were used, such as Wallstent (BSC), a variety of Smart stents (Cordis), and AVE stents (Medtronic Corp). All stents were oversized to the width of the ICA and 20–40 mm in length.
Optimal utilization of the occlusion balloon antiembolization strategy is dependent on patient and lesion selection and appropriate application of the system by using various technical maneuvers to overcome anatomic challenges. Advancing the GuardWire temporary occlusion catheter into a moderate stenosis with a nonangulated bifurcation or with only a moderately angulated takeoff of the ICA poses no technical challenge (Fig 2). In both instances, the extended tip of the temporary occlusion catheter can be shaped and the system can be easily negotiated through the stenotic lesion. However, in cases of preocclusive stenoses, heavily calcified or eccentric lesions, severe distal kinks or tortuosity of the ICA, especially when the ICA takeoff is considerably angulated, it can be difficult and sometimes impossible to advance the temporary occlusion catheter into the distal ICA (Figs 3 and 4). In these situations, placement of a side wire or “buddy wire,” eventual gentle predilation, or a combination of both (Fig 3) may facilitate advancement of the temporary occlusion catheter.
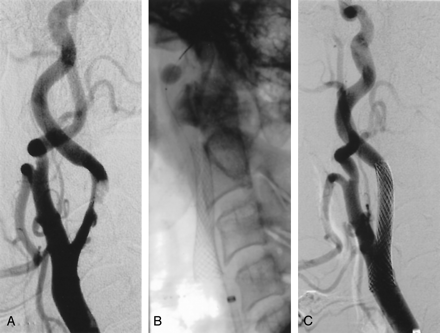
Application of the GuardWire protection system in a 62-year-old man with recent multiple transient ischemic attacks.
A, Right carotid angiogram shows significant stenosis in the ICA.
B, Angiogram shows that the balloon on the temporary occlusion catheter is inflated in the distal cervical segment of the ICA (stent is deployed and postdilated).
C, Carotid angiogram obtained after stent placement.
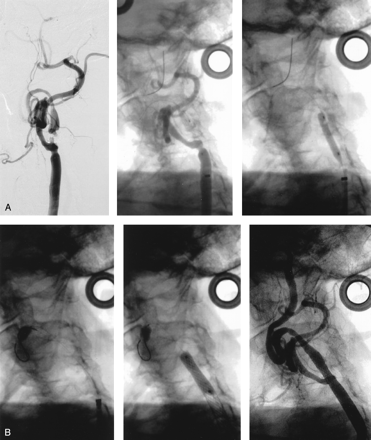
A and B, Illustration of the buddy wire technique in an 82-year-old woman.
A, Left, Left carotid angiogram shows near occlusion of the ICA and significant tortuosity of the distal ICA. Initial attempt to pass the temporary occlusion catheter of the GuardWire protection system through the stenosis was unsuccessful due to the severity of the stenosis. Middle, Angiogram shows a buddy wire positioned through the stenosis and the distal tortuous segment of the ICA. Blood flow through the subocclusive stenosis is hindered by the guidewire with the predilation balloon. Right, Angiogram shows the stenosis predilated with a 3 × 20-mm coronary balloon.
B, Left, Angiogram shows the inflated balloon of the temporary occlusion catheter positioned distal to the stenosis along the buddy wire. Middle, Angiogram shows the postdilated stent. The buddy wire was removed before deployment of the 10 × 24-mm Wallstent. Right, Angiogram obtained after carotid artery stent placement.
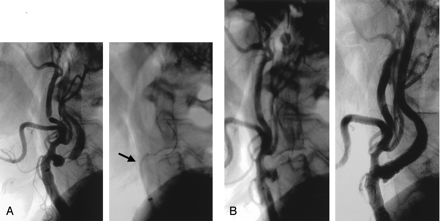
A and B, Illustration of the buddy catheter technique in a 70-year-old man.
A, Left, Left carotid angiogram shows near occlusion of the ICA. The 90° takeoff angulation made advancement of the GuardWire temporary occlusion catheter into the stenotic ICA impossible. Right, Angiogram shows the buddy catheter (5F, 125-cm JR4, arrow) positioned through a 6F guiding sheath, with its tip pointing into the angulated ICA lesion. The temporary occlusion catheter is advanced through the stenosis into the distal segment of the ICA.
B, Left, Angiogram shows that the inflated balloon of the temporary occlusion catheter is occluding the ICA. Right, Angiogram obtained after carotid artery stent placement. Two 10 × 30-mm Smart stents were deployed.
In using the buddy wire maneuver, the lesion is first crossed with a properly angulated, highly torqueable 0.014-inch guidewire (eg, Balance; Guidant Inc), which is placed within the distal ICA. A low-profile angioplasty balloon (eg, Ranger, 2 × 40-mm; SciMed Corp) is then advanced into the stenosis and a low-pressure dilation, if needed, is performed. The balloon catheter is then advanced distally into the ICA, and the 0.014-inch guidewire is exchanged for a 0.014-inch extrasupport wire (eg, Stabilizer-Plus; Cordis Inc). This support guidewire straightens the ICA angulations, and through the now partially predilated stenosis and straightened ICA, the temporary occlusion catheter can be advanced into the distal ICA (Fig 3B). If the tortuosity or angulation of the ICA is not severe, the 0.014-inch support guidewire can be withdrawn after placement of the temporary occlusion catheter. Otherwise, the support wire is left in situ, and after predilation with a regular 4 × 40-mm balloon and depending on the configuration of the bifurcation and the cervical segment of the ICA, the buddy wire is a) withdrawn when the stent is in position to be deployed; b) left in situ and removed after the stent is deployed and postdilated (“jailed” buddy wire); or c) left in situ throughout stent deployment, postdilation, application of the Export catheter, and until the temporary occlusion catheter is retracted. The “jailed” support wire is then removed. It should be noted that all of these maneuvers might provoke significant spasm. The spasm typically resolves after all wires are removed from the ICA. In our experience, these maneuvers have never resulted in any vascular damage or increased embolic load (9). The buddy wire strategy not only facilitates the advancement of the temporary occlusion catheter through the stenosis, but also prevents an unpleasant struggle to try to advance and deploy the stent in a tortuous and angulated ICA.
If the origin of the ICA is so severely angulated that the buddy wire strategy is unsuccessful, a “buddy catheter” system can be used to facilitate placement of the temporary occlusion catheter (Fig 4). An appropriately shaped, 5F, 125-cm-long catheter (eg, 4-cm curved right Judkin catheter [JR4], or an internal mammary artery catheter) is advanced through the guiding sheath and placed with its tip pointing into the ostium of the ICA. Then, through this catheter, the temporary occlusion catheter or a buddy wire is advanced through the stenosis into the ICA. The buddy catheter system can also be used to advance the temporary occlusion catheter through a very tight or fibrous stenosis. One has to remember that the temporary occlusion catheter or the buddy wire length has to be 300 cm when using this strategy. The buddy catheter is then removed over the wire, and the carotid artery stent placement procedure is carried out. The level of the ICA at which the occlusion balloon is inflated is of extreme importance. It should be at least 30 mm distal to the intended distal end of the stent. If this distance is not maintained, the stent delivery system can become imbedded into the temporary occlusion catheter system, with ensuing technical difficulties.
Device Description
The GuardWire system is designed to contain embolic material released during stent placement, preventing the material from reaching the cerebral circulation. The GuardWire is a temporary occlusion and aspiration system that has three components: the temporary occlusion catheter, the MicroSeal adapter, and the Export aspiration catheter. The temporary occlusion catheter is a “balloon-on-a-wire” catheter with a distal elastomeric occlusion balloon (Fig 1). It has a lubricating coating and a flexible radiopaque tip. Crossing profile is 0.036-inch (1 mm, 2.9F). The MicroSeal adapter is used with the temporary occlusion catheter (Fig 1) for the purpose of controlled volumetric inflation and deflation of the occlusion balloon. The first generation of the GuardWire, used earlier in this study, allowed the occlusion balloon to be inflated to two sizes: 5.5 and 6 mm. The second and present generation version has an inflation device that can be set to inflate the occlusion balloon to between 3 and 6 mm in 0.5-mm increments. The Export aspiration catheter is a monorail design that has a distal radiopaque tip marker and a proximal Luer-Lock port. The older GuardWire system had a 300-cm-long 0.018-inch temporary occlusion catheter; the present system is a 0.014 inch and 190 cm temporary occlusion catheter. Because of the shorter length of the current generation GuardWire catheter, only monorail balloons and stents can be used.
Definitions
Technical success refers to the ability to access the carotid artery and to successfully apply the GuardWire system and deploy the stent, with a residual stenosis of 30% or less. Transient ischemic attack is a neurologic deficit that totally resolved in 24 hours. Amaurosis and amaurosis fugax is a permanent or temporary retinal event. Minor stroke refers to a neurologic deficit with an increase in the NIHSS score by three or fewer points of longer duration than 24 hours or complete resolution of neurologic symptoms within 30 days. Major stroke refers to a neurologic deficit with an increase in the NIHSS score by four or more points persisting for more than 30 days.
Study End Points
The study end points included technical success and procedural and composite 30-day frequency of minor stroke, major stroke, death, and myocardial infarction.
Results
Successful carotid artery stent placement was achieved in all cases except one. This failure occurred due to combined significant elongation of the aortic arch and tortuosity of the common carotid artery that prevented placement of the 6F sheath. In 25 patients (8%), advancement of the temporary occlusion catheter through a high-grade stenosis with tortuous vasculature was facilitated by prior placement of a side wire buddy wire and/or predilation (with a 2-mm balloon). In five cases (1.5%), a buddy catheter was required. In eight cases (2.5%), the occlusion balloon spontaneously deflated at an early stage of the procedure, presumably due to a defective valve, and the system was successfully replaced on each occasion. This happened with equal frequency in both the older and newer systems. The valve has to be tested before the system is used. No neurologic events occurred in any of these patients. Eleven patients (3.6%) did not tolerate ICA occlusion. Of these, one patient developed ischemic seizure 17 seconds after ICA occlusion. The procedure was aborted, and carotid artery stent placement was performed on a later day by using a filter antiemboli device. The remaining 10 patients became confused and developed mild to moderate contralateral hemiparesis at 6–7 minutes after ICA occlusion. The intervention was completed, and all patients completely recovered immediately after deflation of the balloon.
The combined rate of all occurrences of stroke and death at 30 days was 2% (a total of seven events), with all of the events occurring during the procedures and none after the patients were discharged. These events included three (0.9%) retinal emboli, three (0.9%) minor strokes, and one (0.3%) fatal stroke. The latter occurred in a patient with symptomatic high-grade (90%) right ICA stenosis and a contralateral occlusion of the left ICA who died from a massive subarachnoid hemorrhage (originating in the high left sylvian fissure) that occurred immediately after the stent placement procedure (10). Notably, the preprocedural brain MR images and cerebral angiograms in this patient were normal. Of the three patients who developed minor stroke, two recovered completely within 30 days. Of the three patients who developed retinal events, two recovered completely within 30 days. In addition, two (0.6%) cases of transient ischemic attack and three (0.9%) cases of reperfusion syndrome occurred. Reperfusion syndrome is usually seen in hypertensive patients and in patients with critical (>90%) stenosis. Clinically, the patients present with transient postprocedural confusional state with headaches, occasionally associated with transient localizing symptoms, and not associated with angiographic abnormalities. A CT or MR image will often show mild hemispheric swelling with effacement of sulci or suggestion of luxury perfusion. The symptoms usually resolve over 24–48 hours, with good control of blood pressure. All of the latter three patients recovered completely.
Angiographically visible vascular dissection of the ICA at the occlusion balloon site occurred in 18 patients (5.9%), with no associated neurologic events (Fig 5). In 13 of these patients, the dissection was notable for retention of contrast medium in the vessel wall; in three, the dissection was notable for a small non–flow-limiting dissection flap; and in one patient, the dissection was characterized by deep disruption of the vessel wall with 50% non–flow-limiting stenosis. One patient had a significant dissection that resulted in an occlusion of the ICA, with no associated neurologic deficit. This was successfully recanalized and a stent was placed.
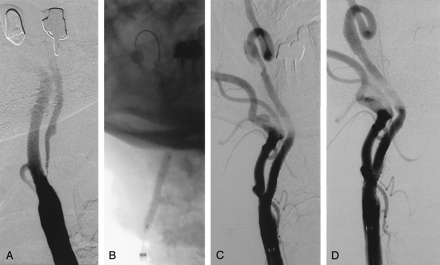
Dissection caused by occlusion balloon in a 79-year-old man.
A, Right carotid angiogram shows near occlusion of the ICA.
B, Angiogram shows that the temporary occlusion balloon is inflated. Predilation was performed with a 4 × 40-mm balloon.
C, Follow-up angiogram obtained after carotid artery stent placement shows significant dissection at the level of the previously placed occlusion balloon. Satisfactory result of carotid artery stent placement with a 9 × 30-mm stent (Medtronic AVE).
D, Follow-up angiogram after placement of a 4 × 23-mm coronary stent in the dissected segment of the ICA.
Discussion
The distal balloon protection technique in brachiocerebral angioplasty was first described by Vitek et al in 1983 (11). The proximal right common carotid artery was temporarily occluded with a miniballoon during balloon angioplasty of the innominate artery. Theron et al (12), in 1990, described a triple coaxial catheter that allowed carotid angioplasty with temporary ICA occlusion and subsequent aspiration of the debris, as well as saline flushing into the external carotid artery. More recently, Henry et al (13) published their experience with carotid artery stent placement in 163 patients (173 treated arteries) and reported an embolic complication rate of 4.6%. In 32 patients who were treated by using Theron’s protection technique, the complication rate of 6.2% was attributed to technical problems with the triple coaxial catheter. Henry et al (5) later published a series of 48 patients (54 carotid arteries) in which the GuardWire protection system was used and reported a 100% technical success rate and a 1.8% embolic neurologic complication rate.
The current study further supports the favorable results of the prior reports with a low rate of embolic neurologic events during carotid artery stent placement. Important to note is the absence of major disabling embolic neurologic events. In the whole group of 303 patients, the only major stroke was hemorrhagic. Particular beneficial effect of the distal balloon protection technique was apparent in the elderly patients. Older age (≥ 80 years) is an important independent predictor of procedural neurologic complications in patients undergoing carotid artery stent placement without antiembolization protection (14). In our early experience, a small group of patients older than 80 years had a significantly higher complication rate during carotid artery stent placement without antiemboli protection in comparison with patients younger than 80 years: 14.7% versus 4.3%, respectively (1).
Balloon occlusion of the ICA during carotid artery stent placement may have a significant effect on cerebral perfusion. Henry et al (5, 13) reported intolerance symptoms in one patient (5%) during balloon occlusion; however, they applied two-stage distal balloon occlusion in 18 (34%) patients in their series by briefly reestablishing blood flow within the ICA midway through the procedure, which contributed to improved tolerance. Our practice has been to perform cerebral angiography with selective injection of bilateral carotid arteries and at least one vertebral artery before carotid artery stent placement. We consider patients with a poorly developed collateral circulation to the affected hemisphere unsuitable for the occlusion balloon antiembolization strategy. Despite this precaution, 3.6% of our patients developed intolerance symptoms during ICA occlusion. Patients thought to be unsuitable for ICA occlusion, based on our screening angiography, underwent carotid artery stent placement with use of a different antiembolization strategy. A complete circle of Willis is present in 50–60% of the population. Variations in the presence or absence of the anterior-posterior communicating arteries are frequent. If not present, leptomeningeal anastomoses are usually insufficient to maintain adequate blood supply to the cerebral hemisphere for a longer period of time when the ipsilateral ICA is occluded. Reversal of the flow within the ipsilateral ophthalmic artery can also contribute to better tolerance to ICA occlusion. Intolerance to ICA occlusion can be instantaneous and dramatic, manifesting immediately with contralateral hemiplegia, aphasia, or ischemic seizure. In these situations, the occlusion balloon must be immediately deflated. This occurred in only one patient in our series. The remaining patients (most older than 80 years) showed only partial intolerance to ICA occlusion, becoming symptomatic approximately 5–6 minutes after ICA occlusion. In this situation, the procedure is expeditiously completed. All of our 10 patients who had partial intolerance to ICA occlusion had complete neurologic recovery immediately after deflation of the occlusion balloon, without any sequelae. Alternatively, ICA occlusion can be staged (13), with the occlusion balloon deflated after removal of 40 mL of blood from the stump of the ICA by using the Export aspiration catheter. After the patient recovers, ICA occlusion is reestablished and the intervention is completed.
Embolization to the intracranial circulation may still occur when the occlusion balloon antiembolization strategy is used during carotid artery stent placement. This may occur through the collateral circulation, when distal occlusion is incomplete, or during deflation of the protection balloon (9). It is important to achieve complete occlusion of the distal ICA and maintain it throughout the procedure and until the final aspiration is completed. Microemboli, detected with transcranial Doppler ultrasonography, are frequently detected during deflation of the protection balloon. A “dead zone” (ie, a space that is inaccessible to the Export aspiration catheter) exists between the occlusion balloon and the arterial wall and could be responsible for these microemboli. In our experience, these microemboli have not been associated with any clinical events. The occlusion balloon is sized (by measurement or experience) to the distal ICA at the beginning of the procedure. Because of underfilling and collapse of the cervical ICA when the ostial segment is severely stenosed, the distal ICA diameter may be underestimated. After predilation of the lesion, it is important to reassess the occlusion of the ICA by means of slow-injection angiography (to avoid flushing the eventual emboli from the stump of the ICA into the external carotid or proximal common carotid artery), and if necessary the size of the protection balloon should be adjusted to achieve complete occlusion. The occlusion balloon should not be oversized; this may increase the risk for intimal damage or dissection to the ICA at the level of placement of the occlusion balloon. The alternative pathway to the intracranial circulation, when the ICA is occluded, is the external carotid artery. Under normal circumstances, communication between the external carotid artery and the ICA (and to a lesser extent between the external carotid and vertebral arteries) is almost always present even if not seen on conventional angiograms. Of all possible connections (facial artery, ascending pharyngeal artery, multiple branches of the internal maxillary artery, superficial temporal artery), the ophthalmic artery is the clinically most important. In the healthy individual, retrograde flow through the ophthalmic artery into the ICA is immediately established upon occlusion of the extracranial segment of the ICA. In patients with high-grade stenosis of the ICA, these anatomically preexistent anastomoses enlarge. Intracranial angiography performed in 11 of our patients during distal ICA occlusion showed the intracranial ICA and ipsilateral middle cerebral artery partially opacified by retrograde flow through the ophthalmic artery. Transcranial Doppler ultrasonographic monitoring in some of these patients confirmed this phenomenon of microembolization into the ipsilateral middle cerebral artery despite complete ICA occlusion (9). This may explain three cases of retinal embolus in our series and a case of amaurosis fugax in another report (13). In our judgment, it is inappropriate to apply the saline flushing technique of the stump of the ICA into the external carotid artery. One also should not forcefully inject contrast material into the common carotid artery when the ICA is occluded. This could result in flushing embolic material into the external carotid artery. One should be especially careful if the external carotid artery is occluded as well; the emboli could be flushed into the proximal common carotid artery, into the arch, or into the right vertebral artery (innominate and right subclavian passway). A complete brachiocephalic angiographic examination is mandatory before carotid artery stent placement to identify any congenital or pathologic vascular contraindications to the ICA balloon occlusion technique. If a large external-to-internal carotid artery or external carotid artery-to-vertebral artery anastomosis is present on the preinterventional cerebral angiogram, use of the distal occlusion device should be avoided during carotid artery stent placement. We also have to take into consideration the possible presence of congenital, embryonic anastomoses between the anterior and posterior circulation, such as the proatlantal, hypoglossal, otic, and trigeminal arteries.
Advancement of the temporary occlusion catheter into the ICA, particularly with associated straightening of a tortuous ICA, can produce vascular spasm. The protection balloon exerting pressure on the ICA wall can also cause spasm. The position of the inflated protection balloon has to be kept stable throughout the procedure. Any untoward movement of this balloon may induce vascular spasm as well. Flow-limiting spasm is rare and almost always disappears after removal of the temporary occlusion catheter (and eventually buddy wire) from the ICA. It is also helpful after completion of the carotid artery stent placement procedure to retract the guiding sheath to the proximal common carotid artery. This maneuver relaxes the bifurcation and stretches the ICA, which by placing the sheath or the guiding catheter within the distal common carotid artery can push the bifurcation cephalad, creating artificial kinks in the ICA. Severe spasm may take several minutes to disappear. Administration of intraarterial nitroglycerin (100–200 mg) will facilitate its resolution.
It is important to distinguish spasm from dissection (Fig 5). Dissection at the level of the occlusion balloon can be caused by oversizing the balloon or by not having the occlusion balloon well stabilized throughout the procedure (ie, by its craniocaudal motion). The most common cause of distal ICA dissection is oversizing the occlusion balloon. The diameter of the ICA at the level of the intended balloon occlusion can be measured at the beginning of the procedure. However, this measurement is not reliable for achieving a complete occlusion of the ICA throughout the procedure. The size of the ICA varies with blood flow. In a severely stenosed ICA, the vessel is partially collapsed and may significantly expand after predilation and stent placement. An appropriately sized occlusion balloon at the beginning of the procedure may become nonocclusive during the procedure. Ideally, the occlusion balloon should be more compliant than the present system and change in size and shape depending on the vessel diameter without exerting too much pressure on the vessel wall. The new-generation system is more flexible with the possibility of inflating the occlusion balloon 3–6 mm by 0.5-mm increments. The occlusion balloon can also be inflated in the petrous segment of the ICA. Although at this level the ICA cannot be distended, owing to its confinement within bone, this location of the occlusion balloon significantly elongates the occluded segment of the ICA from which the debris has to be aspirated and may decrease the efficacy of the system. In the presence of significant tortuosity of the ICA, this location would also be inconvenient for advancement of the Export aspiration catheter. At the end of the stent placement procedure, it is important to carefully examine the neck segment of the ICA at which the occlusion balloon was inflated. This segment of the ICA can be easily missed on the follow-up angiograms obtained after the procedure, as it is located at the level not included on the exposure of the bifurcation and excluded on the intracranial study.
Conclusion
Carotid artery stent placement with distal balloon protection is safe and feasible. The short-term outcome appears to be favorable and needs to be confirmed in multiple institutions. The proper application of this technique is important to ensure the safety and successful outcome of the carotid artery stent placement procedure. In cases of severe tortuosity and angulation of the ICA origin, the buddy wire technique and/or combination with the buddy catheter maneuver and appropriate predilation facilitates placement of the the GuardWire system into the distal ICA. Significant spasm of the ICA is rare. Dissections are uncommon and usually self-limiting. Preprocedural brachiocephalic angiography is essential in selecting patients for this protection strategy. The principal disadvantage of the GuardWire protection system is the temporary occlusion of the ICA. This excludes an important percentage of patients from undergoing this embolic protection strategy. Patients with isolated hemisphere occlusion of the contralateral ICA, as well as those with a well-developed collateral circulation through the external carotid or vertebral artery, are not suitable for carotid artery stent placement with distal ICA occlusion.
References
- Received August 2, 2004.
- Accepted after revision October 26, 2004.
- American Society of Neuroradiology