Abstract
SUMMARY: Histologic patterns of cellular architecture often suggest a tissue diagnosis. Distinctive histologic patterns seen within the peripheral nerve sheath tumor schwannoma include the Antoni A and Antoni B regions. The purpose of this report is to review the significance of Antoni regions in the context of schwannomas.
Microscopically examined histologic patterns of cellular architecture on stained tissue preparations may imply a specific tissue diagnosis. Pathologists rely on these patterns much as radiologists rely on gray-scale patterns on images. The histologic patterns known as Antoni A and Antoni B regions have been recognized as highly suggestive for the peripheral nerve sheath tumor schwannoma. The purpose of this report is to review the significance of Antoni A and Antoni B regions in the context of schwannoma.
What Are Antoni A and Antoni B Patterns?
In 1920, Nils Ragnar Eugène Antoni (1887–1968), a Swedish neurologist and researcher working at the Royal Neurologic Clinic of Stockholm under the supervision of the eminent neuropathologist and now controversial eugenics proponent Frithiof Lennmalm (1858–1924), described 2 distinct patterns of cellular architecture in the peripheral nerve sheath tumors, which would become known as schwannomas.1 Antoni, who would later become professor of neurology at the Karolinska Institute in Stockholm, based his observations on analysis of 30 cases and described a “fibrillary, intensely polar, elongated appearing tissue type” which he called “tissue type A.”1 These highly cellular regions were eventually referred to as Antoni A regions by later authors. Antoni also described seemingly distinct loose microcystic tissue adjacent to the Antoni A regions, and these came to be known as Antoni B regions. Recognition of these patterns has proved useful in the histologic identification of schwannomas.
Nerve Sheath Tumors and Schwannomas
Current concepts in the classification of peripheral nerve sheath tumors have evolved over many decades. In Antoni's time, researchers labored intensely to identify distinguishing features that would allow consistent differentiation of subtypes of these tumors. Now the rubric “peripheral nerve sheath tumor” actually defines several unique pathologic entities including schwannoma, neurofibroma, perineurioma, traumatic neuroma (nonneoplastic; all benign), and malignant peripheral nerve sheath tumor (MPNST).
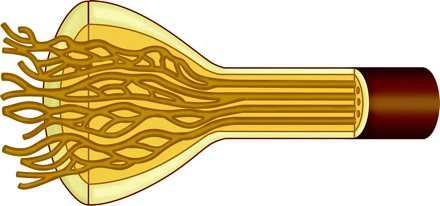
Schwannomas are derived from the myelinating cell of the peripheral nervous system and are composed almost entirely of Schwann cells, which were originally described by the prominent German histologist and physiologist Theodor Schwann (1810–1882). Schwannomas typically grow within a capsule that remains peripherally attached to the parent nerve (Fig 1).2 Most commonly, schwannomas impact the central nervous system (CNS) by involving the vestibular branch of cranial nerve VIII or the dorsal roots of the spinal cord (Figs 2 and 3).3
Diagram of a schwannoma. The tumor (T) originates within a Schwann cell cylinder that surrounds an axon. The tumor subsequently grows, eccentrically compressing the normal adjacent axons. Schwann cell cylinders surrounding axons (light brown), endoneurium (dark yellow), and perineurium (tan and dark brown).
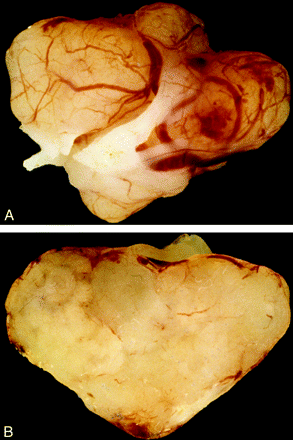
Photograph of surgical resection specimen of an intradural schwannoma arising in a 53-year-old woman. A, The yellowish tan gross specimen exhibits a thin capsule. B, Cut section of the tumor reveals a uniform, solid parenchyma. Architecture of schwannomas may vary and also exhibit cystic or nodular consistency.
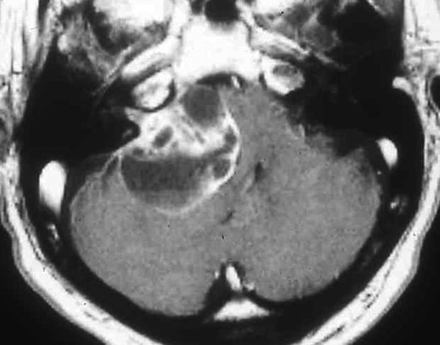
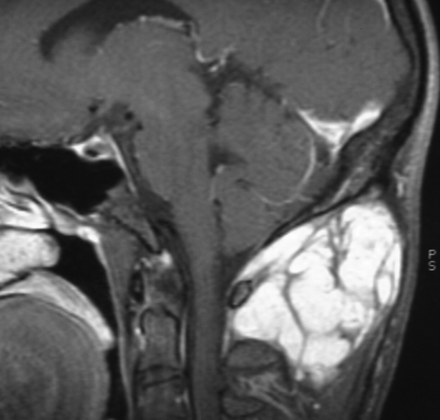
Vestibular schwannoma. Enhanced T1-weighted MR image demonstrating a large predominantly cystic schwannoma arising from cranial nerve VIII within the internal auditory canal. The tumor fills the cerebellopontine angle, compresses the pons, and displaces the fourth ventricle. Debris settles within the dependent portion of the tumor.
By contrast, neurofibromas appear to contain all the cellular elements of a peripheral nerve, including Schwann cells, fibroblasts, perineurial cells, and axons. The tumor cells grow diffusely within and along nerves, causing the nerves to expand radially while entrapping native neural elements within the substance of the tumor (Figs 4 and 5). This intraneural growth pattern, with its entrapped axons, provides a key feature to histologically distinguish neurofibroma from schwannoma. When a neurofibroma involves multiple branches of a nerve plexus or multiple fascicles within a large nerve, it adopts and amplifies the normal anatomic tortuosity to produce a “plexiform” growth pattern that some authors have likened to a bag of worms (Fig 6). Plexiform neurofibromas most commonly occur in the orbit, neck, back, and inguinal areas, most occurring in patients with neurofibromatosis type 1 (NF1).2 Although benign lesions with a generally favorable prognosis, the ultimate treatment outcome for both schwannomas and plexiform neurofibromas depends largely on size, location, and proximity to important anatomic structures.
Diagram of a neurofibroma. The endoneurium (dark yellow) increases in volume. Schwann cell cylinders (light brown) separate from one another and become dolichoectatic. The mature tumor consists of a complex of Schwann cells, axons, and fibrous material within a myxomatous matrix surrounded by a thickened perineurium (tan and dark brown).
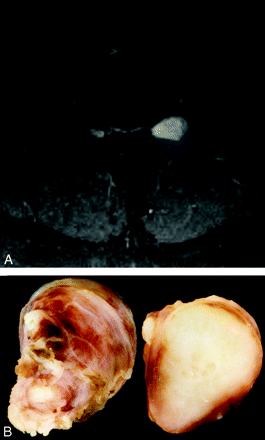
Solitary neurofibroma in a 34-year-old woman. A, Enhanced fat suppressed T1-weighted MR axial image at the L4–5 level demonstrates an enhancing tumor arising from the left L4 root. The neuroforamen is expanded. B, Photograph of cut sections of the gross specimen in the same patient shows a solid heterogenous-appearing tumor, which has grown within and expanded multiple fascicles of the nerve.
Plexiform neurofibroma in a 21-year-old man with no other signs of NF1. Sagittal enhanced T1-weighted MR image demonstrates an enhancing serpentine mass in the pontine soft tissues of the neck.
Other categories of “peripheral nerve sheath tumor” also exist. For example, traumatic neuroma typically arises in the setting of injury to a peripheral nerve, as the name implies (eg, brachial plexus injury associated with obstetric delivery); the resulting mass develops from the nerve's regeneration and a fruitless attempt to re-establish its connections. Histologically, the mass appears as a jumble of benign-appearing nerve minifascicles interwoven with reactive fibrous tissue.2 Perineuriomas are exceedingly rare, representing less than 1% of nerve sheath tumors.4 They most commonly affect peripheral motor nerves, but sensory and cranial nerve involvement has also been reported.5 These tumors are formed by neoplastic perineurial cells arranged into “pseudo-onion bulbs” around axons within the perineurium and endoneurium of nerve fascicles. Thus, axon entrapment distinguishes both perineuriomas and neurofibromas from schwannoma. In contrast, “extraneural” (ie, soft tissue) perineuriomas do not display entrapped axons. The latter are not encountered in the CNS and are, therefore, not discussed further.
The MPNST may develop either de novo or from transformation of a plexiform neurofibroma in a patient with NF1.6 Very rarely, MPNST may arise from a benign schwannoma.7,8 Histologically, MPNST appears as an invasive hypercellular spindle cell lesion with only subtle features to distinguish it from other sarcomas. Clinically, MPNST appears in the same areas as plexiform neurofibroma, but the common sites of origin that impact the CNS are cranial nerve V and the spinal roots; cranial nerve VIII is only rarely involved.9,10 Unlike schwannoma and neurofibroma, which are benign tumors with favorable prognosis, that of MPNST is generally poor. Metastases may develop in the lung, liver, and brain, and, therefore, primary treatment is usually aimed at local control through wide excision when possible. Chemotherapy and radiation therapy are also considerations.
Antoni A regions
Antoni's original work, which dealt with the peripheral nerve sheath tumor type that would ultimately be classified as schwannoma, identified highly cellular zones. These zones were punctuated by nuclei arranged in stacked alignments known as palisades (Fig 7). Within these so-called Antoni A regions, the closely packed elongated nuclei demonstrated a thickened middle portion with tapered ends and resembled the wooden spindles used in textile spinning.1 A decade earlier, the pathologist Jose Juan Verocay noted that bands of these fusiform nuclei alternated with clear zones devoid of nuclei.11,12 These “Verocay bodies” were almost always associated with the densely cellular regions of schwannomas but were by no means pathognomonic of schwannomas and have now been demonstrated in several other tumor types. Interestingly, the palisades within Antoni A regions are more commonly seen in spinal schwannomas compared with intracranial tumors and are often absent in cranial nerve VIII schwannomas.13,14
Photomicrograph of Antoni A tissue within a schwannoma. Wavy, tightly organized nuclear palisades known as Verocay bodies occupy the center of a highly cellular field (hematoxylin-eosin, original magnification ×400).
Electron microscopy of Antoni A tissue demonstrates long interdigitating cell processes covered by a nearly continuous well-formed external layer or lamina and separated by intercellular basement membranes.14–16 Fisher and Vuzevski17 described these cytoplasmic processes as more numerous, longer, and more oriented into parallel bands than those seen in other tumors, such as neurofibromas. These ultrastructural findings correlate with the prominent palisades and Verocay body formation seen in this tissue type.
The nearly ubiquitous basement membrane found in Antoni A regions is rich in laminin, a high-molecular-weight glycoprotein, and is produced by Schwann cells at all stages of development.18,19 Miettinen et al20 investigated benign and malignant soft tissue tumors immunohistologically for the presence of laminin. The results showed intense laminin positivity in schwannomas and neurofibromas and less intense positivity in leiomyomas and leiomyosarcomas, whereas fibrous histiocytomas and fibrosarcomas generally were negative. The immunohistochemical demonstration of attenuated intercellular laminin is, thus, a valuable aid in differentiating between tumors derived from Schwann cells and those derived from fibroblasts.20 Another component of basement membrane, collagen type IV, provides analogous antigen that is often targeted with diagnostic immunohistochemistry in clinical pathology laboratories.
The adhesive properties of laminin are thought to explain the tight organization within Antoni A tissue. Baur et al21 investigated the influence of laminin on cell cultures derived from unilateral vestibular nerve schwannomas. Schwannoma tumor tissue was incubated on both uncoated and laminin-coated culture dishes. Cells cultured with laminin appeared more differentiated with long, fusiform, cytoplasmic processes than cultures without laminin. When laminin was added to the culture medium, cell attachment and proliferation were stimulated with the effect increasing roughly proportional to the dose of laminin.21
Cells cultured with laminin also expressed S-100 protein providing further support for their Schwann cell derivation. S-100 is a highly acidic protein that is found in many neural crest tumors and may play a role in the ionic regulation of nervous tissue.22 Although not entirely specific, S-100 protein is nonetheless a reliable marker for the diagnosis of schwannomas and is especially prevalent in the Antoni A areas.14,22,23 In contrast, neurofibromas only show patchy positivity for S-100, because Schwann cells represent only a subset of the tumor constituency.24 Moreover, MPNSTs contain only limited amounts of S-100 protein in roughly half of the cases. Its expression often corresponds with the degree of differentiation within these typically high-grade tumors.22
Antoni B Regions
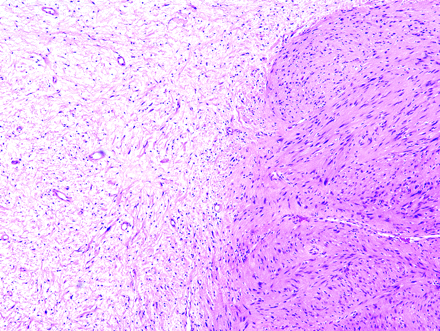
In contrast to the tightly organized Antoni A regions, the Antoni B regions tend to be relatively less cellular and contain more loosely arranged cells (Fig 8).25,26 Although intermingled, islands of type A and type B tissues usually appear fairly well demarcated from one another. The volume of Antoni B regions in any given tumor is variable and can occasionally be scant or completely lacking. Cells within the Antoni B regions are often thin and wispy, separated from one another by microcystic spaces filled with basophilic mucin.27 Microcysts may coalesce into radiologically detectable macrocysts. Interestingly, cranial nerve VIII schwannomas have an abundance of Antoni B tissue, which may account for the frequently observed macrocystic regions (Fig 3). In addition, Antoni B tissue contains many lipid-laden histiocytes, lymphocytes, and small vessels with hyalinized walls.14,16,25,28
Photomicrograph of Antoni A tissue and Antoni B tissue within a schwannoma. The highly cellular Antoni A region on the right of the field is contrasted with the loosely organized hypocellular Antoni B region on left of the field (hematoxylin-eosin, original magnification ×400).
Neoplastic Schwann cells in vitro show increased angiogenic properties similar to other tumor cells.29 The vascularization of these Antoni B areas is varied, sometimes aberrant or bizarre, but often contains degenerative features. Acute and chronic thrombosis, hemorrhage, and hyaline thickening of these ectatic vascular walls can be seen. Areas of frank necrosis further contribute to observed cystic regions.
Within type B tissue, areas of so-called “ancient change” may also be seen. Twisted cytoplasm and enlarged hyperchromatic and markedly atypical nuclei may be present without increased cellular attenuation or mitotic activity to denote anaplastic change. Prominent fibrous stroma is rarely punctuated by attenuated bands of hyalinized cartilage, particularly in mediastinal tumors.30 Cysts as described above, focal calcification, and mucinous and xanthomous change involving the tumor cells are also common and thought to represent a degenerative phenomenon. Xanthomatous changes can produce areas that are yellow to the naked eye and that demonstrate aggregated foamy macrophages. Lymphocytic infiltrates are also relatively common.
An explanation for the presence of 2 distinct Antoni tissue types within the same tumor eluded investigators for many years. Separating the regions in many tumors is a transitional zone with both Antoni A and B features.31 This zone also contains detached segments of basal lamina debris, a diminished presence of S-100 protein, and strands of fibrin suggesting a degenerated Antoni A region.26,31,32 In addition, within Antoni B regions, some investigators have observed granules resembling myelin sheaths within presumably phagocytic vacuoles.31 Given these features, some researchers now contend that the Antoni B areas may embody degenerated Antoni A regions, with the transitional zone representing a stage in the deterioration process.14,31 The hyalinized blood vessels commonly encountered within Antoni B tissue may lead to ischemia, are prone to hemorrhage, and may further contribute to degeneration.14,31
Molecular Biology as a Complement to Histology
In the 80 years since Antoni described the features of schwannoma, pathologists continue to rely on histologic patterns for tumor diagnosis and grading. However, the understanding of molecular biology and protein expression has greatly impacted our understanding of these tumors.
Schwannomas, both sporadic and those associated with neurofibromatosis type 2 (NF2), most commonly show deletion of the NF2 locus known as 22q12.2 (rare translocations have also been reported).33–37 The NF2 gene encodes merlin (ie, schwannomin), a tumor suppressor protein of which the expression is typically lost in most schwannomas. Although the mechanism by which merlin influences the cell cycle is not fully understood, recent studies describe merlin's interaction with and inhibition of several proteins involved in the cell cycle and mitogenic signal intensity transduction. These proteins include the following: MLK3 (mixed lineage kinase 3), which stabilizes the B-Raf/Raf-1 complex within the mitogen-activated protein kinase pathway and is required for schwannoma tumor cell proliferation38; HEI10, which controls the accumulation of cyclin B, a protein critical to cell cycle control39; and GTPase PIKE-L (phosphatidylinositol 3-kinase [PI3Kinase] enhancer), which promotes cell survival by activating PI3Kinase.40
Neurofibromas also demonstrate loss of a tumor suppressor gene NF1, located on chromosome 17q12.41 This gene encodes the tumor suppressor protein neurofibromin. Cells deficient in this protein demonstrate altered actin cytoskeletons, increased cell motility, increased cell proliferation, and elevated activity within the RAS signaling pathway.42–44 Neurofibromin has been identified as a GTPase activating protein for the GTPase oncoprotein K-RAS.45 By facilitating GTPase activity of K-RAS, neurofibromin effectively inhibits K-RAS, preventing K-RAS from activating the tuberous sclerosis complex/mammalian target of rapamycin pathway. In this manner, neurofibromin inhibits proliferation.46,47
Even with this very basic understanding of the different molecular features of schwannoma and neurofibroma, one might predict that future diagnoses will be rendered (and treatments applied) on the basis of genetic or even proteomic analysis. Currently, however, differentiating these entities relies heavily on histologic features, such as those described by Antoni.
Imaging and Antoni Tissue Types
Although imaging alone cannot consistently differentiate benign from malignant nerve sheath tumors or schwannoma from neurofibroma, the Antoni tissue types A and B patterns may have an impact on the neuroimaging features. For example, Gomez-Brouchet et al48 found that a heterogeneous MR imaging appearance of larger tumors was seen more commonly in lesions with a higher ratio of type B to type A tissue. This heterogeneous appearance also seemed related to other pathologic changes, mainly hemosiderin deposits and cystic formation seen within type B tissue. Their data suggested that increasing tumor size probably depends less on proliferation rate than on dystrophic changes, such as hemosiderin deposition and cyst formation and/or on the presence of type B tissue.48 Moreover, larger lesions may be inclined to degenerate as blood supply is compromised leading to degeneration, development of Antoni B tissue, and eventual cystic change on imaging. Similarly, on CT, attenuated portions of schwannomas may correlate with Antoni A tissue, whereas hypoattenuated regions tend to correlate with Antoni B tissue.49,50 A so-called target sign has also been described in some nerve sheath tumors and consists of a central core of hypointensity surrounded by hyperintensity on T2-weighted and enhanced T1-weighted images.51,52 The central region contains fibrocollagenous tissue, and the peripheral region consists of myxomatous tissue; however, histologic correlation often fails to reveal a consistent association with tumor type or Antoni A or Antoni B regions.51
Conclusion
In summary, Antoni A and B tissue types represent distinct histologic architectural patterns that aid in the pathologic diagnosis of schwannomas and may influence their imaging characteristics. Type A tissue is highly cellular and demonstrates nuclear palisading and associated Verocay bodies, which may reflect their prominent extracellular matrix and secretion of laminin. Type B tissue is loosely organized with myxomatous and cystic changes and may represent degenerated Antoni A tissue.
References
- Received January 11, 2007.
- Accepted after revision April 20, 2007.
- Copyright © American Society of Neuroradiology