Abstract
BACKGOUND AND PURPOSE: Stent-protected carotid angioplasty (SPAC) is an option for treating cervical symptomatic high-grade internal carotid artery (ICA) stenosis. So far, knowledge about hemodynamic changes in the early postinterventional phase is limited. The purpose of this study was to evaluate these changes.
METHODS: Thirty-four consecutive patients with a high-grade ICA stenosis (according to European Carotid Surgery Trial criteria) and 10 healthy volunteers were enrolled. Hemodynamics of the cerebral circulation were assessed before and within 6 hours after SPAC. ICA flow volume, cerebral blood volume flow (CBVF), and collateral flow volume were sonographically assessed. The ratio of flow velocities in the middle cerebral artery (MCA) ipsilateral to the ICA stenosis was calculated and compared with that in the contralateral MCA. This ratio was designated rMCA.
RESULTS: Preinterventional CBVF, ICA flow volume, and rMCA were significantly reduced compared with results in healthy volunteers. After SPAC, CBVF, ICA flow volume, and rMCA increased significantly. The rMCA did not exceed 1.0. Collateral flow volume decreased in patients with posterior collateral flow only. Postinterventional CBVF and ICA flow volume in patients did not differ from values in healthy volunteers.
CONCLUSION: Cerebral hemodynamics appear to be impaired in patients with symptomatic high-grade ICA stenosis. After SPAC, hemodynamic parameters normalize within 6 hours. We did not detect hyperperfusion. However, flow volume in the contralateral ICA remains increased in patients with former anterior cross-filling.
Patients with ischemic symptoms due to high-grade carotid stenosis benefit from carotid obliteration therapy (1, 2). Carotid thrombendarterectomy (CTEA) is still the criterion standard for the treatment of atherosclerotic carotid stenosis. However, during the last decade, stent-protected carotid angioplasty (SPAC) has become an alternative therapeutic option. Several controlled multicenter trials are currently being performed to compare both methods (3–5).
Besides ischemic symptoms during CTEA and SPAC, reperfusion damage is a rare but feared complication of carotid obliteration. Little is known about the hemodynamic changes that occur during the early phase after CTEA, and especially after SPAC, and about the pathogenic mechanisms leading to hyperperfusion syndrome (4, 6, 7).
Cerebral hemodynamics can be evaluated by sonographically assessing intracranial blood flow velocities and cerebral blood volume flow (CBVF). CBVF reliably measures global cerebral perfusion and is correlated with cerebral blood flow (CBF), as measured by means of positron emission tomography (PET) and single photon emission CT (SPECT), respectively (8).
The aim of the present study was to assess hemodynamic changes in the cerebral circulation in the early postinterventional phase after SPAC. These changes may suggest or help identify hyperperfusion as the underlying pathogenic mechanism of reperfusion damage in obliteration of a carotid artery stenosis.
Methods
Inclusion and Exclusion Criteria
We enrolled patients with a symptomatic internal carotid artery (ICA) stenosis of high grade according to European Carotid Surgery Trial (ECST) criteria (2). Patients with high-grade carotid artery stenosis and no recent ischemic event but insufficient collateral flow were also included; examples included those with critically reduced flow in the middle cerebral artery (MCA) and reduced vasomotor reactivity. Patients had to be eligible for interventional therapy, which was the only other inclusion criterion. Carotid artery stenosis was detected by means of extracranial duplex sonography with established criteria (9) and reproduced by means of either MR angiography or digital subtraction angiography. All stenoses were due to atherosclerotic carotid artery disease.
We excluded patients with clinically evident cardiac output failure (worse than New York Heart Association grade IIb), those with occlusion of the target vessel, and those with stenosis due to an acute appositional thrombus if they were not candidates for interventional therapy or if they refused participation. Patients were also excluded from the study if the time to postinterventional follow-up sonography exceeded 6 hours.
Diagnostic Workup
All patients underwent routine extracranial and transcranial Doppler and duplex sonography, clinical examination, medical history workup, and cerebral CT or MR imaging. Ischemic symptoms were classified by using the National Institutes of Health Stroke Scale at the onset of symptoms and after the procedure. We noted demographic data including the patients’ age, sex, vascular risk factors, concomitant vascular disease, and type of ischemia (eg, infarct type or transient symptoms). Collateral pathways were sonographically assessed and recorded before interventional therapy. After intervention, they were compared with flow patterns in the circle of Willis.
To evaluate ipsilateral impairment of MCA blood flow velocity, we calculated a ratio of the ipsilateral peak flow velocity to the contralateral peak flow velocity. Ratios before and after SPAC were compared.
Sonography was performed by using a unit (Elegra; Siemens, Erlangen, Germany) with a 7.5-MHz probe for extracranial sonography and a 2.5-MHz probe for transcranial sonography.
Percutaneous SPAC
Patients were considered for angioplasty according to ECST criteria. Additionally, patients with restenosis after CTEA were referred for carotid angioplasty. For 3 days before SPAC and 3 days afterward, all patients began an anticoagulant and an antiplatelet regimen consisting of aspirin 100 mg/day, clopidogrel 75 mg/day, and enoxaparin 2 × 50 IU/kg/day. During the procedure, patients received unfractionated heparin intravenously to increase their activated clotting time to more than 250 seconds. Patients also received low-dose benzodiazepine for sedation shortly before interventional therapy; this sedation was adjusted during the procedure if necessary. SPAC was performed according to published procedure (10). After a 7F introducer catheter was placed in the common carotid artery, a guidewire was navigated through the stenosis into the poststenotic ICA. Endovascular treatment was primarily performed by using a self-expandable stent (carotid Wallstent, 7.0–30 mm; Boston Scientific, Natick, MA). A balloon catheter was then placed to treat the residual stenosis and inflated with 7–12 bar of pressure. SPAC was performed without predilation and without the use of distal protection devices. Treatment was considered successful if the stenotic segment was 20% or less after balloon dilation. In all patients, the intracranial circulation was visualized after treatment to detect embolic vascular occlusion.
Cerebral Blood Volume Flow
Cerebral hemodynamics were monitored by measuring the CBVF before and within 6 hours after stent placement in the carotid artery. CBVF was the sum of flow volumes within both ICAs and vertebral arteries. The flow volume of an artery was calculated by multiplying the mean time-averaged velocity with the area of the vessel by using the following published equation (8): FV = TAVm × (d/2)2 × π, where FV is the flow volume, TAVm is the mean time-averaged velocity, and d is the diameter of the vessel.
Flow volumes were measured, with attention to published limitations with the method (8). Preinterventional flow volume of the stenotic ICA was assessed as far distal to the stenotic lesion as possible so that laminar flow was present. Flow velocity was corrected for the angulation of the sonographic beam. Vessel diameter was measured exactly 90° relative to the vessel wall; values were obtained three times during systole by using the average to calculate flow volume. Vessel diameter and time-averaged mean velocity were measured in the same part of the vessel. Flow volumes of the vertebral arteries were assessed in the V2 segments.
To estimate the input of collateral flow in patients with collateral flow via the anterior communicating artery, flow volumes of both ICAs were measured in 10 healthy volunteers. In patients with collateral flow via the posterior communicating artery, flow volumes of the vertebral arteries were added to a total vertebrobasilar flow volume and compared with values in healthy volunteers. As a control, CBVF was also measured in 10 healthy volunteers with no signs of atherosclerotic artery disease or cardiac disease. Patients and healthy volunteers were matched in terms of age and sex.
Statistical Analysis
Statistical analysis included a comparison of preinterventional and postinterventional CBVF and ICA flow volume in patients with values in healthy volunteers, as well as a comparison of preinterventional and postinterventional CBVF and ratio of flow velocity in the MCA ipsilateral to the ICA stenosis to the flow velocity in the contralateral MCA (rMCA). Subanalysis was performed to compare ipsilateral ICA flow volume and flow volume in the collateral artery before and after intervention. Differences in CBVF, ICA flow volume, vertebrobasilar flow volume, and rMCA between groups were analyzed by using the t test for normally spread, paired variables and the Mann-Whitney U test for non-normally spread, paired variables (SPSS statistical software; SPSS, Chicago, IL).
Results
We enrolled 34 patients with mean carotid stenosis of 87% ± 7.69. Table 1 shows their demographic data. Three patients did not have a history of temporary or permanent cerebral ischemia but were enrolled, because they had a high-grade carotid stenosis and insufficient collateral flow. Among the 34 patients, 18 had a transient ischemic event, and 13 had a cerebral infarction due to carotid stenosis. All patients underwent a routine vascular workup, which revealed 19 concomitant vascular lesions; these were hemodynamically relevant in 16 patients. Four patients were referred because of restenosis after CTEA. Before interventional therapy, intracranial collateral pathways attributed to the symptomatic stenosis were detected in 24 of the 34 patients. An ophthalmic collateral pathway was present in nine patients (Table 2).
Patient demographic data, periprocedural symptoms, and procedural success
Collateral pathways
Thirty SPAC procedures were performed without peri-interventional sequelae. Three patients had a minor transient ischemic attack with resolved symptoms at the time of postinterventional investigation. One of the 34 patients developed persisting neurologic sequelae attributed to SPAC, including mild dysarthria and moderate brachiofacial hemiparesis (Table 1).
In two patients, SPAC was not successful, with residual stenosis of 60% in one case and a high-grade stenosis due to distal carotid dissection after SPAC in another (Table 1). To rule out the hemodynamic influence of residual stenosis and stroke, we excluded these two patients and also the patient with persisting neurologic sequelae from hemodynamic analysis.
Compared with healthy volunteers (Table 3), patients with carotid stenosis had significantly lower CBVF. Volume flow of the ipsilateral stenotic carotid artery was reduced. In patients with collateral flow via the anterior communicating artery, volume flow of the contralateral carotid artery was significantly increased. In patients with posterior collateral flow, total vertebrobasilar volume flow was increased. Table 3 shows the hemodynamic data and levels of significance.
Hemodynamic data in volunteers and patients before SPAC
Postinterventional measurements were performed 2.52 hours ± 1.43 after SPAC. At that time, only one patient had collateral flow attributable to the stented carotid artery. This patient had a postinterventional high-grade stenosis due to dissection of the distal carotid artery and was excluded from the hemodynamic analysis (Tables 1 and 2).
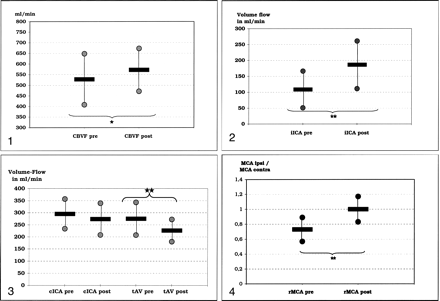
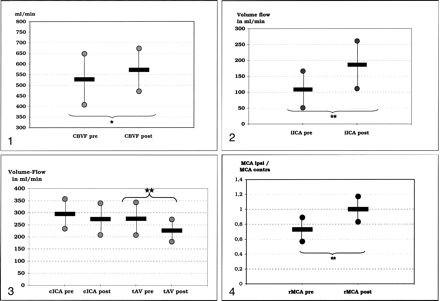
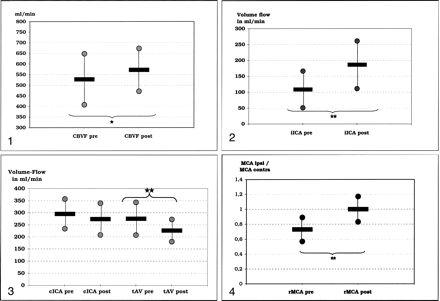
CBVF increased slightly but significantly after intervention (Fig 1). Despite this minor change in CBVF, ipsilateral ICA flow volume was significantly increased (Fig 2). Contralateral ICA flow volume decreased in patients with preinterventional anterior cross-flow, but the results were not significant (Fig 3). Therefore, contralateral ICA flow volume remained increased in comparison with values in healthy volunteers (Table 4). Total vertebrobasilar flow volume in patients with posterior collateral flow decreased significantly (Fig 3).
CBVF before (CBVF pre) and after (CBVF post) interventional obliteration of the ICA. Star indicates P < .05.
Volume flow in the ipsilateral ICA before (iICA pre) and after (iICA post) SPAC. Stars indicate P < .01.
Volume flow of the contralateral carotid artery in patients with anterior cross-filling before (cICA pre) and after (cICA post) SPAC and volume flow of the vertebral arteries in patients with posterior collateral flow before (tAV pre) and after (tAV post) the procedure. Stars indicate P < .01.
Hemodynamic data in volunteers and patients after SPAC
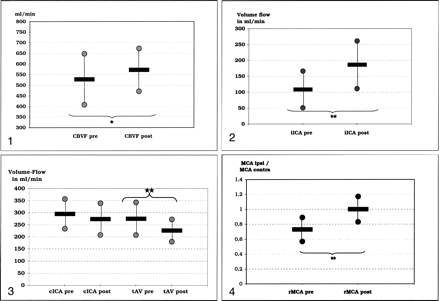
Hemodynamic improvement was also reflected by a restoration of the preinterventionally decreased rMCA. After SPAC, rMCA increased significantly and revealed symmetric MCA flow velocities (Fig 4).
CBVF values and ICA flow volume in patients after SPAC were not different from those in healthy volunteers. However, vertebrobasilar flow volume and contralateral ICA flow volume increased. Table 4 shows hemodynamic data and levels of significance.
Discussion
Cerebral hemodynamics were evaluated by measuring carotid and vertebral blood flow volume and CBF volume, as described elsewhere (8). CBVF has been proved to represent global brain perfusion and reliably demonstrates the same age dependency as CBF values assessed by means of PET and SPECT. When calculated on a standard adult brain weight of 1300 g, CBVF is equivalent to CBF ranging from 45 to 60 mL/100 g/min (11, 12), depending on the age group analyzed (8, 13, 14). In the present study, healthy volunteers had a CBF of 46.7 mL/100 g/min, which represents a normal value. Reliability and validity of the method has been proved by others performing intraobserver and interobserver testing (14, 15). Therefore, we did not reconfirm reliability in this study. Common obstacles with the method (8, 15) were considered. With CBVF measurement, cerebral perfusion can be estimated with a noninvasive, bedside method. It has been used in different clinical settings. The information gained helps us to understand cerebral hemodynamics more accurately, because its correlation with CBF is high (8, 13, 14). Furthermore, volume flow is sensitive to autoregulatory changes under CO2 insufflation, indicating sensitivity of the method for even minor changes in cerebral hemodynamics (16). CBVF describes changes of global cerebral blood supply, whereas changes of local intracerebral perfusion (described with PET and SPECT) might be depicted only indirectly. Furthermore, CBVF is an investigator-dependent method, but it may be a sensitive tool when used by experienced investigators (16).
CBVF measurement was used to evaluate hemodynamic changes before intervention and during the acute postinterventional phase. Although we did not test for vasoreactivity, which is impaired in high-grade carotid stenosis (17, 18), we used a reduction in CBVF, ipsilateral ICA flow volume, and MCA flow velocity (rMCA) as markers of hemodynamic impairment before intervention. Like others (17–21), we found that cerebral hemodynamics were impaired in symptomatic high-grade carotid stenosis. Postinterventionally, both global cerebral perfusion and carotid perfusion increased to normal values without exceeding the parameters found in healthy volunteers. Therefore, we did not detect collateral pathways concerning the stented carotid artery after intervention (22, 23), except in one patient who had postinterventional arterial dissection. Also, we found a significant increase in rMCA immediately after intervention that was not different from values in healthy volunteers; this finding is in accordance with other results (24).
Like CTEA, SPAC poses a risk of hyperperfusion syndrome and stroke (4). However, in a comparative study (Carotid and Vertebral Artery Transluminal Angioplasty Study [4]), intracerebral hemorrhage was lethal in only the endovascular arm. The underlying pathogenic mechanism remained unknown, but the authors speculated that ICH was due to a difference in hemodynamic change, among other causes (4, 5).
In the present study, none of the patients developed a hyperperfusion syndrome, underlining the fact that hyperperfusion syndrome is a rare complication (25, 26), although some groups report a higher incidence in SPAC (27, 28). This complication is most often described during the subacute postinterventional phase (28–30), although in CTEA, a doubling of CBF during surgery identifies patients at high risk (31). At a mean interval of 2.5 hours after recanalization, we did not detect any sign of ipsilateral or global hyperperfusion, which has been encountered in other series (31). However, we observed normalization of hemodynamic parameters, as has been demonstrated in peri-interventional transcranial Doppler monitoring (24). Persistence of reduced vasoreactivity has been described after carotid obliteration (18). This may be the leading pathogenic mechanism (32–34), but we did not test for it in this study. Nevertheless, our findings underline the fact that hyperperfusion syndrome may not be due to increased ipsilateral perfusion (eg, real hyperperfusion).
Our findings had some limitations. First, we did not differentiate between patients with and those without elevated mean arterial pressure. However, because we did not detect elevated volume flow in the ipsilateral ICA or elevated flow velocity in the ipsilateral MCA, we probably would not have found any difference. Also, we did not test for vasoreactivity, which would have helped to highlight preinterventional hemodynamic impairment and postinterventional improvement in cerebral hemodynamics. Finally, we used a single-examination approach with a delay between reopening of the vessel and measurement of volume flow and MCA flow velocity. As a result, the study might have missed a hyperperfusional state occurring immediately after intervention.
Conclusion
We were able to show that measurement of CBVF is applicable in patients with carotid artery disease. We confirmed that global cerebral perfusion, as well as ipsilateral perfusion, is compromised in patients with symptomatic high-grade carotid stenosis, although we did not test for vasoreactivity. Furthermore, we observed normalization of global and ipsilateral cerebral perfusion after intervention, with no sign of hyperperfusion during the acute phase after SPAC. Hyperperfusion syndrome is a rare complication in patients undergoing SPAC that may not be due to real hyperperfusion, but rather, a persistence of impaired vasomotor reactivity of the MCA, which may be vulnerable to arterial pressure changes. To gain further insight, studies are needed to evaluate durability of vasomotor impairment after intervention and also to assess cerebral perfusion during the subacute phase after intervention.
References
- Received August 20, 2003.
- Accepted after revision January 5, 2004.
- Copyright © American Society of Neuroradiology