Abstract
BACKGROUND AND PURPOSE: The development of a new polyp or mass in the radiation field of a previously treated carcinoma is usually an ominous sign of a recurrent cancer, but rarely may it be caused instead by a nonmalignant process. The purpose of this study was to document the MR appearance of unusual nonmalignant polyps or masses (NMPMs) in the nasopharynx and sphenoid sinus arising after radiation treatment of nasopharyngeal carcinoma.
MATERIALS AND METHODS: The MR imaging reports of patients undergoing imaging after radiation therapy for nasopharyngeal carcinoma were reviewed retrospectively to identify patients with unusual polyps and masses in the nasopharynx. The MR images of those patients with no evidence of malignancy on biopsy or follow-up were reviewed.
RESULTS: The MR imaging reports of 1282 patients were reviewed, and 11 patients (1%) with NMPMs in the nasopharynx or sphenoid sinus were identified. Two patterns were identified: contrast enhancing nasopharyngeal polyps ranging in size from 1 to 5 cm (n = 5) and sphenoid sinus masses consisting of a nonenhancing mass filling a nonexpanded sinus (n = 4) and a heterogeneous enhancing mass expanding the sinus (n = 2). Osteoradionecrosis produced a large defect in the roof of the nasopharynx causing direct communication with the sphenoid sinus (n = 6). Histology revealed granulation tissue in all of the patients with variable amounts of fibrin and inflammatory cells. A direct infective etiology was not proved in any patient.
CONCLUSION: NMPMs in the nasopharynx and sphenoid sinus are rare complications after radiation therapy to the skull base, but the radiologist needs to be aware of their appearance so that they can be considered in the differential diagnosis of suspected tumor recurrence.
The rapidly dividing cells in the mucosal membranes of the pharynx and paranasal sinuses are very sensitive to the effects of radiation. The severity of radiation damage after treatment of head and neck cancers is related to radiation dose and potentially is increased by the use of chemotherapy and altered fractionation schedules of radiation therapy.1 The naso-pharynx and sphenoid sinus are especially vulnerable to the effects of radiation treatment of nasopharyngeal carcinoma, because they receive the full radiation dose. Acute mucositis is a consistent clinically visible adverse effect during the standard course of radiation therapy for head and neck cancers. It starts around the second to third week of treatment and usually subsides several weeks after the end of treatment.2 Likewise, acute radiation change can be seen on MR imaging. In the pharynx, these MR abnormalities often resolve, whereas in the paranasal sinuses there is a high incidence of persistent minor abnormalities, including mucosal thickening and fluid levels, months or years after treatment for nasopharyngeal carcinoma.3–5 Rarely a mucocele may form in the sphenoid sinus.6 However, there are some patients who go on to develop severe delayed radiation effects resulting in the formation of unusual nonmalignant polyps and masses (NMPMs) in the nasopharynx and sphenoid sinus. These radiation-induced injuries cause both clinical and radiologic problems with distinction from recurrent cancer, as well as being a cause of serious morbidity and even mortality. The aim of this study was to describe these abnormalities in patients undergoing MR imaging after radiation therapy for nasopharyngeal carcinoma.
Methods
The local ethics committee granted ethical approval for this retrospective study. The MR imaging reports of patients undergoing imaging after radiation therapy treatment for nasopharyngeal carcinoma between 1995 and 2006 were reviewed to identify patients with exophytic polyps in the nasopharynx or a mass in the sphenoid sinus that did not have the typical features of posttreatment inflammatory sinus changes (mild thickening and mucosal enhancement and/or secretions of high T2 and low T1 signal intensity). The clinical records of these patients were reviewed, and those with evidence of local malignancy on biopsy and follow-up were excluded from the study. The MR images of the patients with NMPMs were reviewed. All of the patients underwent MR imaging on a 1.5-MR unit (Gyroscan; Philips, Best, the Netherlands) and all of the patients over this time period underwent the same standard MR protocol consisting of an axial fat-suppressed T2-weighted sequence (TR/TE, 2500/100 ms; echo-train length, 15; FOV, 22 cm; section thickness, 4 mm, with no intersection gap; matrix size, 256 × 202), coronal T2-weighted turbo spin-echo (TR/TE, 2500.100 ms; echo-train length, 14; FOV, 22 cm; section thickness, 4 mm, with no intersection gap; matrix size, 256 × 202), axial T1-weighted spin-echo (TR/TE, 500/20 ms; FOV, 22 cm; section thickness, 4 mm, with no intersection gap; matrix size, 256 × 202), and contrast-enhanced T1-weighted spin-echo images by using a 512 × 512 matrix in the axial and coronal planes after a bolus injection of 0.1 mmol/kg of gadolinium dimeglumine (Schering; Berlin, Germany). In addition to the standard protocol above, most patients underwent a T1-weighted sequence postcontrast with fat saturation, and some sequences were also performed in a sagittal plane.
Results
The MR imaging reports of 884 patients undergoing 1597 MR examinations were available for review. Eleven patients (1%) with delayed onset of NMPMs in the nasopharynx or sphenoid sinus were identified (9 men and 2 women; age range, 39–71 years; mean age, 54 years). All of the patients were referred because of suspected tumor recurrence based on symptoms or endoscopy findings during clinical follow-up or previous imaging. Treatment was composed of conventional radiation therapy (n = 3); conventional radiation therapy plus either chemotherapy (n = 2), stereotactic radiation therapy (n = 1), brachytherapy (n = 2), or nasopharyngectomy for local tumor recurrence (n = 1); and hyperfractionated radiation therapy (n = 1). In 1 patient, details of radiation treatment were unknown.
Ten patients had a history of chronic nasopharyngitis (ranging from 2 to 11 years), one of whom also had chronic osteomyelitis of the skull base after nasopharyngectomy. Nasopharyngeal biopsies identified candida in 3 patients, 1 of whom also showed fungal hyphae. Acid fast bacilli were not identified, though 3 patients had a history of pulmonary tuberculosis either before or after treatment.
NMPMs were seen on MR images obtained 2 to 14 years (mean, 8 years; median, 10 years) after radiation therapy. Details of the radiologic findings are shown in the Table and On-line Table 1. In summary, 2 patterns of NMPMs were identified. The first was a nasopharyngeal polyp (n = 5), which ranged in size from 1 to 5 cm and showed mixed heterogeneous T2 signal intensity and marked contrast enhancement, the larger polyps having stellate areas of reduced enhancement (Figs 1–⇓⇓4). The second was a sphenoid sinus mass, which consisted of nonenhancing mass filling a nonexpanded sinus (n = 4; Fig 5) and a heterogeneous enhancing mass expanding the sinus (n = 2; Fig 6). All of the patients had some degree of inflammatory mucosal change in the paranasal sinuses or radiation damage and posttreatment scar tissue in the paranasopharyngeal regions, but none of the NMPMs appeared directly infiltrative. There was direct communication between the nasopharynx and sphenoid sinus via a large osteoradionecrotic defect in the bony roof of the nasopharynx/floor of the sphenoid sinus in 6 patients, 2 with nasopharyngeal polyps and 4 with a nonenhancing sphenoid sinus mass.
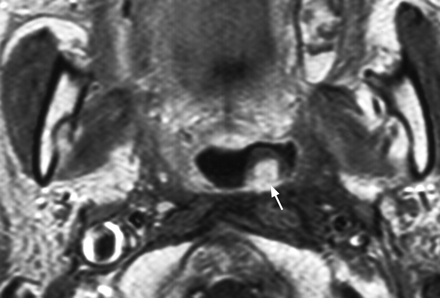
Axial T1-weighted postcontrast MR image in a 44-year-old man with a small contrast-enhancing polyp (arrow) arising from the posterior wall of the lower nasopharynx 11 years after chemoradiotherapy. Posttreatment scarring is present in the left lateral nasopharyngeal wall, partially effacing the parapharyngeal fat, and around both carotid sheaths.
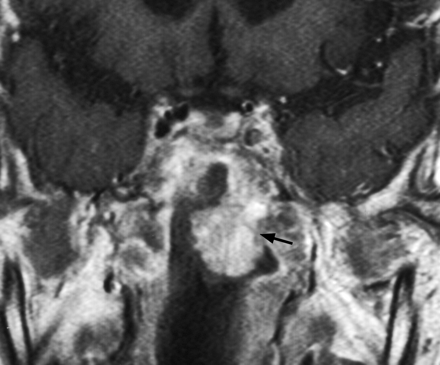
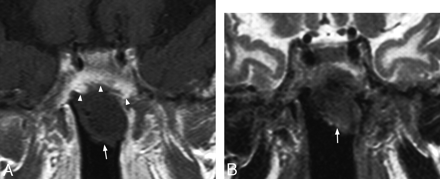
Coronal T1-weighted postcontrast MR image in a 71-year-old man, 11 years after radiation therapy, with a contrast enhancing polyp (arrow) with less enhancing stellate areas centrally, arising at the junction of the sphenoid sinus and roof of the nasopharynx where there is an osteoradionecrotic bony defect.
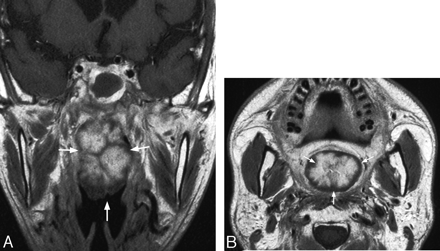
A, Coronal and (B) axial T1-weighted postcontrast MR image in a 40-year-old man with a 5-cm rapidly growing contrast-enhancing polyp with a more central area of reduced contrast enhancement radiating to the periphery, 2 years after treatment with conventional radiation therapy plus a stereotactic radiation therapy boost. Inflammatory changes and retained secretions are present in the sphenoid sinus.
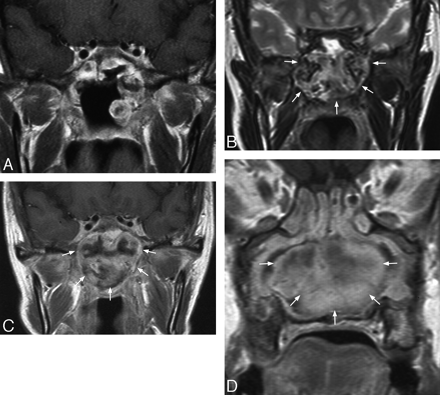
Coronal T1-weighted postcontrast MR image in a 42-year-old woman with enhancing polyps in the left lateral wall of the nasopharynx and sphenoid sinus and a large defect in the sphenoid sinus floor, 11 years after conventional radiation therapy and 3 years after a nasopharyngectomy for local tumor recurrence (A). These polyps remained static before rapidly increasing in size on MR imaging 32 months later to form a large heterogeneous mass in the nasopharynx expanding into the sphenoid sinus and nasal cavity. On the T2-weighted image, the mass shows heterogeneous mixed signal intensity (B), and on the T1-weighted image postcontrast there is heterogeneous enhancement (C) with a less enhancing stellate area centrally in the nasal cavity component (D).
A, Coronal T1-weighted postcontrast MR image in a 69-year-old man with osteoradionecrosis causing a large defect in the sphenoid sinus floor and a nonenhancing mass (arrow) within the sphenoid sinus that also shows thickened enhancing mucosa in the roof of the sinus (arrowheads), 5 years after radiation therapy (“rhinolith” at surgery). B, Coronal T2-weighted MR image in the same patient showing the mass is of low-intermediate signal intensity (arrow) and radiation-induced injury in the white matter of the inferior aspects of both temporal lobes.
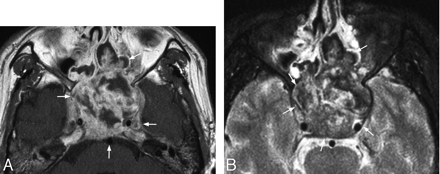
A, Axial T1-weighted postcontrast MR image in a 39-year-old man with a heterogeneous enhancing mass causing expansion of the sphenoid sinus (arrows) 4 years after radiation therapy. B, Axial T2-weighted MR image in the same patient showing a heterogeneous mass (arrows) with foci of very low T2 signal intensity presumed to be old hemorrhage.
Radiologic findings, histology, and follow-up in 6 patients with a nonmalignant mass in the sphenoid sinus
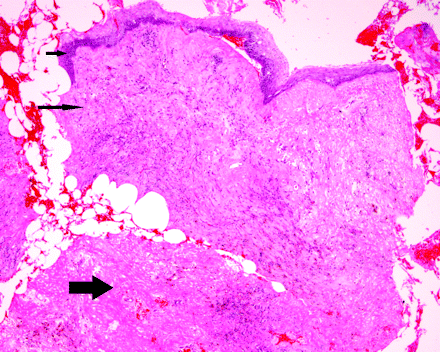
Histology revealed variable amounts of granulation tissue, fibrin, and inflammatory and epithelial cells but no malignant cells (Fig 7). Details of histology and clinical/radiologic follow-up are shown in the Table and On-line Table 1.
Photomicrograph showing a polypoidal mass that is lined by benign squamous epithelium (short arrow) with proliferation of granulation tissue in the underlying stroma (long arrow) together with a fibrin deposit (thick arrow).
Discussion
The target volume for radiation treatment of nasopharyngeal carcinoma routinely covers the nasopharynx and at least the lower half of the sphenoid sinus, and, hence, these areas are vulnerable to the effects of radiation. In addition, the thin plate of bone separating the floor of the sphenoid sinus from the roof of the nasopharynx is especially vulnerable to osteoradionecrosis, resulting in bony defects that allow direct communication between the sinus and the pharynx. Two nonmalignant patterns of disease are identified in this study, nasopharyngeal polyps and sphenoid sinus masses, though there is some overlap in these 2 patterns. The exophytic markedly enhancing nasopharyngeal polyps, composed of variable amounts of granulation tissue, fibrin, and inflammatory cells, could grow to an alarming size and fill the nasopharyngeal cavity. The sphenoid sinus masses were divided according to whether they showed contrast enhancement. All of the patients with noncontrast-enhancing masses had osteoradionecrosis in the floor of the sphenoid sinus, causing a large bony defect. These masses are believed to represent inspissated secretions/concretions, a view supported by the case of a large “rhinolith” protruding down into the nasopharynx, which was removed surgically (Fig 5). In the 2 patients with a contrast-enhancing sphenoid sinus mass, there was no large bony defect in the sphenoid sinus floor, thereby preventing any decompression into the nasopharynx, and in both cases the sphenoid sinus showed progressive expansion (Fig 6). Simple mucoceles have been reported previously as a complication of radiation therapy for nasopharyngeal carcinoma6–9 and are believed to result from obstruction by scar tissue. It is postulated that the expansion of the sphenoid sinus in this study was caused by ongoing chronic inflammatory and radiation changes obstructing the sinus ostium. Repeated hemorrhages from telangiectasia are common in irradiated mucosal tissues and may have played a role also in the expansion of the sphenoid sinus mass, one patient revealing an organized hematoma on the surgical excision biopsy. In addition, both patients showed areas of very low T2 signal intensity in the sphenoid mass (Fig 6). Given that neither patient had evidence of a fungal infection,10 it is postulated that this low T2 signal intensity represented old hemorrhage. These nonmalignant sphenoid masses are particularly difficult to treat, and one patient died as a result of repeated intracranial infection.
Most patients who undergo follow-up imaging after the treatment of nasopharyngeal carcinoma will do so because of the clinical suspicion of local tumor recurrence, a diagnosis that can be difficult to distinguish from NMPMs by endoscopy. In general, the MR appearance of recurrent nasopharyngeal carcinoma is similar to that of the original primary tumor. This cancer tends to have a long base along the nasopharyngeal wall from which it bulges into the nasopharyngeal cavity and infiltrates into the deep tissues. It is usually of homogeneous intermediate T2 and low/intermediate T1 signal intensity and shows only moderate homogeneous contrast enhancement on nonfat suppressed T1-weighted MR images. In contrast, the nonmalignant nasopharyngeal polyps in this study were more exophytic, protruding into the nasopharyngeal cavity without any deep infiltrative component, and on the postcontrast images where there was more marked contrast enhancement resulting from the inflammatory/granulomatous reaction. In the larger polyps, a more stellate central area of reduced enhancement could be seen, resulting in less homogeneous enhancement than is found in nasopharyngeal carcinoma. The degree of contrast enhancement is also useful in distinguishing nasopharyngeal carcinoma from the 2 forms of nonmalignant sphenoid masses, the latter showing either no enhancement or marked, often very heterogeneous, enhancement.
Unfortunately, nasopharyngeal carcinoma occasionally incites a granulomatous reaction causing more marked enhancement, and, therefore, exclusion of recurrent tumor has to rely on biopsy and follow-up. However, whereas an initial biopsy is usually required to confirm the diagnosis, it is important that the radiologist should include NMPMs in the initial differential diagnosis so that the clinician does not have to embark on a fruitless series of repeated biopsies chasing an MR imaging diagnosis of recurrent tumor. The role of PET in these patients is unclear, but it is known from 2 case reports that avid uptake of fluorodeoxyglucose–positron-emission tomography is not only restricted to recurrent tumor but may also be found in a radiation-induced mass in the nasopharynx11 and osteoradionecrosis in the skull base.12
Radiation-induced sarcoma is rare but a recognized sequela of radiation therapy for nasopharyngeal carcinoma, with these tumors frequently arising at a similar time to NMPMs. Unfortunately, the MR appearance of radiationinduced sarcoma may be similar to NMPMs, with both disease entities causing heterogeneous masses of mixed signal intensity that show marked contrast enhancement. On the other hand, radiation-induced sarcomas are usually much more infiltrative and destructive than NMPMs and do not tend to cause large exophytic type masses. The main difficulty for MR diagnosis lies in distinguishing between a radiation-induced sarcoma of the sphenoid sinus and the expansile contrast-enhancing form of the nonmalignant sphenoid mass, with the correct diagnosis relying on biopsy and follow-up.
The etiology of NMPMs is probably a combination of radiation-induced injury and chronic infection originating in the nasopharynx, sinus, or underlying bone. Infection is thought to play a role in the development of radiation-induced mucositis, with colonization by gram-negative enterobacteria being implicated.13–15 Candida,16 fungi, and tuberculosis may also have a role. Tuberculous infection has been reported after radiation therapy for nasopharyngeal carcinoma17,18 and produces a range of MR appearances in the nasopharynx, which include contrast-enhancing granulation polyps.19 Although tuberculosis was not isolated from the nasopharynx of any patient in this study, 3 patients did have a history of pulmonary tuberculosis. Unfortunately, it is difficult to determine the contribution of infection, because many microbes in the nasopharynx are normal commensals rather than pathogens. In addition, the interpretation of nasal swab cultures is difficult in this clinical setting, whereas repeated biopsies should be avoided because of the concern that it will aggravate the radionecrosis or bleeding. Furthermore in the case of tuberculosis, repeated biopsies often remain negative.20,21
NMPMs were rare in this study, found in only 1% of patients treated previously by radiation therapy for nasopharyngeal carcinoma. (Two of these cases have been reported previously.22,23) This figure probably underestimates the true incidence, because regular follow-up MR imaging is not routinely performed at our institution, not all of the MR imaging reports could be obtained, and the initial documentation of the complications was based on the MR reports rather than review of the MR images. However, from our experience, these abnormalities are generally quite rare.
In summary, the nasopharynx and lower portion of the sphenoid sinus receive the full radiation dose during treatment for nasopharyngeal carcinoma, and osteoradionecrosis of the thin bony roof of the nasopharynx can result in a common compartment between the sinus and pharynx. NMPMs may be found in this region composed of contrast-enhancing polyps in the nasopharynx, nonenhancing “rhinoliths” in the sphenoid sinus, and enhancing masses expanding the sphenoid sinus. The appearances of these NMPMs are unusual for local tumor recurrence but can be similar to radiation-induced sarcomas, and the final diagnosis will rely on biopsy and follow-up.
Footnotes
indicates article with supplemental on-line table.
References
- Received November 6, 2007.
- Accepted after revision January 12, 2008.
- Copyright © American Society of Neuroradiology