Abstract
Summary: Cutaneous neuroendocrine carcinoma, first described in 1972, is an aggressive disease usually occurring in sun-exposed skin. Other sites have been described, however; such tumors occasionally occur within the nasal fossa. A high rate of metastasis (>30%) explains the poor prognosis. Descriptions of the imaging features of these tumors, mainly located in cutaneous region, are rare. We therefore present the imaging features of two cases of Merkel cell carcinoma involving the sinonasal region, suggestive of a hypervascular tumor.
Merkel cell carcinoma (MCC) was first described in 1972 by Toker, who identified five cases of dermal trabecular carcinoma (1). MCC is a highly aggressive neoplasm of neuroendocrine origin, with a predilection for the head and neck regions in elderly patients. It has marked propensities for local and distant metastasis and recurrence (2). Surgical therapy is indicated for the primary lesion, with or without postoperative radiation therapy.
Case Reports
Case 1
A 92-year-old woman presented with a 10-month history of right nasal obstruction, rhinorrhea, and reduced visual acuity of the right eye. Her medical history was negative except for tachycardia. Physical examination showed proptosis of the right eye. Abdominal sonography was normal.
Native CT of the head and neck showed a large, homogeneous soft-tissue mass of the right nasal fossa without calcifications (Fig 1A and B) involving the adjacent paranasal sinuses and destroying the nasal septum, medial wall of the orbit, and roof of the ethmoid.
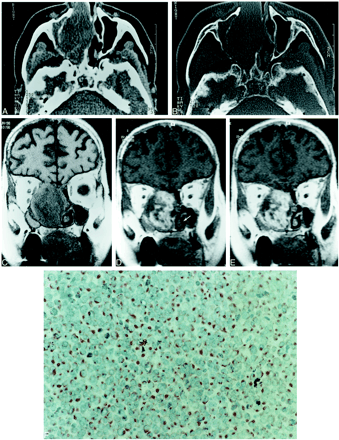
Case 1.
A, Axial, unenhanced CT scan through the inferior turbinate shows a large, homogeneous soft-tissue mass in the right nasal fossa.
B, Bone-window image (same level as A) shows aggressive destruction of the nasal septum and intersinonasal wall.
C, Coronal T1-weighted imaging with parameters 500/14/4 (TR/TE/excitations) shows the large tumor of intermediate signal intensity with extension into the skull base. Note the retention of mucosal secretion within the right maxillary sinus, of discrete higher signal intensity (arrowhead).
D and E, Dynamic T1-weighted imaging with parameters 500/14/4 (TR/TE/excitations) shows early enhancement (D) of the tumor 37 seconds after contrast medium administration (white arrow). Note that the mucosa overlying the inferior turbinate is not yet enhanced. At 75 seconds after contrast medium administration (E), the tumor is more enhanced. Notice the linear enhancement of the dura mater (arrowhead).
F, Immunostaining for keratin. Positivity has a distinct perinuclear dot-like quality, a characteristic feature of Merkel-cell carcinoma.
On MR imaging, the large soft-tissue mass was of intermediate signal intensity on T1-weighted (Fig 1C) and T2-weighted imaging. Dynamic T1-weighted imaging after contrast medium administration showed maximal enhancement within the first 30 seconds (Fig 1D and E).
The mass was very aggressive, in close relationship to the right olfactory nerve. We noted upper extension into the skull base. The dura mater showed linear enhancement. Laterally, the lamina papiracea was destroyed with extraconal fat involvement; however, the signal and shape of the right medial rectus muscle were unremarkable.
Anterior rhinoscopy showed a large reddish mass within the nasal fossa, filling the nasal vestibule. Posterior rhinoscopy revealed filling of the right choana.
Biopsies done under local anesthesia established the diagnosis of MCC on the basis of the following histologic findings: very rich tumoral vascularization and a uniform population of cells with round-to-oval nuclei, inconspicuous nucleoli, evenly dispersed chromatin, ill-defined cytoplasm, abundant mitotic activity with marked cytoplasm activity for neurospecific-enolase (Fig 1F), negative staining for S-100 protein, and positive markers for cytokeratin.
The patient and her family declined any kind of treatment.
Case 2
A 95-year-old woman presented with nasal obstruction and progressive right-sided loss of vision. Physical examination showed blindness in the right eye and right-sided infraorbital hypoesthesia.
The CT scans revealed a voluminous homogeneous mass without calcifications, expanding the right nasal fossa and destroying the nasal septum and the lamina papiracea (Fig 2A). We noted extension into the superior orbital fissure.
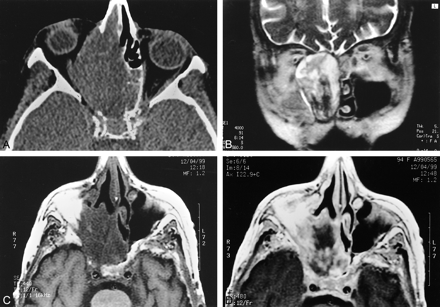
Case 2.
A, Axial unenhanced CT scan image shows a large homogeneous mass without calcifications destroying the lamina papiracea and the nasal septum.
B, Coronal T2-weighted imaging with parameters 4000/91/3 (TR/TE/excitations) shows stretching of the right optical nerve by the tumor, which shows intermediate signal intensity with cystic areas.
C and D, Six months after chemo- and radiotherapy, T1-weighted images before and after contrast material administration (parameters 480/12/2 [TR/TE/excitations]) show large necrotic intratumoral areas.
MR imaging showed a large expansive mass within the right nasal fossa, of intermediate signal intensity on T1- and T2-weighted images (Fig 2B) and enhanced after contrast medium administration. The tumor had invaded the right pterygopalatine fossa and the orbit, with wide involvement of the extraconal fat. The right optical nerve was stretched with dilatation of its arachnoid sheath.
Anterior rhinoscopy showed a large reddish mass filling the right nasal fossa. Histologic findings after biopsies were similar to those of the first case.
The patient underwent radio- and chemotherapy. The MR images at 6-month follow-up showed a dramatic decrease in tumor size with large cystic necrotic areas (Fig 2C and D).
Discussion
MCC is a rare tumor first described by Toker in 1972 as a trabecular carcinoma of the skin (1). This aggressive tumor arises from Merkel cells in the basal layer of the dermis. The origin of the Merkel cells is controversial; there is evidence supporting their origin both from the neural crest as well as from transitional cells in the basal layer of the epidermis (2).
With electron microscopy, Tang and Toker (3) identified membrane-bound, neurosecretory, dense-cored granules in the malignant cells. Because the only mature cell in the skin containing these granules is a touch receptor known as the Merkel cell, the name of MCC was given to the disease. They also noted the presence of neuron-specific enolase (NSE), a marker for cells of neuroendocrine system. Gu et al (4) suggested that NSE immunostaining is a reliable, simple, and highly specific method for identification of Merkel cells.
MCC mainly affects older patients, with a mean age of 70 years and a slight predominance among women. The most common primary sites are located in the head and neck, especially the cheek and eyelids (5). The extremities, trunk, and mucous membranes also can be involved.
Although the majority of the tumors are found on sun-exposed sites, there have been reports of MCC involving the nasal cavity (6) and palatal mucosa (7). One recent report described a case of MCC of the external auditory canal invading the intracranial compartment in an 86-year-old woman (8). The tumor showed a homogeneous, intermediate signal on T1- and T2-weighted imaging and enhanced massively and homogeneously after contrast medium administration. To our knowledge, there have been no published imaging reports of MCC involving the nasal fossa.
Long-term prognosis for patients with MCC is poor. Pergolizzi et al (9) have reported the estimated 1-, 2-, and 3-year survival rates to be 88%, 72%, and 55%, respectively, and an estimated 25% to 35% of the patients ultimately will die of the disease. The local or regional recurrence rate is >30%, often occurring within the first year (6). Regional-node metastases, reported in 40% to 80% of the cases, usually are present at initial presentation. MCC metastasizes widely; distant metastases occur with a reported frequency from 24% to 36% within 2 years after diagnosis (10). Sites include the regional lymph nodes, liver, bone, brain, lung, and skin (11).
Gollub et al (12) reported that lymph node metastasis was the most common manifestation of regional disease. They showed a high attenuation on contrast-enhanced CT. Liver metastases had a target-sign appearance or showed uniformly low attenuation. Skin lesions appeared as cutaneous rounded nodules, isoattenuated to slightly hyperattenuated relative to muscle.
To minimize the chance of local recurrence and nodal and distant metastases, MCC should be treated aggressively. The treatment of choice is a wide local excision with a margin of 2.5 to 3 cm, and adjuvant radiotherapy is recommended (9). In our two patients, one refused any kind of treatment, and the second underwent radio- and chemotherapy because her poor heart condition contraindicated surgery.
Malignant sinonasal tumors are rare. The most common are squamous cell carcinomas, adenocarcinomas, adenoid cystic carcinomas, lymphomas, and melanomas (13). These tumors are of intermediate signal on T2-weighted images and enhance variably after contrast medium administration.
Squamous cell carcinoma is the most frequent epithelial tumor of the nose and paranasal sinuses. The majority of patients are male and over 65 years of age. On CT and MR imaging, bone destruction of the sinus walls may be shown. The tumor itself may show areas of punctate calcifications, and it is variably enhanced after intravenous contrast medium administration.
Adenocarcinoma of the nasal fossa is a relatively rare condition. Age distribution shows a peak occurrence in the sixth and seventh decades with a marked predominance of men. This type of carcinoma most often enhances with intravenous contrast medium administration, and calcifications may occur. Some patients show a sclerotic reaction of the walls of affected sinuses. There is moderately high signal intensity on T2-weighted imaging.
Adenoid cystic carcinoma, in contrast, has its peak distribution in the fourth decade, with a slight female preponderance. The most frequent spread of this neoplasm is perineural, which may appear on CT and MR imaging as obliteration and enlargement of nerve sheaths.
Although the nasopharyngeal region is a potential site of lymphoma, actual involvement of the nose and paranasal sinuses is rare (14). Lymphomas arising in the nose or paranasal sinuses are of the non-Hodgkin's type. On CT and MR studies, they can mimic the much more common entities of sinusitis, polyposis, granulomatous processes, and benign and malignant neoplasms. They often are seen as bulky masses of soft-tissue density with no calcifications, with possible expansion, erosion, or infiltration on CT imaging. On MR imaging, the signal is of moderate intensity on T2-weighted images and is not enhanced after contrast agent administration.
Patients with malignant melanoma of the nasal or paranasal sinuses have a poor prognosis (14). Less than 1% of all melanomas arise in the nasal cavity or adjacent sinuses; most lesions originate in the maxillary sinus. On CT imaging, the nasal mass shows no characteristic attenuation features, and associated bone destruction often is present that involves the maxillary antrum, the ethmoids, or the cribriform plate area. On MR studies, it appears as a mass of high signal intensity on T1-weighted images and low signal intensity on T2-weighted images and can be moderately to markedly enhanced after contrast agent administration.
Hypervascular masses of paranasal sinuses also are rare. Their diagnosis is based on the following imaging features: dilatation of the feeding vessels (eg, the maxillary artery in the pterygopalatine fossa), hypertrophy of peritumoral vessels, intratumoral flow voids, and early and intense enhancement on dynamic postcontrast T1-weighted images. The differential diagnosis should include metastatic carcinoma from primary tumors of genitourinary system, because there is a strong predominance of renal cell carcinoma (80%), neuroendocrine carcinoma, paraganglioma, ectopic meningioma, and olfactory neuroblastoma. Olfactory neuroblastoma is the most common neuroectodermal tumor of the nasal fossa, presenting with a bimodal incidence of one peak after 50 years. A high location in the nasal fossa, the presence of punctate intratumoral calcifications and cystic areas, and intracranial extension through the cribriform plate are the main suggestive features of this tumor, which may have a relatively intermediate to high signal intensity on T2-weighted imaging and show strong enhancement after contrast agent administration.
Differentiation from metastatic renal cell carcinoma and other neuroendocrine carcinomas is based on biopsy results and specific immunostaining. If Merkel cell tumor is under discussion, pretherapeutic tumor staging should include the regional and retropharyngeal nodes, because their involvement at initial presentation is thought to be high in reference to other sites of Merkel tumor. Conversely, a tumor of the nasal fossa should be checked in patients referred for epithelial lymph node metastasis of an unknown primary tumor to the retropharyngeal nodes, high jugulocarotid nodes, or submandibular gland nodes.
In patients over 70 years who present with a hypervascular mass of the nasal fossa, MCC should be included in the differential diagnosis. Abundant mitotic activity and NSE positivity are the main points to establish the correct histologic diagnosis. Tumor location, the age of the patient, and a lack of tumoral calcifications can suggest the diagnosis. When MCC is considered, a careful check-up that includes imaging of the regional lymph nodes, lung, liver, and brain should be performed preoperatively.
Footnotes
1 This work was presented in part at the ASHNR meeting, Washington, DC, May 4–7, 2000.
2 Address reprint requests to Louisa Azizi, MD, Service de Radiologie, Hopital Saint Antoine, 184 Rue du Faubourg Saint Antoine, 75012 Paris, France.
References
- Received August 17, 2000.
- Accepted after revision January 2, 2001.
- Copyright © American Society of Neuroradiology