Abstract
SUMMARY: We describe MR spectroscopy in 2 patients with frontal sinus mucoceles that showed a dominant metabolite peak at 2.0-ppm chemical shift, simulating N-acetylaspartate (NAA) of normal neuronal tissue. In vitro analysis of postsurgical mucocele samples confirmed that the signal at 2.0 ppm was arising from the methyl moiety of an N-acetyl compound. This is probably caused by N-acetylgalactosamine or N-acetylglucosamine, which are glycoproteins found in normal respiratory mucus produced by the paranasal sinus epithelium.
In vivo MR spectroscopy has been applied to the study of a variety of intracranial diseases and may be useful in improving diagnosis of focal lesions such as cerebral neoplasm and infection.1–4 At long echo times (TEs), the metabolic constituents of normal brain include creatine (Cr), choline (Cho) and N-acetylaspartate (NAA). NAA is the dominant metabolite in normal brain tissue, detected at 2.0-ppm chemical shift (Fig 1) and is a putative neuronal marker present in healthy brain tissue.5 We describe 2 patients with frontal sinus mucoceles in whom dominant metabolite peaks at 2.0 ppm were detected, mimicking normal NAA.
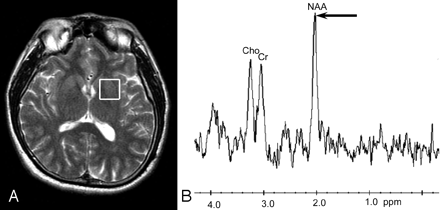
In vivo MR spectroscopy of normal brain. Axial T2-weighted MR image shows a single voxel of interest for MR spectroscopy (white box) placed within the brain parenchyma in a healthy volunteer (A). The corresponding in vivo MR spectrum shows the normal dominant peaks of NAA at 2.0 ppm (arrow), Cr at 3.0 ppm, and Cho at 3.2 ppm (B).
Materials and Methods
Two women, aged 33 and 74 years, were studied with MR imaging and in vivo MR spectroscopy on a 1.5T clinical scanner. In vivo MR spectroscopy was performed by using single-voxel MR spectroscopy point-resolved spectroscopy localization: the scanning parameters were 1500/136 ms (TR/TE); voxel dimensions, 2 × 2 × 2 cm; and FOV, 24 cm. Semiautomated spectral postprocessing was performed by using Functool (PROBE-P, Version 8.3, GE Healthcare, Milwaukee, Wis). The voxel of interest was placed completely within the sinus mucocele; normal brain tissue was excluded.
Both patients underwent surgical drainage of the mucoceles within 7 days of MR imaging; surgical procedure and recovery were uneventful. The surgical specimens were collected and immediately frozen in liquid nitrogen and stored in a freezer (at −78°C). The thawed samples were dissolved in heavy water (D2O), and the clear solutions were subsequently transferred into 5-mm nuclear magnetic resonance (NMR) tubes for 1D proton and 2D correlated spectroscopy (COSY) in vitro NMR spectroscopic analysis (AVANCE 500-MHz spectrometer equipped with pulse-field gradient units and a cryoprobe, Bruker Biospin, Rheinstetten, Germany). 1D proton NMR spectroscopic data acquired were processed and analyzed by XWIN-NMR, Version 3.1 (Bruker Biospin). 2D data acquired were processed by NMRPipe6 and analyzed by NMRView.7 2D COSY spectra were used for identification of brain metabolites. In t2, 1024 complex points were collected, whereas there were 512 points in t1, both covering a spectral width of 4595 Hz. The spectra were also zero-filled to a 1024 × 1024 matrix size.
Case Reports
Case 1
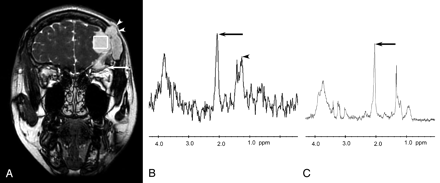
A 33-year-old woman presented with blurring vision in the left eye. Clinical examination revealed a left supraorbital mass, and MR imaging showed the mass to be arising from the frontal sinus with both intracranial and intraorbital extension. Its contents were hyperintense on both T1- and T2-weighted images, (Fig 2A) and there was no enhancement following intravenous contrast injection. In vivo MR spectroscopy showed a large peak at 2.0 ppm. The Cho and Cr metabolite peaks (3.2 and 3.0 ppm) expected to be in normal brain parenchyma were not positively identified (Fig 2B). At surgery, thick yellow necrotic tissue and inspissated squames were found. The dura was not breached. There was no bacterial growth. In vitro NMR spectroscopy of the mucocele contents revealed the main metabolite peak at 2.0 ppm, confirming the in vivo MR spectroscopy findings (Fig 2C).
33-year-old woman with frontal sinus mucocele. Coronal T2-weighted localizer image shows the voxel of interest for MR spectroscopy (white box) within a mucocele of high signal intensity (A). The mass is clearly extra-axial and compresses the frontal lobe brain parenchyma, which is excluded from the voxel. Note the involvement of the superior aspect of the right orbit (arrow) and destruction of the outer table of the skull (arrowheads). In vivo MR spectrum (B) shows a dominant peak at 2.0 ppm (arrow) and another at 3.9 ppm. There is also a complex multiplet between 1.2 and 1.4 ppm (arrowhead), probably from a combination of lipid and lactate. In vitro 1D proton NMR spectroscopy (C) of the mucocele contents removed at surgery confirms the dominant peak at 2.0 ppm (arrow) and other peaks at various positions of chemical shift.
Case 2
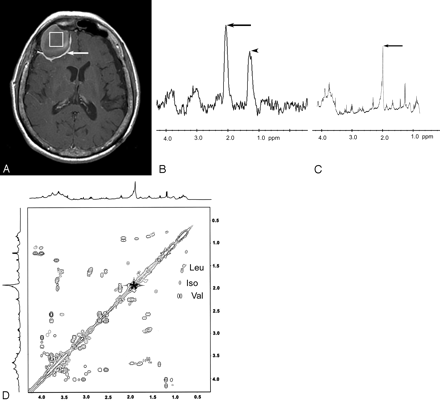
A 74-year-old woman presented with headaches and seizures for 2 weeks. MR imaging revealed a right frontal intracranial extra-axial mass, arising from the frontal sinus. The mass had a T2-weighted high-signal-intensity rim and low-signal-intensity center and was compressing the adjacent frontal lobe, causing inflammatory enhancement of the dura mater (Fig 3A). In vivo proton MR spectra show a large dominant peak at 2.0 ppm but absence of Cr or Cho peaks (Fig 3B). During frontoethmoidectomy, thick viscid yellow mucus, which showed gram-negative bacilli, was found. The dura mater and brain were not involved. In vitro 1D NMR spectroscopy of mucocele contents confirmed the dominant peak at 2.0 ppm (Fig 3C), and its 2D COSY revealed that this peak did not correlate with other protons via proton-proton couplings (Fig 3D). On the basis of the proton chemical shift and 2D correlations, we found that the signal intensity at 2.0 ppm was arising from the CH3 moiety of N-acetyl compounds, instead of acetate. Further analysis of 1H-13C heteronuclear single-quantum correlation (HSQC) (not shown) of the same sample confirmed our conclusion. Several minor metabolites such as leucine, isoleucine, and valine were also identified according to the COSY correlations.
74-year-old woman with frontal sinus mucocele. Axial postcontrast MR image (A) shows the voxel of interest placed completely within the right frontal mucocele. Dural contrast enhancement (arrow) confirms the extra-axial location of the mucocele. There is no visible contamination of the voxel by normal brain tissue or scalp fat. In vivo proton MR spectrum (B) shows dominant peaks at 2.0 ppm (arrow) and 1.2 ppm (arrowhead), the latter assigned as lipid breakdown products. In vitro 1D NMR spectrum (C) of the mucocele sample after surgery confirms the main metabolite peak at 2.0 ppm (arrow). The 2D COSY spectra (D) show an N-acetyl compound at 2.0 ppm (asterisk), as well as cross-peaks for leucine (Leu), isoleucine (Iso) and valine (Val).
Discussion
In 2 patients with frontal sinus mucoceles, we found a dominant peak at 2.0 ppm, which resembled the expected NAA spectral peak of normal brain parenchyma, even though there was no brain tissue included in the voxel of interest. NAA is a marker of normal neuronal function and, together with Cho and Cr, forms the main normal metabolites detectable by in vivo MR spectroscopy at long TEs.5 A mucocele, on the other hand, represents an end stage complication of a chronically obstructed paranasal sinus.8,9 It occurs most commonly in the frontal sinus (66% of mucoceles) and results from accumulation of mucoid material produced by paranasal sinus epithelial cells. Mucoceles are caused by a chronically obstructed sinus ostium with enlargement of the bony walls.8 Rarely, mucocele can extend intracranially, but because they are extra-axial lesions, mucoceles seldom involve the brain parenchyma.8 Therefore, in vivo MR spectroscopy studies of mucoceles should not show evidence of any of the metabolic constituents of normal brain parenchyma.
Initially, we suspected that our observations were due to inaccurate voxel prescription or contamination by normal brain tissue NAA included in the voxel of interest as a result of the partial volume–averaging effect. However, in both patients, the voxel of interest was placed in the center of the mucocele cavity, away from surrounding normal brain, and contamination was unlikely. Furthermore, the metabolite at 2.0 ppm was also present on in vitro NMR spectroscopy of the mucocele specimen obtained at surgery. During frontoethmoidectomy, the dura mater was not breached, the liquid specimens obtained from both patients did not contain any normal brain, and therefore, the metabolite could not have originated from brain tissue.
In vitro spectroscopic examination of the mucocele contents with 1D and 2D techniques confirmed that the metabolite at 2.0 ppm was indeed due to the presence of N-acetyl compounds. Although in vivo MR spectroscopy has been used in clinical practice to help differentiate brain diseases such as neoplasms and abscess,1–4 in vitro NMR spectroscopy of biologic samples, typically obtained after surgery or biopsy, is also useful.10 In vitro 1D NMR spectroscopy at very high magnetic field strength (typically at 400 MHz or equivalent to 9.4T) has been used to study neoplasm with very high spectral resolution. However, even though 1D NMR spectroscopy has a simple pulse sequence and is relatively easy to perform, the complexity of biologic samples, which do not contain pure chemical compounds but rather a mixture of substances, makes metabolite peak assignment difficult. For instance, the multiple overlapping metabolites in the region of 1.3 and 0.9 ppm, including lactate, lipids, and proteins, are impossible to accurately analyze by using conventional 1D spectra. To overcome this limitation of uncertainty and overlapping peaks, one can use the second dimension in 2D NMR spectroscopy11 to provide information about the cross-peaks and help to identify the metabolic compounds.
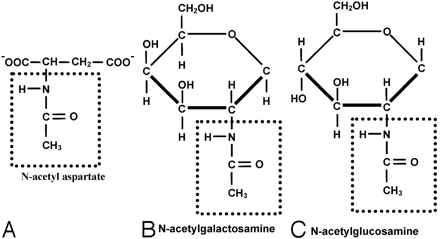
MR spectroscopy depends on the principle of specific chemical bonds resonating under a magnetic flux, giving rise to identifiable and characteristic peaks, measured in parts-per-million chemical shift.5 Thus, spectroscopy does not identify specific compounds but rather detects certain chemical species such as CH3 or ethyl groups by their chemical bonds. Therefore, a variety of different compounds with a common chemical bond may be detected, occupying the same parts-per-million chemical shift. In the normal brain, the NAA peak is due to the CH3 bonds of the N-acetyl group (box in Fig 4A) resonating at 2.0-ppm chemical shift. This resonance is thus not specific to NAA, and any other N-acetylated compound with the same methyl group chemical bonds may also be responsible for our findings on in vivo and in vitro spectroscopic studies.
Chemical structure of NAA and N-acetylated glycoproteins. The common CH3 moiety of N-acetyl compounds (box) in NAA (A) and N-acetyl glycoproteins (B and C) is highlighted. MR spectroscopy will detect the electrons in the CH3 chemical bond, and the metabolite peak at 2.0 ppm will be positive for NAA as well as N-acetyl glycoproteins.
Although we have confirmed that the metabolite detected at 2.0 ppm is not a constituent of normal brain NAA and is consistent with the presence of an N-acetyl compound, the nature of this compound in mucoceles still needs to be established. There has been a great deal of clinical interest in the cellular and chemical pathology of human mucus, especially in diseases of mucus hypersecretion such as asthma, chronic bronchitis, and cystic fibrosis, and the structure and function of the respiratory mucus has been extensively studied.12 Normal mucus, composed of 96% water and 4% mucus glycoproteins (also called mucins), exhibits high viscosity and often forms a gel, helping to lubricate and forming a protective physical barrier on epithelial surfaces.12 In the paranasal sinus, this mucus biofilm is secreted by glands and epithelial goblet cells, similar to other respiratory mucous membranes. Mucins from respiratory tract secretions, cervical, and gastrointestinal mucus have similar physical and chemical properties and structural design, with several different families of very large complex molecules of exceptional mass, size, and complexity.13
The chemical structure of mucus that makes up the mucocele contents has also been well studied. Mucus glycoproteins have complex carbohydrate chains covalently attached to their polypeptide backbones.13 One of the characteristic features of mucins is their high (generally >50%) content of carbohydrates, primarily in the form of oxygen linkages to oligosaccharides (O-glycosidic linkages). There are only 8 sugars (monosaccharides) that make up these oligosaccharides, including N-acetylgalactosamine (GalNAc), N-acetylglucosamine (GlcNAc), N-acetylneuraminic acid (NeuAc), galactose, glucose, mannose, xylose, and fucose.12 Therefore, the most likely metabolite in the mucocele specimen to be responsible for the N-acetyl CH3 resonance at 2.0 ppm would be the N-acetylated sugars GalNAc, GlcNAc, and NeuAc found in the mucin glycoproteins (Fig 4B, -C). In our specimens, we were unable to purify a single chemical metabolite from the mixture of viscid mucus; the infected mucus that constitutes the mucocele contents is probably a mixture of different compounds.
In addition to the large N-acetyl peak, traces of low-molecular-weight amino acids such as leucine, isoleucine, valine, alanine, and lactate were also identified on 2D COSY in 1 patient. However, our in vivo MR spectroscopy was not sensitive enough to confirm the presence of these amino acids. These findings would be consistent with metabolites that have been detected in cerebral abscess by other investigators by using in vivo MR spectroscopic methods4; gram-negative bacilli were detected in 1 of our patients. The difference in MR spectral patterns between sinus mucocele and cerebral abscess, in which similar amino acids, alanine, acetate, and succinate have been reported, still remains to be studied.4 We believe this to be the first description of in vivo MR spectroscopy in intracranial mucocele and in identification of N-acetyl mucus compounds, confirmed by in vitro studies. In our spectra, several unknown peaks were detected including a broad peak centered at 3.8 ppm, possibly containing superimposed smaller peaks (Fig 2). We could not identify these metabolites, and further study of mucocele glycoproteins may benefit from purification and structural analysis by using high-resolution HSQC and heteronuclear multiple bond correlation (eg, with carbon in the second dimension) or mass spectrometry.
Clinical in vivo MR spectroscopy is being used to study a variety of intracranial disease. In our cases, the large dominant peak at 2.0 ppm caused by mucus in intracranial mucoceles may be mistaken for contamination by normal brain and may represent a potential pitfall in the interpretation of in vivo MR spectroscopy. In vitro study especially with 2D COSY was useful in resolving the different chemical species and suggests that the methyl moiety in the N-acetyl group of mucus glycoproteins was the probable cause of the peaks identified.
Footnotes
This work was partly supported by research grants from Biomedical Research Council (BMRC) 01/1/33/19/199 and Singhealth/NHG-PTD-03004.
References
- Received November 3, 2005.
- Accepted after revision November 22, 2005.
- Copyright © American Society of Neuroradiology