Abstract
SUMMARY: Our aim was to better understand and improve the accuracy of the preoperative diagnosis of intraspinal MM by a combined analysis of MR imaging and pathologic findings. All 5 patients had undergone unenhanced and contrast-enhanced MR imaging examinations. All tumor samples had immunohistochemical reactions to HMB-45, vimentin, S-100, EMA, and Leu-7 antibodies. All 5 cases were located in the intradural extramedullary compartment. Two cases had multifocal lesions, and 3 cases were solitary. Two cases showed homogeneously strong enhancement, and 3 cases showed moderate enhancement on contrast-enhanced T1WI. The tumor cells had positive reactions to HMB-45, vimentin, and S-100 antibodies. MR imaging plays an important role in the detection and diagnosis of intraspinal MM. Final diagnosis should be based on histopathology and IHC examinations.
ABBREVIATIONS:
- EMA
- epithelial membrane antigen
- HMB-45
- human melanoma black-45
- IHC
- immunohistochemistry
- MM
- meningeal melanocytoma
Primary melanocytic neoplasms of the CNS are lesions arising from melanocytes of the leptomeninges. Melanocytes are melanin-producing cells that derive from the neural crest during embryogenesis and then migrate to the skin, mucous membranes, and CNS.1 Melanocytes are often concentrated at the base of the brain and on the ventral surface of the cervical spinal cord. Melanocytic neoplasms of the CNS are rare and most frequently are metastatic in origin. Primary melanocytic lesions of the CNS include neurocutaneous melanosis, MM, primary leptomeningeal melanomatosis, and melanoma.
MM is a rare primary benign melanotic tumor of the leptomeninges, which occurs anywhere in the cranial or spinal region but most commonly in the supratentorial and thoracic spine regions.2 It was first described by Limas and Tio3 in 1972 with a prolonged clinical course and benign histologic features.4 From 1972 to 2009, <60 cases of intraspinal MM were reported.5 Here, we report 5 cases of intraspinal MM. To our knowledge, this study is the largest collection of intraspinal MM cases with MR imaging to date.
Materials and Methods
We searched the data base of our hospital for all intraspinal tumors from 2003 to 2010. The clinicopathologic data and MR imaging findings of 5 cases of MM were analyzed retrospectively. The patients included 1 woman and 4 men with the average age of 49 years. The main clinical symptoms were gradually worsening debilitation of the lower limb (n = 5), leg numbness (n = 3), lumbodynia (n = 1), dorsodynia (n = 1), and dizziness (n = 1). The On-line Table shows the clinical data and MR imaging findings of all patients.
The patients were examined with MR imaging at 1.5T (n = 3) or 3T (n = 2). Spin-echo or fast spin-echo T1WI and T2WI in the axial, coronal, or sagittal planes were acquired. The corresponding contrast-enhanced T1WI was obtained after intravenous injection of 0.1 mmol/kg of gadopentetate dimeglumine (Magnevist; Schering, Berlin, Germany).
Two neuroradiologists reviewed the MR images separately. All lesions were removed by open surgery. HMB-45, vimentin, S-100, EMA, and Leu-7 antibodies were purchased from Dako North America (Carpinteria, California). The final diagnoses were established by 2 pathologists after reviewing the clinical process, the macroscopic protocols, and the histologic slides.
Results
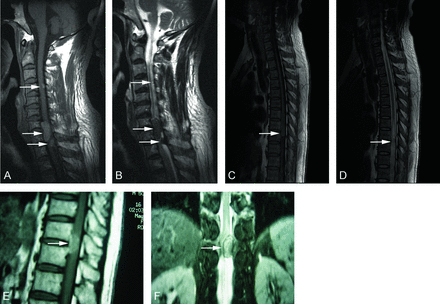
All 5 cases were located in the intradural extramedullary compartment, with the results revealing that the spine was obviously compressed (Figs 1 and 2). All lesions were tightly attached to the dura mater. Two cases were multifocal and others had solitary lesions. The single lateral intervertebral foramen was widened (n = 1). T1WI showed homogeneous hyperintensity (n = 2), moderate hyperintensity (n = 2), and isointensity (n = 1); T2WI showed homogeneous hypointensity (n = 3), moderate hyperintensity (n = 1), and isointensity (n = 1). Two cases showed homogeneously strong enhancement, and others showed moderate enhancement on contrast-enhanced T1WI (Fig 1 and On-line Table).
In case 1 (A and B), 3 lesions are shown on sagittal nonenhanced MR images of the cervical vertebrae at the C4-T2 level. In case 2 (C and D), the lesion is shown on sagittal nonenhanced MR images of the thoracic vertebrae at the T7-T8 level. In case 3, the lesion is shown on sagittal (E) and coronal (F) nonenhanced MR images of the thoracic vertebrae at the T11-T12 level.
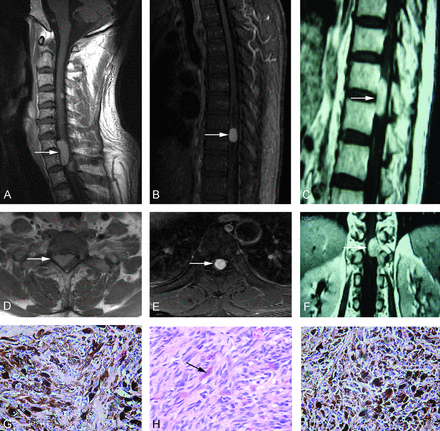
All the tumors showed well-defined, round, or ovoid masses. Three cases were solitary, and others were multifocal. The cut surfaces and peripheral meninges of the tumors were all in black. Tumor cells were spindle, epithelioid, or polygonal with round vesicular nuclei, prominent nucleoli, and plenty of melanin granules in the cytoplasm. They had a uniform cytologic appearance and lacked evidence of atypia or significant mitotic activity. Tumor cells demonstrated storiform vasocentric arrangements. In case 2, the amount of melanin-containing cells was <10%, while in the other 4 cases, >10% of tumor cells had melanin granules in the cytoplasm, even masking the morphology of the nucleus (Fig 2). Tumor cells had a positive reaction to anti-HMB-45, vimentin, and S-100 antibodies but a negative reaction to anti-EMA and Leu-7 (Fig 3). All 5 cases were finally diagnosed as intraspinal MM. All patients underwent surgical resection and radiation therapy. To date, 4 patients are alive (22–37 months after surgery; mean, 28 months), and follow-up MR imaging showed no signs of tumor recurrence. One patient with multifocal lesions developed malignant melanoma and died of metastasis 13 months after surgery.
In case 1, the sagittal (A) and axial (D) contrast-enhanced T1WI and morphologic changes (G, HE original magnification ×400) of the lesion are seen. In case 2, the sagittal (B) and axial (E) contrast-enhanced T1WI sequences and morphologic changes (H, HE original magnification ×200) of the lesion are seen. In case 3, the sagittal (C) and axial (F) contrast-enhanced T1WI sequence and morphologic changes (I, HE original magnification ×400) of the lesion are shown.
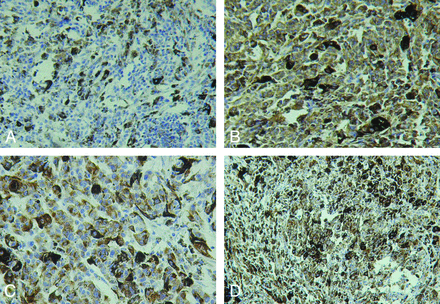
IHC detection of MM (DAB stain). The tumor cells have a negative reaction to the EMA antibody (A, original magnification ×200) but positive reactions to vimentin (B, ×200), HMB-45 (C, original magnification ×400), and S-100 (D, original magnification ×200).
Discussion
Melanocytes originate from the neural crest and are normally found in the leptomeninges.2,4 These cells are present in the leptomeninges of healthy adults and may result in primary leptomeningeal pigmented tumors, including pigmented meningioma, malignant melanoma, MM, melanotic schwannoma, and melanoblastosis.6,7 Malignant melanoma and MM are of the same origin and represent the malignant and benign ends of the tumor spectrum, respectively.5
MM was first coined by Limas and Tio3 in 1972. It is a very rare, pigmented, and slow-growing benign neoplasm.8 However, malignant transformation of melanocytoma to malignant melanoma has been reported.5,9 To date, approximately 112 cases of MM have been reported,5 and most were found in the cerebrum. The tumor occurs almost exclusively in the posterior fossa and spinal cord.6,7,10,11 The annual incidence of MM is estimated to be 1 case per 10 million people, and women are vulnerable.10 Our hospital has an extremely large collection area for patients from Guangdong province and 4 neighboring provinces. The orthopedics center in our hospital is very famous and is also a referral center for spinal tumors. Therefore, we have collected 5 cases of patients with MM in the past 7 years. The incidence is not affected by race or genetic risk factor, and there are no literature reports about MM, to our knowledge. MM may occur at any age but is most commonly seen in the fifth decade. The average age in our series is 49.2 years, which is consistent with that in other reports.10,11
Approximate 59 cases of intraspinal MM have been reported.5 Nineteen cases were founded in the intramedullary location, while others were located in the extramedullary area.5,9 The clinical signs and symptoms depend on its location and may present as myelopathy and seizures.10,12 The symptoms of our patients were nonspecific, mainly caused by mass compression.
Most MMs are commonly found as a solitary lesion, but they can also present as multifocal or disseminating along the arachnoid and dura mater.13,14 To date, approximately 4 cases with multifocal lesions have been reported.15,16 Ali et al6 considered multifocal MM as a distinct pathologic entity that seems to portend an aggressive clinical course and a poor prognosis, even when malignant pathologic features are absent.
In our study, 2 patients with multifocal lesions did not show malignant pathologic features compared with those with solitary lesions, but 1 patient with multifocal lesions developed malignant melanoma and died of metastasis 13 months after surgery. It has been reported that MM might transform into malignant melanoma9,17 and then cause distant metastasis.28 Our study also supports multifocal MM possibly predicting a poor outcome.
MR imaging is the first choice for spinal tumors. On MR imaging, MM is generally an attenuated extra-axial lesion attached to the dura, which may not have a distinct margin.5 It is typically hyperintense on T1WI and hypointense on T2WI and enhances homogeneously with gadolinium.10,16⇓⇓–19 The characteristic signal intensity of MM is thought to be caused by the paramagnetic susceptibility effect of the melanin pigment or hemorrhagic foci; hence, its radiologic signal intensity is variable and inconstant.16,19,20 However, differences in duration, different amounts of melanin, and hemorrhage in malignant melanoma may influence the signal intensity of tumor cells on MR imaging.9,14,19,20
Amelanotic melanoma and melanoma without hemorrhage are iso- or hypointense on T1WI and moderately hyperintense on T2WI.21⇓–23 Isiklar et al24 reviewed the MR images of 30 patients with intracerebral melanoma and sorted these into 4 groups: melanotic pattern (hyperintense on T1WI, hypointense on T2WI), amelanotic pattern (hypointense or isointense on T1WI and hyperintense or isontense on T2WI), indeterminate or mixed pattern (MR imaging characteristics not conforming to the first 2 categories), and hematoma pattern (MR imaging exhibiting only hematoma characteristics).27 In our study, cases with >10% of melanin-containing cells showed a melanotic pattern, while case 2 did not have typical MR imaging and was then proved to have <10% melanin-containing cells by histopathology.
Wadasadawala et al22 considered the following aspects useful for the differential diagnosis of MM: 1) Location: Pigmented meningioma is located in the extra-axial area; melanotic schwannoma is located in the cerebellopontine angle; MM and primary melanoma are often located in the high cervical cord or perimedullary area, whereas metastatic melanoma is located in the supratentorial or gray-white matter junction. 2) Number: The lesion of pigmented meningioma, MM, or primary melanoma is often single, while metastatic melanoma is multiple. 3) Hemorrhage: The pigmented meningioma and melanotic schwannoma do not have a hemorrhagic focus, while MM and primary and metastatic melanoma often have it.22 4) Dural involvement: Dural involvement and the “dural tail” sign are often observed in meningioma, MM, or meningeal melanocytosis, but metastatic melanoma seldom presents dural involvement.
Because the differential diagnosis of MM from other pigmented malignancies is difficult on the basis of only the imaging information, a final diagnosis is dependent on the results of pathologic examination. Grossly, MM usually presents as a solitary well-circumscribed black mass, while lesions in red, white, tan, brown, or blue have been reported.10,23,24 MMs may be mistaken for meningioma at gross inspection because they are attached to the meninges. MM cells tend to compress adjacent tissues instead of infiltrating them. However, local invasion of neural structures has been reported in some cases.6,25,26 The cytologic feature of MM is uniform, lacking in atypia and mitotic activity and showing different degrees of melanization.12,27⇓⇓–30
IHC staining is always indispensable for differentiating MM from other pigmented tumors. MM is characterized by a positive reaction to HMB-45, vimentin, and S-100 proteins12 and a negative reaction to EMA and Leu-7.7,20 Meningioma cells lack the expressions of HMB-45 and S-100, and a positive reaction to EMA differentiates pigmented meningioma from MM.7,12 The absence of a reaction to Leu-7 excludes the diagnosis of melanocytic schwannoma.7,28 Vimentin usually appears in MM but is only rarely present in malignant melanoma. Ultrastructurally, desmosomes are characteristic of meningioma but are generally absent in MM.3,5,19 However, a differential diagnosis between MM and malignant leptomeningeal melanoma may present some difficulties.20 Because the histologic characteristics of MM and melanoma are overlapping, thorough physical and radiologic examinations of the patient are required to exclude a primary cutaneous, ocular, or mucosal melanoma.5,10
Conclusions
We report 5 rare cases of intraspinal MM. To date, to our knowledge, this is the largest collection of intraspinal MM cases with MR imaging. MR imaging plays an important role in the detection and diagnosis of intraspinal MM. The different amounts of melanin-containing cells or hemorrhage may influence the MR imaging signal intensity of tumor cells. Final diagnosis should be based on histopathology and IHC examinations.
Acknowledgments
The authors thank Mou Ren-Qi for his assistance with the study.
Footnotes
The first 2 authors contributed equally to this work.
References
- 1.
- 2.
- 3.
- 4.
- 5.
- 6.
- 7.
- 8.
- 9.
- 10.
- 11.
- 12.
- 13.
- 14.
- 15.
- 16.
- 17.
- 18.
- 19.
- 20.
- 21.
- 22.
- 23.
- 24.
- 25.
- 26.
- 27.
- 28.
- 29.
- 30.
- Received September 16, 2011.
- Accepted after revision November 18, 2011.
- © 2012 by American Journal of Neuroradiology