Abstract
PURPOSE: Our goals were to assess image quality of time-resolved contrast-enhanced MR angiography (CE MRA), by using 3D data acquisition along with a parallel imaging technique that can improve temporal resolution and to compare this technique with 3D-time-of-flight (TOF) MRA in the postoperative assessment of extracranial (EC)–intracranial (IC) bypass surgery.
METHODS: On a 1.5T imaging system, we performed CE MRA by using a 3D fast field-echo sequence in combination with a parallel imaging technique, to obtain images in the coronal plane centered at the postoperative site. Our patient group comprised 17 patients, including 13 after superficial temporal artery–middle cerebral artery (MCA) anastomosis, 3 after external carotid artery-MCA anastomosis, and one after extracranial vertebral artery–posterior cerebral artery anastomosis. Visualization of the anastomosis and the distal flow on the CE-MRA images was assessed comparatively with that on 3D-TOF MR angiograms obtained at the same time. In 6 patients, we also compared the efficiency of visualization on CE-MRA images with that on conventional angiograms.
RESULTS: A temporal resolution of 0.8 s/frame could be achieved with the technique employed. The bypass was better demonstrated postoperatively on CE-MRA images than on 3D-TOF MR angiograms in 13 patients (76%), whereas the 2 methods were equivalent in 4 patients (24%). Good correspondence of results was observed in the 6 patients for whom CE MRA and conventional digital subtraction angiography (DSA) images were compared.
CONCLUSION: CE MRA by using the parallel imaging technique can increase image acquisition speed with sufficient image quality. This technique is at least equivalent to 3D-TOF MRA to evaluate the postoperative status of EC-IC bypass.
Extracranial–intracranial (EC-IC) bypass surgery has been used to increase the cerebral blood flow and halt extension of the affected area or reduce the risk of future strokes in patients with ischemic cerebrovascular disease (1–3). Furthermore, several kinds of bypass surgery are performed in patients with cerebral aneurysm, in which the parent artery often has to be sacrificed. In these cases, conventional angiography has been used as the most reliable method to assess the patency of the EC-IC anastomosis postoperatively. Recently, however, several other modalities—including MR angiography (MRA), sonography, and CT angiography—have also been reported to be useful for such assessment (4–7).
Time-resolved contrast-enhanced (CE) MRA, in which a rapid T1-weighted sequence is performed in combination with bolus intravenous injection of a gadolinium-based contrast agent, provides useful anatomic and hemodynamic information concerning intracranial vascular lesions. To generate “time-resolved” images, 2D sequences were previously used (8–14). More recently, however, a technique using an effective k-space sampling technique that provides good temporal resolution even with 3D data acquisition has been reported (15). Multiple arrays of receiver coils are used in the parallel imaging technique, where the imaging time is reduced by a factor equal to the number of coils used (16, 17). Application of this technique to contrast-enhanced carotid MRA has already been reported (18).
In this study, we assessed whether CE MRA with 3D data acquisition by using parallel imaging as well as an efficient method of k-space sampling might be useful in the postoperative assessment of EC-IC bypass, by providing images comparable to those obtained in conventional 3D-time-of-flight (TOF) MRA and digital subtraction angiography (DSA).
Methods
We obtained CE-MRA images from 17 consecutive patients referred to us for MR imaging following EC-IC bypass during a 2-year period. The subjects consisted of 7 men and 10 women aged 23–74 years (mean age, 51.2 years). Of these patients, 13 had undergone superficial temporal artery (STA)–middle cerebral artery (MCA) anastomosis for MCA disease (n = 8), internal carotid artery (ICA) disease (n = 2), or Moyamoya disease (n = 3), 3 had undergone external carotid artery (ECA)–MCA anastomosis for ICA disease by using a radial artery graft, and one had undergone extracranial vertebral artery (VA)–posterior cerebral artery (PCA) anastomosis by using a radial artery graft for bilateral occlusion of the vertebral arteries due to dissection and its therapy. The interval between surgery and the MR study ranged from 2 days to 4 years and 10 months (mean, 15.1 months).
On a 1.5T imaging system, in addition to obtaining conventional T1-weighted, T2-weighted, and fluid-attenuated inversion recovery (FLAIR) images, and MR angiograms by using a 3D-TOF technique, we also performed CE MRA by using a 3D fast field-echo sequence in conjunction with a parallel imaging technique and segmented k-space sampling technique. For the parallel imaging technique, we employed a 5-channel array coil with a time reduction factor of 2.0. As described in detail elsewhere (19), for the segmented k-space sampling technique, we divided the k-space into 6 segments and acquired data from the central part and one of the remaining 5 segments alternately—that is, after collecting the data from the central segment, the data from the remaining 5 segments were collected, starting with the closest one. The accumulated data were then reconstructed as one image. This technique allowed the speed of data collection to be trebled as compared with that in the conventional methods of reconstruction. In all the patients in this series, the scanning was conducted in the coronal plane, by placing a section centered at the postoperative site as visualized in the precontrast MR images. The other imaging parameters were as follows: TR/TE/excitations, 3.1/0.9/1; flip angle, 20°; imaging matrix, 128 × 256; field of view, 26 × 28 cm; partition thickness, 7.5 mm; number of partitions, 10 (section thickness, 75 mm). No interpolation was done in postprocessing. At the start of the scanning, 7 mL of gadolinium was injected via the antecubital vein at the rate of 3 mL/s, followed by 15 mL of saline for flushing, by using a power injector. The postprocessing that involved maximum-intensity projection of the acquired data, obtained after subtracting a set of source images in the early phase from subsequent sets, and gray-scale reversal usually took <15 minutes. For 3D-TOF MRA used in comparison, we performed image reconstruction without employing targeted maximum-intensity projection.
Visual assessment of the CE-MRA images from all of the 17 patients was performed, and the visualized images of the bypass on both the CE-MRA images and 3D-TOF MR angiograms were scored on a 3-point scale as follows: 2 = good; 1 = fair; and 0 = poor. A score of 2 was assigned when the anastomosis and secondary or more distal branches of the recipient vessels connected to the anastomosis or graft artery could be demonstrated. A score of 1 was assigned when the anastomosis and only recipient branches immediately distal to the anastomosis or graft artery could be demonstrated. A score of 0 was assigned when neither the anastomosis nor any of the recipient branches could be visualized. As we assessed both EC and IC postoperative vessels, it was difficult to include a precise control group in this study for comparison. In 6 patients, we also assessed conventional DSA images obtained within one month after the MR study in terms of the visualization of the bypass flow. All the CE-MRA images and MR angiograms were reviewed by 2 radiologists in a blinded fashion to the finally assessed status of the bypass surgery and/or the findings in the conventional DSA images. In cases whose conventional DSA was not available, we used 3D-TOF MR angiograms as the standard determining the conditions of the bypass by comparison with the corresponding EC and IC vessels on the contralateral side. In cases of disagreement in the assessment of any of the angiograms, the final judgment was reached by consensus. Interobserver concordance of the review of the 3 kinds of angiograms between the 2 readers was evaluated by the kappa test.
Results
The CE-MRA and 3D-TOF MR angiographic images were not degraded by patient motion or technical failure in any of the patients and were rated as being of good diagnostic quality. A temporal resolution of 0.8 s/frame was obtained with our CE-MRA technique, which was twice that obtained with the CE-MRA technique employed before the introduction of the parallel imaging technique.
At the initial assessment, the 2 readers’ ratings agreed in 16 (94%) patients with a kappa value of 0.77 (substantial reproducibility), in 14 (82%) patients with a kappa value of 0.58 (moderate reproducibility), and in 6 (100%) patients with a kappa value of 1.00 (perfect reproducibility) in the assessment of CE-MRA images, 3D-TOF MR angiograms, and conventional DSA images, respectively.
Of the 16 patients who underwent STA- or ECA-MCA bypass surgery, the patency of the anastomosis was visualized on the CE-MRA images in all the patients, except in one whose surgery had been performed at another institution. His CE MRA demonstrated failure of surgery, which was first suspected on 3D-TOF MR angiograms. In all of the remaining 15 patients who had successful STA- or ECA-MCA bypass surgery, CE MRA demonstrated flow from the parent artery to M2 or more distal branches. In the ratings of these 15 patients, CE MRA was rated better than 3D-TOF MRA in 12 patients, whereas it was rated as being equally efficient in 3 patients. In the former group of 12 patients, CE MRA allowed visualization of the distal M2 branches and their flow dynamics could be compared with that on the contralateral side (Fig 1). In the patient who had VA-PCA bypass surgery, CE MRA provided better visualization of the flow to the bilateral PCAs and even to the distal basilar artery than 3D-TOF MR angiograms. Overall, CE MRA allowed better visualization of the postoperative bypass than 3D-TOF MRA in 13 patients (76%), whereas the 2 methods were equally efficient in 4 patients (24%). The rating of CE MRA corresponded to that of conventional angiography in all of the 6 patients who underwent both the imagings (Fig 2).
A 57-year-old man 7 months after STA-MCA anastomosis for stenosis of the right MCA.
A, 3D-TOF MR angiogram shows patent anastomosis between the 2 STA branches and distal branches of the right MCA (M2).
B–D, Selected frames of CE MRA show corresponding findings. Flow to the distal parts of the M2 branches is better demonstrated in these images than by MRA.
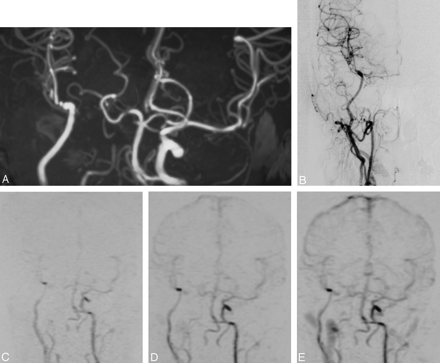
A 43-year-old woman 3 weeks after EC-MCA anastomosis by using a radial-artery graft for occlusion of the right ICA for treatment of a large ICA aneurysm.
A, 3D-TOF MR angiogram shows a patent radial artery graft and distal MCA branches. Note that the more distal MCA branches cannot be fully visualized due to the limited section thickness.
B, Conventional DSA images confirm the patent anastomosis and good distal flow.
C–E, Selected frames of CE MRA show corresponding findings.
Discussion
Postoperative EC-IC bypass is required not only to be patent, but also to provide sufficient blood flow to hypoperfused areas. As stated above, conventional angiography, MRA and CT angiography have been employed in the postoperative assessment of bypass grafts and the distal blood supply. Ultrasonography has also been used for the assessment, both intraoperatively and postoperatively (20, 21). Until now, single-photon emission CT or positron-emission tomography and perfusion study by MR imaging or CT have been considered as the most suitable techniques for the assessment of cerebral blood flow through a bypass graft. It has been believed that MR imaging with diffusion-weighted scanning is of greater value than CT to diagnose early postoperative lesions, including new infarction related to technical failure or homodynamic changes and multifocal hemorrhage with cerebral edema due to normal perfusion pressure breakthrough.
Our results indicated that CE MRA may be at least as useful as 3D-TOF MRA, if not of greater value, for the anatomic evaluation of EC-IC bypasses. Despite the limited number of cases in this study, the findings on CE-MRA images corresponded to those on conventional DSA images in terms of visualization of the branches distal to the anastomosis. These features of CE MRA are considered to be especially advantageous, because the imaging can be performed in combination with MR imaging, including diffusion-weighted imaging, with only the minimal invasiveness associated with the injection of gadolinium.
We consider that our results owe much to technical aspects of our CE-MRA scanning technique. CE MRA can be performed with either 2D or 3D data acquisition. In previous reports, 2D sequences have been employed, with excellent temporal resolution, whereas 3D techniques, which are widely used in time-resolved MRA of other regions of the body, were scarcely employed for the brain, mainly because of the rapid intracranial circulation time. Despite 3D data acquisition, however, we could still achieve a temporal resolution of as good as 0.8 s/frame, by using parallel imaging in combination with the segmented k-space sampling technique. In-plane spatial resolutions for the 2 MR techniques were 2.0 × 1.1 mm for time-resolved, CE MRA and 1.0 × 0.8 mm for 3D-TOF MRA. Although these in-plane sizes of pixels and the partition thickness (5 mm) were not so small, we could still clearly visualize the anastomoses, which are only a few millimeters in diameter, as well as their fine distal branches by our CE MRA. We believe that the maximum-intensity-projection, which effectively increases the signals from vessels containing gadolinium in the postprocessing of the 3D data set in CE MRA, was effective for good visualization of the small arterial branches despite lower spatial resolution when compared with 3D-TOF MRA. In addition, we consider that images of multiple phases of CE MRA allowed clear visualization even when the blood flow through a bypass route was rather slow.
The size of the field of view that we selected was the smallest allowed by our hardware and sequence. This resulted in a rather large voxel size, as stated above. The field of view, however, enabled us to visualize long bypasses, such as an EC-MCA anastomosis by using a radial-artery graft, for their entire length (Fig 2). This is another advantage of CE MRA over 3D-TOF MRA, which is generally limited by the section thickness. In addition, with our protocol of injecting 7 mL of gadolinium for a single scan, it is possible to perform CE MRA in an additional plane if indicated. Actually, in 2 patients in this series, we additionally obtained sagittal CE-MRA images, the image quality of which was the same as that of the initial coronal scans. This is probably because, due to subtraction, gadolinium injected for the initial scan does not degrade images of the second scan.
Because our technique provides serial images with good temporal resolution, their pixel-by-pixel analysis, similar to that in perfusion imaging performed by using the dynamic susceptibility contrast technique, may provide quantitative hemodynamic information from the scanned area. This is currently being investigated at our institution.
Conclusion
For postoperative evaluation of EC-IC bypasses, CE MRA can be performed with minimal invasiveness and a short examination time in addition to MR imaging, which can sensitively assess changes in the brain parenchyma. CE MRA by using the parallel imaging technique as well as the segmented k-space sampling technique is effectively used to assess patency of EC-IC bypasses.
Footnotes
Presented as “Postoperative Assessment of External Carotid-Internal Carotid Bypass by MR Digital Subtraction Angiography Using Parallel Imaging”at the 42nd annual meeting of the American Society of Neuroradiology, June 7–11, 2004, Seattle, WA.
References
- Received January 13, 2005.
- Accepted after revision March 22, 2005.
- Copyright © American Society of Neuroradiology