Abstract
BACKGROUND AND PURPOSE: The aim of this retrospective study was to report the incidence, clinical presentation, and midterm clinical and imaging results of endovascular parent vessel occlusion of 11 patients with 13 distal cerebellar artery aneurysms.
MATERIALS AND METHODS: Between January 1995 and December 2006, 2201 aneurysms were treated in our institution. Thirteen aneurysms in 11 patients were located on distal cerebellar arteries (incidence, 0.6%), 8 of them arising from vessels feeding small arteriovenous malformations. There were 6 men and 5 women, ranging from 44 to 70 years of age. One patient with a superior cerebellar artery aneurysm presented with isolated trochlear nerve palsy. Ten patients presented with subarachnoid and intraventricular hemorrhage, and most patients were in poor clinical condition on admission. Aneurysm location was the superior cerebellar artery in 3, the anterior inferior cerebellar artery in 5, and the posterior inferior cerebellar artery in 5. Two patients had 2 aneurysms each.
RESULTS: Eleven aneurysms were treated by simultaneous coil occlusion of the aneurysm and parent artery or occlusion of the parent artery just proximal to the aneurysm. Clinical follow-up was at a mean of 16.5 months (range, 2–40 months). Infarction in the territory of the occluded vessel was apparent on follow-up imaging in 5 of 11 patients, all without functional impairment.
CONCLUSION: Distal cerebellar artery aneurysms are rare. Most patients present with poor-grade hemorrhage. Endovascular parent vessel occlusion is effective in excluding the aneurysm from the circulation. In most patients, adequate collateral circulation prevents infarction in the territory of the occluded vessel. In this series, when infarction did occur, the clinical consequences were limited.
Aneurysms located on distal portions of the cerebellar arteries are uncommon, and their underlying pathology, presentation, natural history, and clinical management are poorly understood.1,2 Only case reports or small case series have been published, and no precise information is available in the literature regarding the management of distal cerebellar artery aneurysms or the associated long-term results.1–16 Several approaches have been proposed to surgically treat these aneurysms, depending on their size and location. Although direct clipping is possible in some cases, sophisticated surgical techniques such as hypothermic cardiac arrest and bypass construction are often needed.9–12,14–16 On the other hand, endovascular therapy, consisting of occlusion of the parent artery with coils or glue, is simple to perform and often effective.2–7,13 However, this deconstructive approach may lead to infarctions in the territory of the occluded cerebellar artery if collateral circulation is insufficient.
The aim of this retrospective study was to report the incidence, clinical presentation, and midterm clinical and imaging results of endovascular parent vessel occlusion of 11 patients with 13 distal cerebellar artery aneurysms.
Patients and Methods
Between January 1995 and December 2006, 2201 aneurysms were treated in our institution. Surgery was performed in 967 aneurysms and endovascular treatment, in 1134 aneurysms. Of 2201 treated aneurysms, 13 aneurysms in 11 patients were located on distal cerebellar arteries, resulting in an incidence of 0.6% of treated intracranial aneurysms. All 13 aneurysms were treated by endovascular techniques. Characteristics of patients and aneurysms are summarized in the Table. There were 6 men and 5 women with a mean age of 61 years (range, 44–70 years). One patient with a distal partially thrombosed superior cerebellar artery (SCA) aneurysm presented with isolated trochlear nerve palsy. Ten patients presented with subarachnoid and intraventricular hemorrhage, in 2 patients with associated vermian hematoma. Three patients, all with a distal anterior inferior cerebellar artery (AICA) aneurysm, presented with, besides hemorrhage, ipsilateral facial palsy and sensory hearing loss. Eight of 10 patients who presented with hemorrhage were in poor clinical condition, with hydrocephalus needing emergency external ventricular drainage. Two patients had a second episode of hemorrhage before treatment of the aneurysm. One patient with a distal posterior inferior cerebellar artery (PICA) aneurysm had undergone surgery at another institution, but the surgeon was unable to locate the aneurysm.
Clinical, imaging, and treatment characteristics of 11 patients with 13 distal cerebellar artery aneurysms
Aneurysm location was SCA in 3, AICA in 5, and PICA in 5. Two patients had 2 distal aneurysms each: both on 1 PICA in 1 patient and 1 each on the AICA and PICA in the other patient. In 6 of 11 patients, a small arteriovenous malformation (AVM), supplied by the affected vessel, was apparent but not considered the cause of hemorrhage. One patient with an AICA aneurysm had an additional middle cerebral artery aneurysm that was coiled in the same session.
Endovascular Treatment
Endovascular treatment was performed with the patient under general anesthesia on a biplane angiographic unit (Integris BN 3000 Neuro; Philips Medical Systems, Best, the Netherlands). On this unit, 3D angiography has been available since 1999. In earlier cases, multiple angiographic projections were made to evaluate the anatomy of the aneurysm and parent vessel. In later cases, 3D angiography was performed immediately after diagnosis of an aneurysm. The type of endovascular treatment depended on the anatomy of the aneurysm neck and parent vessel as well as on the location of the aneurysm, either on the mainstem or a distal branch. After treatment, subcutaneous heparin was administered in therapeutic dosage for 48 hours.
Results
Results are summarized in the Table. Eleven wide-necked or fusiform aneurysms were treated by simultaneous coil occlusion of the aneurysm and parent vessel or coil occlusion of the parent artery just proximal to the aneurysm: occlusion of the SCA rostral or caudal trunk in 3, AICA mainstem occlusion in 4, AICA rostral branch occlusion in 1, and PICA cortical segment occlusion (distal to the cranial loop) in 3 aneurysms. One PICA with 2 aneurysms distal to the cranial loop was occluded with glue proximal to the aneurysms.
Clinical and Imaging Follow-Up
All 11 patients survived the hospital admission period. Clinical follow-up was at a mean of 16.5 months (range, 2–40 months). None of the patients had an episode of rebleeding after treatment. In the 3 patients with a distal AICA aneurysm with facial and vestibule-cochlear nerve palsy accompanying hemorrhage, there was no recovery from these symptoms at follow-up. Infarctions in the territory of the occluded cerebellar artery (or the branch of the cerebellar artery) were apparent on follow-up MR imaging or CT in 5 of 11 patients; the remaining patients had no infarctions. One of 3 patients with a partial or complete SCA occlusion developed a small clinically silent infarction. Two of 5 patients with AICA occlusions developed small clinically silent infarctions in the brachium pontis. Two of 4 patients with a PICA occlusion de-veloped partial PICA infarctions; both patients were independent on follow-up and are able to walk without gait imbalance. Of 6 patients with a concomitant cerebellar AVM, 1 was treated with surgery and 4, with γ-knife radiosurgery. One patient is scheduled for radiosurgery.
In general, in patients who developed infarctions after mainstem or branch occlusion of a cerebellar artery, possible symptoms may have been masked by decreased level of consciousness after severe hemorrhage during a stay in the intensive care unit or by cranial nerve dysfunction from the onset. One patient was discharged to a nursing home as a result of diffuse infarctions after vasospasm and died 4 months later of pneumonia. In the other 8 patients who presented with hemorrhage, after revalidation, no cerebellar symptoms were apparent. One patient with an AICA aneurysm, who was independent on discharge, died 2 months later at home of an unknown cause. Autopsy revealed no rebleeding from the AICA aneurysm or infarctions in the supplied vascular territory. The 7 remaining patients eventually were independent. Functional impairment, if any, was mostly determined by cognitive decline and memory loss after severe subarachnoid and intraventricular hemorrhage and not by the therapy-induced infarctions. The trochlear nerve palsy as the presenting symptom of a SCA aneurysm in 1 patient was almost cured on follow-up 1 year later.
Representative Cases
Case 1.
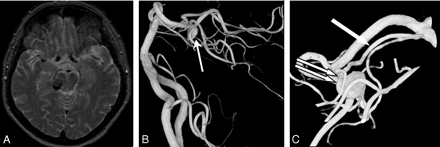
A 59-year-old woman (patient 1) presented with diplopia of subacute onset. On clinical examination, isolated right trochlear nerve palsy was apparent. MR imaging and angiography demonstrated a partially thrombosed aneurysm, distal on the rostral trunk of the right SCA. The aneurysm and the parent artery were occluded with coils. MR imaging 4 weeks later showed complete thrombosis of the aneurysm without infarctions. Twelve months later the trochlear nerve palsy was almost cured (Fig 1).
A 59-year-old woman with isolated right trochlear nerve palsy. A, MR image demonstrates a partially thrombosed aneurysm lateral to the brain stem. B, A 3D angiogram shows a distal SCA aneurysm (arrow). C, Detail of the 3D angiogram reveals an aneurysm without a neck (double arrow) on the rostral trunk of SCA (arrow).
Case 2.
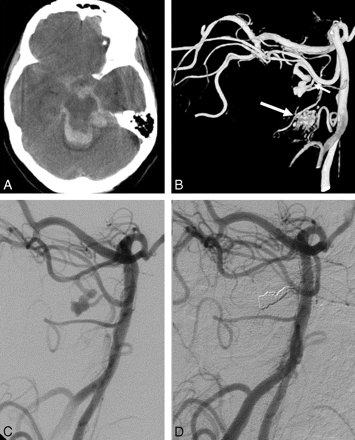
A 68-year-old woman (patient 2) was admitted in poor clinical condition with a subarachnoid and intraventricular hemorrhage with a vermian hematoma. After intubation, an external ventricular drain was placed. Angiography showed an aneurysm on the caudal trunk of the left SCA and a small AVM supplied by the same SCA and the ipsilateral AICA. The aneurysm was occluded with coils, including the parent caudal trunk; the rostral trunk of the SCA remained patent. MR imaging performed 3 weeks later showed, besides the hematoma, a hemorrhagic infarction in the territory of the occluded caudal SCA trunk. Clinically, she gradually recovered completely and is independent in daily activities (Fig 2).
A 68-year-old woman with hemorrhage from a distal SCA aneurysm. A, CT scan shows subarachnoid and intraventricular blood and vermian hematoma. B, A 3D angiogram demonstrates a fusiform aneurysm of the caudal trunk of the SCA (small arrow). Also a small AVM is apparent (large arrow). C and D, Angiograms before (C) and after (D) occlusion of the aneurysm and parent vessel with coils.
Case 3.
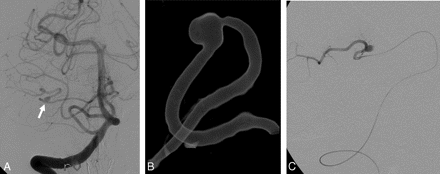
A 69-year-old woman (patient 7) was found comatose at home. A CT scan showed subarachnoid and intraventricular hemorrhage with a thick clot of blood in the right cerebellopontine angle and hydrocephalus. She was intubated, and an external ventricular drain was placed. Angiography revealed a small aneurysm without a neck on the distal right AICA. This aneurysm was occluded with coils together with the AICA itself. She made a remarkable recovery during the following weeks and was discharged home after 3 weeks in good clinical condition. Findings on follow-up brain MR imaging were normal; there were no infarctions in the AICA territory. One morning 2 months later, she was found dead in bed. Autopsy failed to disclose a cause of death. In particular, there was no rebleeding from the aneurysm, and there were no infarctions in the cerebellum or brain stem (Fig 3).
A 69-year-old woman with hemorrhage from a distal AICA aneurysm. A, A right vertebral angiogram shows a small distal AICA aneurysm (arrow). B, Detail of 3D right vertebral angiogram shows a distal AICA aneurysm without a neck. C, A microcatheter in the AICA just proximal to the aneurysm. The AICA was occluded with coils at this level.
Case 4.
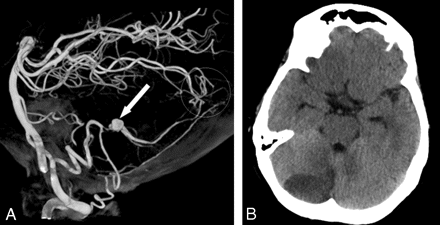
A 57-year-old woman (patient 9) was admitted after sudden coma. After intubation, a CT scan showed subarachnoid and intraventricular hemorrhage with a vermian hematoma and hydrocephalus. An external ventricular drain was placed. Angiography demonstrated an aneurysm on the medial branch of the distal left PICA, and this aneurysm was occluded with coils, including the medial PICA branch. CT 1 week later showed an infarction in the medial PICA territory. The patient slowly recovered during the following weeks and was transferred to a rehabilitation center after 2 months. One year later she was discharged home in good clinical condition and independent on daily activities. She is able to walk without assistance. The small AVM was treated with radiosurgery (Fig 4).
A 57-year-old woman with hemorrhage from a distal PICA aneurysm. A, A 3D angiogram demonstrates an aneurysm on the medial cortical branch of the right PICA (arrow), supplying a small torcular AVM, not well demonstrated in this threshold setting (circle). The aneurysm was occluded with coils, including the parent vessel. B, CT scan after 2 weeks reveals infarction in the medial territory of the right PICA.
Discussion
Anatomic and Clinical Considerations of Cerebellar Arteries
In the posterior fossa, a wide variation in arterial distribution exists. Specific areas of the brain stem and cerebellum cannot always be predictably allotted to a particular cerebellar artery. The effects of occlusion of a cerebellar artery range from clinical silence to death, as a result of infarction of portions of brain stem or cerebellum. Acute occlusion of any one of the cerebellar arteries is frequently associated with vomiting, dizziness, and inability to stand or walk. Recovery and survival of many patients after occlusion of a major cerebellar artery is attributed to adequacy of collateral circulation.17 In general, proximal occlusion of a cerebellar artery may result in a large ischemic area when collateral supply is insufficient, but with adequate collateral flow, no ischemia will occur at all. On the other hand, distal side branch occlusions have a higher probability of inducing ischemia, but the affected area will be limited.
SCA
The SCA is the most constant of the cerebellar arteries. It usually arises as a single trunk from the basilar artery and bifurcates into a rostral and caudal trunk. The SCA may also arise as a double trunk. It courses below the oculomotor nerve, encircles the brain stem near the pontomesencephalic junction, passing between the trochlear and trigeminal nerves. Its proximal portion courses medial to the free edge of the tentorium cerebelli, and its distal part passes below the tentorium. Hemispheric branches arise from the rostral and caudal trunks and give rise to the precerebellar arteries that supply the dentate and deep cerebellar nuclei as well as the inferior colliculi and the superior medullary velum. Anastomoses between the SCA and the AICA are frequent. Perforating direct and circumflex branches usually arise from the main trunk or proximal rostral and caudal trunk and supply the superior and middle cerebellar peduncles, the cerebral peduncle, and the collicular region. The cortical territory of the SCA is more constant than that of the AICA and PICA but is reciprocal with them. The SCA usually supplies most of the tentorial surface and frequently the adjacent upper part of the petrosal surface of the cerebellum. The rostral trunk supplies the vermian and paravermian area, and the caudal trunk supplies the hemisphere on the suboccipital surface.
Occlusion of the SCA produces a distinctive clinical picture that results from infarction of the cerebellum, dentate nucleus, brachium conjunctivum, and long sensory pathways in the tegmentum of the rostral pons. The onset is marked by vomiting, sudden dizziness, and the inability to stand or walk. Additional symptoms may occur, such as ipsilateral intention tremor, ipsilateral Horner syndrome, contralateral loss of pain and temperature, nystagmus, contralateral disturbance of hearing, and loss of emotional expression.17
AICA
The AICA originates from the midbasilar artery, courses through the central part of the cerebellopontine angle, and encircles the pons near the abducent, facial, and vestibulocochlear nerves. After sending branches to the nerves entering the acoustic meatus and to the choroid plexus protruding from the foramen of Luschka, it passes around the flocculus on the middle cerebellar peduncle to supply the lips of the cerebellopontine fissure and the petrosal surface of the cerebellum. It commonly bifurcates near the facial-vestibulocochlear nerve complex to form a rostral and a caudal trunk. The AICA gives rise to perforating arteries to the brain stem, middle cerebellar peduncle, the choroid plexus, and arteries supplying the inner ear and the vestibulocochlear and facial nerves.
Occlusion of the AICA results in syndromes related predominantly to the lateral portions of the brain stem and cerebellar peduncles, including palsies of the facial and vestibulocochlear nerves caused by involvement of the nerves and their nuclei. The most prominent symptom is vertigo, often associated with nausea and vomiting, followed by a facial paralysis, ipsilateral deafness, sensory loss, and cerebellar disorders. Patients can show nystagmus, ipsilateral loss of pain and temperature sensation on the face and corneal hypesthesia, Horner syndrome, cerebellar ataxia and asynergia, and an incomplete loss of pain and temperature sensation on the contralateral half of the body. All of the syndromes caused by its occlusion are not identical because of the variability of the AICA.17
PICA
The PICA has the most variable course and area of supply of the cerebellar arteries. It arises from the vertebral artery near the inferior olive and passes posteriorly around the medulla. At the anterolateral margin of the medulla, it passes between the rootlets of the hypoglossal, glossopharyngeal, vagus, and accessory nerves. Then it courses around the cerebellar tonsil, enters the cerebellomedullary fissure (caudal loop), and passes posterior to the lower half of the roof of the fourth ventricle. On exiting the cerebellomedullary fissure (cranial loop), its branches are distributed to the vermis and to the suboccipital cerebellar surface. Most PICAs bifurcate into a medial and a lateral trunk. The medial trunk supplies the vermis and adjacent part of the hemisphere, and the lateral trunk supplies the cortical surface of the tonsil and the hemisphere. The proximal PICA gives off perforating medullary branches in its course around the medulla. The perforating arteries have numerous branches and anastomoses that create a plexiform pattern on the medullary surface.
The consequences of a PICA occlusion vary and range from a clinically silent occlusion to infarction of portions of the brain stem or cerebellum, with swelling and tentorial herniation. The syndrome of occlusion of the PICA is referred to as the lateral medullary syndrome or Wallenberg syndrome and may include vomiting, dysphagia, dysarthria, hoarseness, ataxia, dizziness, vertigo, ipsilateral numbness of the face, loss of pain and temperature on the contralateral half of the body, nystagmus, and ipsilateral Horner syndrome. Occlusion of the branches of the PICA distal to the medullary branches produces a syndrome resembling labyrinthitis and includes rotatory dizziness, nausea, vomiting, inability to stand or walk unaided, and nystagmus without appendicular dysmetria.17
General Discussion
Distal aneurysms of cerebellar arteries are rare lesions accounting for 0.6% of all treated aneurysms in our practice. Similar low incidences have been reported previously.1,2 Most of our aneurysms presented with severe subarachnoid and intraventricular hemorrhage, and most patients were in poor clinical condition on admission, needing emergent intubation and external ventricular drainage. Additional clinical symptoms on presentation may be explained by the intimate relation of cerebellar arteries with the cranial nerves: 3 of 5 distal AICA aneurysms presented with, besides hemorrhage, ipsilateral facial palsy and sensory hearing loss that persisted at follow-up. One partially thrombosed distal SCA aneurysm presented with isolated trochlear nerve palsy. Most aneurysms involved the circumference of the parent vessel without an apparent neck. In 6 of 11 patients, a concomitant small AVM was supplied by the involved vessel. This association with AVMs has also been established in other studies.9–11,14
Although surgical treatment in experienced hands is associated with good results, access to the distal cerebellar arteries may be challenging, needing destructive far-lateral transcochlear or translabyrinthine approaches.8,11,14–16 Because most distal cerebellar artery aneurysms involve the circumference of the small parent vessel, clipping with sparing of the parent artery is mostly impossible; this problem leaves trapping (with risk of ischemia) the only surgical option. In addition, surgery is often complicated by cranial nerve dysfunction, in view of the intimate relationship of the cerebellar arteries with cranial nerves III–XI.15,16 In selected cases, sophisticated surgical techniques such as hypothermic cardiac arrest and bypass surgery on distal cerebellar arteries16 are needed to treat distal cerebellar artery aneurysms. These techniques are not available in most neurosurgical centers. Because distal cerebellar artery aneurysms are rare, not many surgeons will gain experience in the treatment of these aneurysms. In addition, most patients are in poor clinical condition and are not good surgical candidates in the acute phase.
Although surgery for distal cerebellar artery aneurysms is often difficult, limited to good-grade patients and associated with substantial morbidity, endovascular parent vessel occlusion is technically easy and can also be performed in the acute phase of hemorrhage in poor-grade patients. A substantial portion of distal cerebellar artery aneurysms is located on a distal branch and not on the mainstem, thereby limiting the area of possible induced infarction. In cases in which collateral circulation is insufficient, the area of infarction usually remains relatively restricted. In our small series, in 5 of 11 patients, infarctions were obvious on imaging follow-up but were either asymptomatic from the onset (brachium pontis infarctions after AICA occlusion) or not interfering with functional status on follow-up (partial SCA and PICA infarctions). Possible acute symptoms may have been masked by decreased levels of consciousness or cranial nerve dysfunction from the onset in critically ill patients in intensive care.
Additional 3D angiography was valuable in evaluating the anatomy of the aneurysm and its relation with the parent vessel and in determining a working projection for catheterization and coil occlusion. Our endovascular approach was effective in excluding the aneurysms from the circulation: No rebleeding occurred during hospital admission or on follow-up at a mean of 16.5 months. Our follow-up protocol after parent vessel occlusion did not include follow-up angiography in all patients, because reopening of an occluded vessel harboring a distal aneurysm is very unlikely. Despite the fact that most patients were in poor clinical condition on admission, recovery to an independent functional state followed in all but one. In 3 of 5 patients with a distal AICA aneurysm, central nerve VII and VIII palsy accompanied acute hemorrhage and did not recover at follow-up. In the remaining 2 patients, AICA occlusion did not induce dysfunction of these nerves. Altogether, functional status on last follow-up was mainly determined by either cranial nerve dysfunction from the onset or cognition and memory disturbances as a result of subarachnoid and intraventricular hemorrhage and not by the presence or extent of therapy-induced infarctions.
Similar good outcomes after endovascular treatment of distal cerebellar arteries have been reported in previous studies.2–7
Conclusion
Distal cerebellar artery aneurysms are rare vascular disorders with an incidence in our practice of 0.6%. Most patients present with severe subarachnoid and intraventricular hemorrhage. Endovascular parent vessel occlusion is simple to perform and is effective in excluding the aneurysm from the circulation. In most patients, adequate collateral circulation prevents infarction in the territory of the occluded vessel. In this series, when infarction did occur, the clinical consequences were limited.
References
- Received December 13, 2006.
- Accepted after revision January 12, 2007.
- Copyright © American Society of Neuroradiology