Abstract
Summary: We present the angiographic and MR imaging course of a 62-year-old man with a right parietal high-flow arteriovenous malformation (AVM), which was diagnosed because of seizures. A spontaneous, complete, and asymptomatic occlusion of the AVM was confirmed by a second angiography 3 months later. The possible mechanisms leading to the occlusion are discussed, and a brief review of the literature is given.
Arteriovenous malformations (AVM) are a rare cause of neurologic symptoms. The incidence is estimated at about 0.02–0.05% of the population (1). The AVM can be localized in any part of the central nervous system. Many different systems of classification have been proposed (2).
Apart from seizures and focal neurologic deficits, bleeding is the most frequent complication of AVM. The risk of hemorrhage depends on localization and previous bleeding and is estimated to be about 2–4% per year (3, 4) with a 30% risk of death. Therapeutic options comprise surgery for superficially located AVMs, radation therapy, and endovascular procedures, but all of these treatment options have an inherent risk of complications.
Unfortunately, a spontaneous occlusion—complete or incomplete—seems to occur in only 1–3% of all patients with a cerebral AVM (5). In 70% of such cases, the cerebral AVM occludes after symptomatic intracerebral or subarachnoid hemorrhage (5–7). Several possible pathologic mechanisms are still being discussed (5), with thrombosis of the nidus of the AVM (8–10) as the most important factor.
We describe the angiographic and MR imaging follow-up examinations of a man with a right parietal, high-flow AVM. A spontaneous, complete, and asymptomatic occlusion of the AVM was revealed by a second angiography.
Case Report
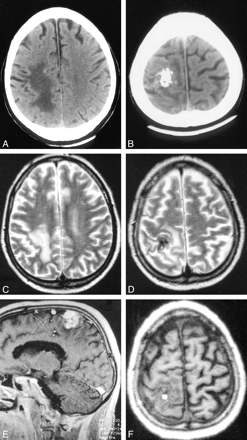
The 62-year-old otherwise healthy man suffered from focal epilepsy with an initial left-sided sensory aura since 1995. Imaging workup with cranial CT or MR imaging was not performed before January 2000. At that time, the patient suffered a status epilepticus with a postseizure paresis of the left arm and leg. Cranial CT revealed a strongly calcified lesion in the parietal lobe with hypoattenuating white matter (Fig 1A and B). The MR images showed an AVM with diffuse hyperintensities in the surrounding white matter in T2-weighted scans, but no signs of an acute or old hemorrhage. These white matter lesions were thought to represent gliosis (Fig 1C and D). Delayed T1-weighted images, obtained 20 minutes after injection of chelated gadolinium, showed a breakdown of the blood-brain barrier surrounding the AVM nidus (Fig 1E).
Cranial CT and MR imaging performed at presentation and 3 months.
A and B, Initial cranial CT scan shows a lobulated, calcified, 1.5-cm lesion in the central region with surrounding hypoattenuating white matter.
C and D, In axial T2-weighted images (4800/85 TR/TE), the lesion shows a hypointense signal because of calcifications, while the white matter changes appear hyperintense.
E, Sagittal reconstruction of T1-weighted fast low-angle shot 3D images (16.1/7 TR/TE) 20 minutes after chelated gadolinium injection reveals a slight contrast enhancement at the AVM nidus.
F, Three months later, one of the main draining veins appears hyperintense in this nonenhanced T1-weighted images, probably because of thrombus.
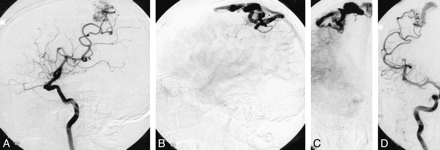
Arterial digital subtraction angiography showed a right central AVM with a nidus size of 2 cm, which was supplied mainly by two arterial feeders from the right middle cerebral artery and also by small feeders from the right anterior cerebral artery. The AVM was drained by two ascending cortical veins into the superior sagittal sinus (Spetzler grade II: eloquence 1, venous drainage 0, size 1) (Fig 2).
Digital subtraction angiograms of the right interior cerebral artery. Lateral views (A, B) and anteroposterior views (C, D) in early arterial and late parenchymal phases show the high-flow central AVM, with two main feeders from the middle cerebral artery and venous drainage via superficial cortical veins into the superior sagittal sinus
Despite high-dose anticonvulsive medications (carbamazepine 900 mg and valproic acid 2800 mg daily), the seizures continued. Treatment of the AVM was deemed necessary to reduce the risk of a bleeding complication. Because of the location in the central region, stereotactic radiation was preferred to surgery or embolization. Blood coagulation tests were normal.
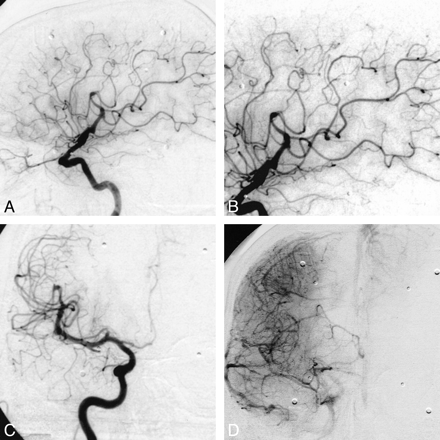
A second digital subtraction angiogram was obtained in May 2000 to plan the stereotactic radiation procedure (Fig 3). Surprisingly, we found a complete occlusion of the AVM with a pronounced caliber reduction in the former feeders. The extracranial vessels were elongated; however, we found no predisposing source of cerebral emboli, such as a focal stenosis or a flow-related aneurysm. Because of the change in cerebral hemodynamics, the hypoplastic A1 segment of the right anterior cerebral artery was now supplied by anterograde vessels. On MR and follow-up cranial CT images, there were no signs of a recent or old intracerebral or subarachnoid hemorrhage and, especially, no progress of the white matter changes around and below the AVM nidus. However, the thrombosis of the main draining vein (Fig 1F) and the normal flow of the superior sagittal sinus could be shown with MR imaging. The exact time of AVM obliteration remains unclear, because no headache or progression of the neurologic symptoms indicated the thrombosis.
Follow-up digital subtraction angiograms of the right interior cerebral artery. Lateral views (A, B) and anteroposterior views (C, D) 4 months later fail to show the AVM nidus. Note the normal lumens of the former feeders
Discussion
Spontaneous obliterations of cerebral AVM are rare; only 50 cases have been reported in the literature. If only adult, neurologically controlled patients are considered, in whom an angiography was performed before and after spontaneous occlusion, this number is reduced to 29, including the patient presented here.
Of these 29 patients, 20 became symptomatic with a primary intracerebral (n = 18) or subarachnoid (n = 2) hemorrhage (9–19). In the remaining nine patients (Table), seizures (n = 4) or headache (n = 4) were the leading clinical symptoms (5–7, 20–22); in one patient, the cerebral AVM was an incidental finding (23). The interval between the initial angiographic diagnosis and the documented occlusion of the AVM ranged from 3 months (5) to 21 years (20). Furthermore, one of the nine patients had a secondary intracerebral hemorrhage related to the AVM, just before the documented occlusion of the AVM (antecedent bleeding, Patient 5) (6).
Patients with spontaneous, complete obliteration of an AVM without initial hemorrhage
Many factors predisposing to a spontaneous AVM obliteration have been discussed, including the arterial feeders as well as the venous draining veins (5). Most often, the thrombosis of the AVM nidus has occurred secondary to an intracerebral or subarachnoid hemorrhage. Thus it seems more likely that the hematoma, with its subsequent mass effects, proximally compresses the draining veins near the nidus rather than the arterial feeders.
In some patients, the nonfilling of the lesions may be explained by compression of the veins from bleeding or swelling of the brain after head trauma or neurosurgical manipulations. In the case of a preceding subarachnoid hemorrhage, vasospasms of the feeders may reduce blood flow within the AVM to a critical value and induce a spontaneous thrombosis. In our patient, we can exclude such mechanisms because of the lack of clinical, cranial CT, and MR imaging signs of subarachnoid hemorrhage during follow-up.
Analysis of the 29 reported patients shows that several factors appear to be associated with spontaneous occlusion of cerebral AVM. In cases with detailed descriptions of the venous drainage (n = 19), a single draining vein was found in 16 patients. Taking only the nine patients without an initial intracranial hemorrhage leading to the diagnosis, four of six cases showed a single draining vein (Table). Spontaneous thrombosis of the draining veins related to a hypercoaguable state is described in children (24) and in women taking high-dose oral contraceptives. After intravenous injection of an x-ray contrast agent, the flow in the veins may be reduced with a subsequent increase in the risk of thrombosis. On the other hand, contrast agent has been reported to have an anticoagulant effect, and there have been no reports of an elevated risk of venous thrombosis after intraarterial injection of contrast agents.
Patients with spontaneous obliteration of an AVM have a mean age of 40 years. Most of the AVMs are located in the parietal lobe (16 of 29). Less frequently, they are found in the frontal lobe (n = 5), in the basal ganglia and thalamus (n = 3), in the occipital lobe (n = 3) or in the temporal lobe, perisylvian cortex, or cerebellum (all n = 1). No difference was found concerning the right or left cerebral hemisphere.
Occlusion of the feeding arteries may be caused by cardiac or arterial emboli. In rare cases, these arterio-arterial emboli result from flow-related aneurysms of the feeders (25). In our patient, we found elongation and kinking of the vessels, but no stenosis or aneurysm. Patients with a single arterial feeder may be more predisposed to complete embolic occlusion of an AVM. However, of the 29 patients with a spontaneous AVM occlusion, a solitary feeder was reported only in 30% (six of 20), implying that the draining veins may be more important.
Histologic and experimental investigations show that turbulent flow within the feeders may induce occlusion of the vessel. Histopathologic examinations have revealed focal collections of interstitial connective tissue and smooth muscle cells (called “fibromuscular cushions”), which occur in the arterial components and only occasionally in the arterialized draining vein of an AVM (26). These fibromuscular cushions may severely narrow the lumen of the vessel, resulting in occlusion. Experiments in rabbits have shown that creation of high-flow arteriovenous fistulas leads to similar changes in the otherwise healthy arterial wall (27). The high-flow angiopathy starts with endothelial damage and destruction, followed by platelet adherence. Spontaneous reendothelialization leads to irregularities of the lumen. Moreover, platelets adhere to the exposed subendothelial matrix and release factors, such as thromboxane and prostaglandin, that cause platelet aggregation, activating a clotting cascade and eventually forming a stable platelet or thrombus (28). However, such hemodynamically induced vessel alterations are found mainly in large AVMs that have turbulent flow because of the large shunt volumes. In contrast, most of the 29 reported spontaneous AVM obliterations revealed a smaller nidus. The nidus was smaller than 3 cm in 14 of 29 cases and between 3 and 6 cm in 11 patients. Only two patients had an AVM > 6 cm (in two cases, the nidus size was not reported). Therefore, these findings also may support the importance of pathologic processes in the draining veins.
Histologic analysis of the nidus 4 weeks after diagnosis of an obliterated AVM (5) showed some patent vascular channels. Expression of vascular endothelial growth factor and its receptors, Flt-1 and Flk, was shown within the vascular endothelium and the subendothelial layers. This may reflect an ongoing process of thrombosis rather than a single, final occurrence. These findings also may reflect an active angiogenic process, with possible neovascularization occurring within the lesion in association with thrombosis. However, only one case of recanalization after complete thrombosis has been reported in the literature (29). Nevertheless, in patients with spontaneous occlusion of an AVM, an angiographic follow-up is indicated to be certain that recanalization has not occurred.
Footnotes
1 Address reprint requests to Hilmar Krapf, MD, Department of Neuroradiology, University of Tübingen, Hoppe-Seyler-Str. 3, D- 72076 Tübingen, Germany.
References
- Received January 24, 2001.
- Copyright © American Society of Neuroradiology