Abstract
SUMMARY: We report a case of large temporal giant cell reparative granuloma in a 72-year-old man. MR imaging depicted a right temporal expansile multiloculated lesion, with hyper- and hypointense signal areas on T2-weighted images, heterogeneously enhancing after gadolinium administration. Cortical thinning and bone remodeling of the temporal squamous portion were better seen on CT. The patient underwent surgery, and the diagnosis was achieved by the correlation of imaging, histologic, and laboratory findings.
The term “giant cell reparative granuloma” (GCRG) was introduced by Jaffe in 1953 as a benign lesion originating from an inflammatory response due to intraosseous hemorrhage following trauma.1,2 It is a non-neoplastic fibro-osseous lesion,1,3 mostly found in the mandible and maxilla.4–6 Temporal bone location is less frequent and was first described in 1974 by Hirschl and Katz,7 with only 21 cases reported since then in the literature, according to a Medline search by using the key words “GCRG” and “temporal.” We present a case of temporal GCRG and also review the imaging and histologic correlation, according to other reports.
Case Report
A 72-year-old man presented with a 1-year right progressive hypoacusia, dizziness, vertigo, and right temporal pain. He had previous episodes of repeated middle ear infections, mainly in the right side. There was no head trauma history.
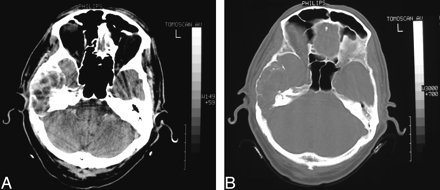
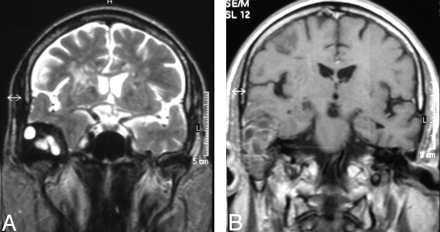
Head CT showed a large heterogeneous right temporal bone lesion (3.2 × 3.8 × 4.9 cm in greatest diameters), with cortical thinning and bone remodeling of the temporal squamous portion, along with lytic involvement of tympanic cavity, mastoid cells, and posterior wall of glenoid fossa; the lesion had a major intracranial component (Fig 1A, -B). Brain MR imaging disclosed a well-circumscribed round multiloculated right temporal extra-axial lesion. On T2-weighted sequences, it was mostly hyperintense with hypointense septations; after gadolinium administration, there was heterogeneous enhancement of the lesion and adjacent temporal dura. The lesion expanded mostly to the temporal fossa, with a slight mass effect on the temporal parenchyma but no edema signs, and, to a much lesser extent, to the glenoid fossa, with no evidence of condylar destruction (Fig 2A, -B).
A, Axial non-enhanced CT scan (soft-tissue algorithm) shows a heterogeneous right temporal bone lesion, with cortical thinning and bone remodeling of the temporal squamous portion and lytic destruction of middle ear (B, axial CT scan, bone algorithm).
A, Coronal T2-weighted image shows a right temporal extra-axial lesion, mostly hyperintense but with surrounding and septal areas of low signal intensity. Note a mild mass effect on the temporal parenchyma. There is no evidence of condyle destruction of the mandible. B, Contrast-enhanced coronal T1-weighted image shows the lesion occupying the right middle ear, which enhances heterogeneously after gadolinium. There is enhancement in the adjacent temporal dura.
These findings were consistent with a slow-growing expansive osseous lesion, with a myriad of possible diagnoses, including: benign epithelial and nonepithelial lesions, infectious and inflammatory lesions, or histiocytosis.8 Cholesterol granuloma was ruled out because of an absence of hyperintense areas on T1-weighted images.8 Sarcoma or metastasis was less likely because of the lack of rapid-growth imaging signs.
In August 2004, the patient was referred to the neurosurgery department for right temporal craniotomy and lesion excision (total removal, macroscopically) along with radical mastoidectomy. At surgery, the lesion was grayish, infiltrative, partially cystic, and calcified. There was dural (but no cortex) invasion, being replaced by Neuropatch (B. Braun Melsungen, Melsungen, Germany). After surgery, the patient had ipsilateral facial nerve palsy.
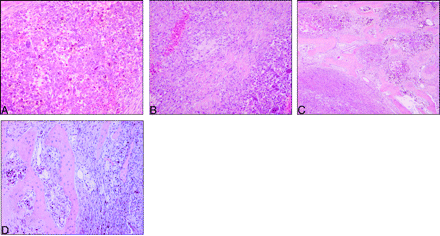
The entire surgical specimen was submitted for pathologic evaluation. Microscopically, the lesion was heterogeneous (Fig 3A–D) and composed of multinucleated osteoclast-like giant cells and spindle-shaped fibroblastic and polygonal xanthomatous cells in variable numbers. The mitotic index was low, necrosis was absent, and the lesion contained extensive areas of old hemorrhage and focal areas of neoformed osseous trabeculae at the advancing edge of the lesion. Tumor cells expressed vimentin and CD68 without immunoreactivity for CAM 5.2, AE1/3, CD31, S-100 protein, and HMB 45.
GCRG light microscopy discloses heterogeneous areas with multinucleated cells (A), alternating with spindle and xanthomatous cells (B). The lesion infiltrates the adjacent bone (C), both depicting similar heterogeneous features (D). Note signs of recent (B) and old hemorrhage (A, C, D).
Subsequent MR imaging revealed a small tumor remaining around the right condyle.
The patient had temporal pain relief after lesion removal and participated in an outpatient rehabilitation program, resulting in progressive recovery of facial motor deficits.
Discussion
The pathologic features of the lesion narrowed down the several diagnostic possibilities to the following: tenosynovial giant cell tumor of temporomandibular joint, brown tumor associated with hyperparathyroidism, GCRG, and giant cell tumor (GCT). Tenosynovial GCT of the temporomandibular joint was excluded by lesion topography on MR images: The lesion abutted the joint focally instead of originating from it. Brown tumor associated with hyperparathyroidism was excluded by laboratory studies disclosing normal serum calcium, phosphorous, and parathormone levels. Paget disease, frequently associated with GCT, was less likely because of the absence of a diffuse bone thickening appearance with mixed densities.8,9
The most challenging differential diagnosis encompasses GCT versus GCRG, due to their histologic resemblances.4 Nonetheless, the presence of giant cell osteoclast-like low-mitotic-rate signs of osteoid and new bone production and extensive hemorrhage with hemosiderin deposits favored GCRG.10
GCRG is a rare lesion,1,5,11 thought to be related to previous trauma or inflammation,12 but its pathogenesis remains unknown.1,13 GCRG mostly affects children, young adults, and women.4,11 However, according to GCRG cases reported in temporal locations, sex and age distributions generally differ from those in other locations and trauma is less frequent (Table).
| Author(s) | Age (y)/Sex | Trauma | Initial Symptoms |
|---|---|---|---|
| Hirschl and Katz, 19747 | 36/F | Yes | Hearing loss |
| Colclasure et al, 198122 | 10/M | NR | Hearing loss |
| 22/M | NR | Hearing loss and mass | |
| Tesluk et al, 198923 | 56/M | No | Mass, pain |
| Ciappetta et al, 199024 | 25/M | Yes | Dysphagia, pain, protrusion |
| Cohen and Granada-Ricart, 199325 | 4m/F | No | Mass |
| Lewis et al, 199419 | 32/F | Yes | Hearing loss |
| Nemoto et al, 199515 | 36/M | No | Hearing loss, tinnitus |
| 28/M | No | Hearing loss, tinnitus | |
| Maruno et al, 199714 | 3/F | No | Facial weakness, hearing loss |
| Ung et al, 199826 | 36/F | No | Hearing loss |
| Liu et al, 200110 | 44/M | No | Hearing loss, tinnitus |
| 72/M | No | Hearing loss | |
| Khodaei et al, 200117 | 36/M | NR | Pain, tinnitus, vertigo |
| Sharma et al, 200218 | 12/M | Yes | Facial weakness, hearing loss, mass, tinnitus, vertigo |
| Matsui et al, 200227 | 41/M | NR | Mass |
| Yoshimura et al, 200216 | 38/M | NR | NR |
| Boedeker et al, 20031 | 17/F | No | Hearing loss |
| Mohammed et al, 20016 | 38/M | NR | Hearing loss, bilateral headache |
| Montero et al, 20034 | 60/M | NR | Hearing loss |
| Kim et al, 20035 | 50/M | No | Hearing loss, tinnitus |
Note:—NR indicates not reported.
Temporal GCRG case reports
In the Table, 10 of the 21 cases (47.5%) were not related to trauma. Only 4 patients had positive trauma history. Ages ranged from 4 months to 72 years, with a mean of 32 years. There was a predominance of 15 men versus 6 women. The most frequent associated symptom was hearing loss.
In the available MR imaging findings of the reported cases mentioned in the Table,1,5,6,10,14–19 the lesions always had a hypointense component in long repetition time (TR) sequences and heterogeneous enhancement, although a multiloculated appearance similar to that in our case report was only described by Mohammed et al,6 Nemoto et al,15 and Sharma et al.18 The hypointense areas on long TR sequences probably reflected hemosiderin and bone formation.
The first temporal GRCG case was described by Hirschl and Katz7; they reviewed 23 cases initially diagnosed as GCT and concluded that 18 were GCRG. GCRG is considered a benign lesion6 that presents as a mass with cortical thinning20 and potential local aggressiveness.1 When localized in the temporal region, associated symptoms include hearing loss, tinnitus, pain, vertigo, and facial palsy.1
Some authors regard GCRG as the last phase of aneurysmal bone cyst and consider theses lesions as solid aneurysmal bone cyst variants,21 because of the overlap of clinical and histologic features shared by GCRG and aneurysmal bone cyst.21 Despite these histologic similarities, GCRG has no large blood-filled spaces, a finding that is at variance with aneurysmal bone cyst.21
After complete removal of GCRG, prognosis is good, with a 10%–15% rate of recurrence.1 If the removal is partial, the recurrence rate is higher.1 Radiation therapy could be an alternative treatment, but the risk of sarcomatous degeneration makes radiation therapy controversial in this setting.1,5
Conclusion
GCRG is rare, especially in the temporal location. The differential diagnosis with GCT is of the utmost importance to establish a suitable follow-up and treatment plan. There are no specific imaging features for the diagnosis of GCRG; thus, it relies on close correlation with pathology and clinical and laboratory data. Additionally, we emphasize the absence of trauma history in our patient, in accordance to recent reports of GCRG.
References
- Received July 19, 2005.
- Accepted after revision September 30, 2005.
- Copyright © American Society of Neuroradiology