Abstract
BACKGROUND AND PURPOSE: The association of cortical organization disorders with facial hemangiomas or vascular malformations has been described in only a few reports. The purpose of this study was to show the close association of these cutaneous anomalies with cortical dysplasias and intracranial vascular abnormalities.
MATERIALS AND METHODS: Five patients, all women, with cutaneous vascular abnormalities, 4 with hemangioma and 1 with vascular malformation, were studied with MR and MR angiography.
RESULTS: All 5 of the patients showed cortical dysplasia. The cutaneous lesions involved the left frontal region, ipsilateral to the cerebral hemisphere with cortical dysplasia, in all of the patients. Four patients had seizures that responded well to antiepileptic drugs. Hemispheric hypoplasia was associated with the cortical dysplasia in all 5 of the patients. Arterial abnormalities were found in all of the patients, consisting of aplasia of the ipsilateral internal carotid artery in 2, persistence of the trigeminal artery in 2, persistence of both proatlantal arteries and double kinking in the internal carotid artery in 1, and origin of both anterior cerebral arteries from the same internal carotid in all 5 of the patients, 1 of whom also showed an intracavernous anterior cerebral artery origin of the same side of the hemispheric hypoplasia and polymicrogyria. Seizures and mild psychomotor delay could be caused by the cortical dysplasia and the hemispheric hypoplasia.
CONCLUSIONS: The presence of many congenital vascular abnormalities in this series suggests that facial hemangioma and vascular malformations may be in close relationship with cortical and vascular abnormalities. The reason that the vascular and cortical abnormalities occurred in the left side in all 5 of the patients and the mechanism underlying the association of both malformations are unclear. A genetic origin is suggested.
Cutaneous hemangioma and vascular malformation complex, the most frequent neurocutaneous syndrome, was first described in 1978.1 This syndrome associates the following2,3: 1) cutaneous hemangioma or vascular malformation, most commonly located in the face; 2) absence of carotid arteries, vertebral arteries, or both, most frequently ipsilateral to the facial vascular abnormality; 3) persistence of embryonic arteries, most commonly the trigeminal; 4) cerebellar ipsilateral hypoplasia with or without Dandy-Walker syndrome; and 5) aortic arch coarctation or cardiac malformation. The ipsilateral eye may be affected if palpebral hemangioma occludes the eye for a long time.4 Cutaneous features are well known from the clinical3,5 and histologic6 points of view. The magnitude of this syndrome, which may involve any region of the body, has seldom received comment.2,5,7,8 This syndrome is known as “cutaneous hemangiomas: vascular anomaly complex”2,7; posterior fossa malformations, hemangiomas, arterial anomalies, coarctation of the aorta and other cardiac defects, and eye abnormalities3; and Pascual-Castroviejo type II syndrome.9 The association of facial hemangioma with disorders of neuronal migration or cortical organization was first described in 199510 and very seldom reported since.11,12 The purpose of this study was to correlate the facial vascular abnormalities with cortical and subcortical brain disorders affecting the ipsilateral hemisphere and with cerebral vascular malformations.
Materials and Methods
Among 48 patients with cutaneous facial hemangioma or vascular malformation, only 15 had complete imaging studies that included MR imaging; MR angiography (MRA) with visualization of the carotid and vertebral systems in the sagittal, coronal, and axial views; and the origin of the arteries in the aortic arch and the subclavian arteries. Five (33%) of the 15 patients, all female, had cortical dysplasia associated with facial vascular abnormalities. Indications for the examinations in the care patients were the facial hemangioma, seizures that were present in 4 of the 5 girls, and moderate psychomotor delay. The series consisted of 5 girls whose age at the time they were studied under our care ranged from 6 months to 16 years. Two of them (patients 1 and 3) also had cerebral conventional arteriographic study that was performed during the first visit to our service several years before MR imaging and MRA studies. Four had hemangioma and 1 had vascular malformation, in agreement with the classification of Mulliken and Glowacki,6 that involved the territory of the first sensory branch of the trigeminal nerve. In 4 patients, the hemangioma also involved a part of the facial zone innervated by the second sensory branch of the trigeminal nerve. Their present ages range from 21 months to 21 years. Only 1 patient (patient 1) has been reported on before.10
Results
The table shows the observed clinical, vascular, and cerebral abnormalities of our 5 patients. Patient 1 had cortical dysplasia with cerebral hemispheric hypoplasia of the left side and absence of the ipsilateral internal carotid with vascularization of both cerebral hemispheres through the contralateral internal carotid (Fig 1). She also showed absence of the left carotid canal in the basal views of the plain skull films. Patient 2 showed left frontal polymicrogyria, persistence of the ipsilateral trigeminal and both proatlantal arteries, double kinking of the extracranial portion of the ipsilateral internal carotid, and origin of both anterior cerebral arteries from the internal carotid of the nonaffected side. Patient 3 showed left cerebral polymicrogyria, enlarged lateral ventricle and hemispheric hypoplasia, and ipsilateral internal hypoplasia of the carotid and its intracranial branches (Fig 2).
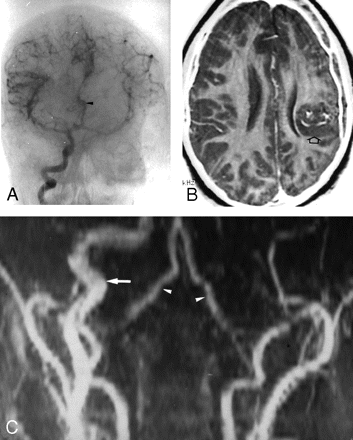
A, Patient 1. Right carotid arteriography. Both cerebral hemispheres are supplied by the right internal carotid artery. Enlarged anterior cerebral artery (arrowhead). B, Same patient. Axial T2-weighted (200/100) image shows an abnormal small left hemisphere with a round gray matter mass (arrow), which is criss-crossed by several large vessels. C, Coronal MRA of the neck shows absence of the left internal carotid artery and hyperplasia of the right internal carotid artery (arrow). Normal size of both vertebral arteries (arrowheads).
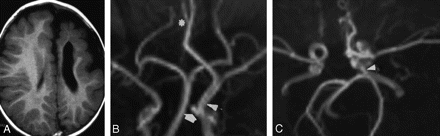
Patient 3 at 16 years of age. A, Axial T1-weighted MR image shows hypoplasia of the left cerebral hemisphere with enlarged lateral ventricle and polymicrogyria in the same hemisphere. B, MRA study of the same patient. The axial view shows right internal carotid artery hyperplasia with vascularization of both anterior cerebral hemispheres from a large right anterior cerebral artery (large arrowhead) and poor vascularization of the left cerebral hemisphere. Collateral vascularization from the ophthalmic (arrow) and the external carotid arteries to the middle cerebral artery in the left side (small arrowhead).
Observed clinical, vascular, and cerebral abnormalities of 5 patients
Patient 4 showed left cortical dysplasia and cerebral hemispheric hypoplasia, Dandy-Walker syndrome with left cerebellar hemispheric hypoplasia, and left carotid aplasia with vascularization of both cerebral hemispheres through the right internal carotid artery. Patient 5 showed left cerebral polymicrogyria-pachygyria, severe left cerebral hypoplasia, persistence of the trigeminal artery, and intracavernous origin of the left anterior cerebral artery (Fig 3). Origin of both anterior cerebral arteries from the same side with filling through the anterior communicating artery was seen in all of the patients and was located on the affected side in 1 (patient 5) and on the nonaffected side in 4 patients (patients 1–4). Location of the anterior communicating artery was very distal along the course of the anterior cerebral artery in all of the patients. Two cases (patients 2 and 5) showed persistent trigeminal artery, being ipsilateral to the cortical dysplasia and the vascular abnormalities in both patients.
A, Patient 5. Axial T1-weighted MR image shows hypoplasia of the left cerebral hemisphere with enlarged lateral ventricle and polymicrogyria of the anterior part of the hemisphere. B, Coronal view of MRA shows left carotid artery hyperplasia and intracavernous origin of the left anterior cerebral artery from which the right anterior cerebral artery (asterisk) also originates, and persistence of the trigeminal artery (arrow). The vertebrobasilar system is normal. C, Axial view of MRA shows the presence of the trigeminal artery in the left side (arrowhead).
Seizures, mostly of focal type, with onset of the electroencephalographic discharges in the affected hemisphere, were more frequent and resistant to antiepileptic drugs in patients 3 and 5, who had more severe hemispheric hypoplasia. Mental level was below borderline in 1 (patient 5), borderline in 3 (patients 1–3), and appeared normal in 1 patient (patient 4), though her young age (6 months) did not permit an objective evaluation.
Discussion
Cerebellar malformations, most commonly hemispheric hypoplasia with or without Dandy-Walker syndrome, were reported in 8 (50%) of 17 patients2 and 3 (100%) of 3 patients.13 In published reports on Dandy-Walker syndrome, the association with cutaneous vascular abnormalities is often observed.14,15 Cerebral hemispheric changes, particularly neuronal migration or cortical organization disorders, in association with cutaneous vascular malformations, have been seldom reported in association with cutaneous vascular malformations of the face.10–12 Cutaneous hemangiomas and vascular malformations are 3 times more frequent in women than in men,8 and, in our series, cortical and subcortical alterations were found only in females. The 2 patients reported by Aeby et al11 were also women. Only the patient reported by Grosso et al12 was a boy. All 8 of the patients described with neuronal migration or cortical organization disorders had cutaneous vascular abnormalities in the frontal region and palpebral area, though, in most patients, hemangiomas also involved other ipsilateral facial zones, especially the nose and the upper lip. In patient 2 of Aeby et al,11 the hemangioma also affected the skin of the neck, trunk, and upper and lower extremities of the ipsilateral or the contralateral side. Seven of the 8 patients had cutaneous hemangioma, and only our patient 3 had vascular malformation, in agreement with the classification given by Mulliken and Glowacki6 in 1982. Cortical dysplasia affected the ipsilateral hemisphere of the cutaneous hemangioma and vascular malformation in all of the patients. Only 1 patient of Aeby et al11 showed parasagittal polymicrogyric and similar changes in the contralateral hemisphere as well, though the changes were less severe than in the ipsilateral side. In 7 of the 8 patients, the cutaneous vascular anomaly affected the left frontal region, and only patient 1 of Aeby et al11showed the hemangioma on the right side. Although the mechanism that causes left facial and cerebral side involvement and the presentation of the disease almost exclusively in women have not yet been clarified, we suggest a possible association of genetic and environmental factors. In our series of more than 40 patients with this syndrome, the prevalence of females:males is 3:1 and facial left:right hemangioma or vascular malformation also is 3:1. Despite the high prevalence of the association of the cerebellar malformation with cutaneous facial hemangiomas and vascular malformations,2,8,13 only patient 4 showed ipsilateral cerebellar hypoplasia and Dandy-Walker malformation. Agenesis of the ipsilateral internal carotid artery has been reported in the first patient described with association of facial hemangioma and cortical dysplasia,10 which corresponded with patient 1 of our series.
The reported patients with cortical dysplasia10–12 and all of the patients included in the present series also showed hypoplasia of the cerebral hemisphere with the cortical-subcortical anomaly. Seizures of the patients with cortical dysplasia were most often controlled with antiepileptic drugs, whereas some of them did not require any medication.10–12 Contralateral hemiparesis and mild mental delay of most patients are caused not only by the cortical dysplasia but also by the cerebral hemispheric hypoplasia. Despite the severity of the cortical dysplasia and the cerebral hemispheric hypoplasia, hemispherectomy to control epilepsy was not necessary in the reported patients to date. Seizures associated with unilateral polymicrogyria in patients without facial hemangioma are controlled with antiepileptic medication in most patients.16
The mechanism that causes polymicrogyria has not yet been fully clarified. Polymicrogyria was believed to be an arrest of neuroblast migration17 but also a lesion acquired by an intrauterine vascular problem.18 Ischemia has been related to disorders of neuroblast migration.19 Alterations of neuronal migration have been found in several syndromes,20 most of them hereditary, starting in the first 20 weeks of gestation and without involvement of vascular structures. Cortical dysplasia associated with facial hemangioma is known from the recent years,10–12 probably because of the lack of neuroradiologic study of these patients until the description of the syndrome,1 though the existence of cutaneous vascular abnormalities, hemangiomas and vascular malformations, was well known, especially by dermatologists. Diverse injuries, such as ischemic and/or hypoxic events on the pial surface, can produce retraction of radial glial fibers. Neuroblasts that cannot complete migration end up as heterotopic neurons. The mechanism underlying the association between facial hemangioma or vascular malformation and brain dysplasia is unclear. Several theories, however, try to explain the possible etiologic mechanisms of migrational disorders in the corticosubcortical zone of the cerebral hemispheres. The radial pattern of distribution of watershed infarcts of the fetal and neonatal cortex that do not occur in older children is imposed by immature blood vessels, which penetrate the cortex from the meninges perpendicular to the brain surface.20 Migrating neuroblasts in this zone tend to cuff these vessels in the presence of ischemia, leaving columns of perivascular aggregates of neuroblasts.21,22 The high prevalence of arterial abnormalities in the patients of our series (absence of internal carotid, persistent embryonic arteries, especially trigeminal and proatlantal, the origin of both anterior cerebral arteries from the same internal carotid, and the dysplastic appearance of the intracranial arteries in some patients11) suggests a possible relation of vascular abnormalities with corticosubcortical disorders. There is a direct relation between the corticosubcortical malformations and the vascular territories supplied by the abnormal arteries in the 5 patients of our series. The presence of abnormalities that affected the anterior cerebral arteries (all patients) and the persistence of embryonic arteries, such as trigeminal (patients 2 and 5) and proatlantal (patient 2), as well as the Dandy-Walker malformation, occurs at approximately 4–5 weeks. Because vascular cerebral abnormalities and cerebellar malformation occur during the same embryonic dates, it is probable that the cortical dysplasia was contemporary to the vascular malformations in our patients. An interaction between genetic and environmental factors has been suggested as a possible mechanism for the association of arterial and cortical malformations.11 Vascular endothelial growth factor is a key regulator of physiologic angiogenesis during embryogenesis and is also implicated in pathologic angiogenesis associated with a lot of conditions.23 Some other genes, such as homeobox and Eph genes and ephrin proteins, have been linked to capillary24,25 and neural26,27 development. Other candidate genes could be those that are involved in the differentiation of neural crest cells.28
References
- Received August 17, 2006.
- Accepted after revision January 20, 2007.
- Copyright © American Society of Neuroradiology