Abstract
Summary: We describe an approach to measuring cerebral blood flow (CBF) based on independent measurements of cerebral blood volume (CBV) and mean transit time (MTT) with calculation of CBF by using the central volume theorem: CBF = CBV / MTT. This permits optimization of the individual acquisitions and analyses. In particular, measurement of CBV during contrast infusion, rather than simultaneously with MTT from a single bolus, yields values more consistent with those of other methods.
Routine measurement of cerebral perfusion by MR imaging remains an elusive goal with qualitative publications still outnumbering quantitative studies. Our laboratory is exploring alternatives for calculating cerebral blood flow (CBF), applying the central volume theorem, CBF = cerebral blood volume (CBV)/ mean transit time (MTT), with independent measurements of CBV and MTT, which permits individual optimization of the MR imaging acquisitions and contrast schedules. In preliminary studies,1 T2* contrast imaging at 1.5T requires TR ≥ 2500 milliseconds to eliminate T1 inflow effects for optimal CBV but TR ≤ 1500 milliseconds for accurate fitting of bolus concentration–time curves.1,2 These mutually exclusive requirements and other results suggested that measuring CBV by contrast infusion but MTT by using a bolus would be advantageous.3,4 In this study, 3 different strategies of measuring CBF are compared in healthy volunteers to test the hypothesis that optimized independent measurements of CBV and MTT would yield perfusion values more consistent with those from positron-emission tomography (PET), single-photon emission tomography (SPECT), and xenon CT.
Methods
All protocols were approved by our institutional review board. Five healthy subjects (3 women; age range, 21–25 years) were recruited from medical center staff and students.
MR images were acquired on a GE Signa 1.5T MR imaging system by using a standard head RF coil (GE Medical Systems, Milwaukee, Wis). Each session included all 3 MR imaging sequences, a “clinical bolus,” a “quantitative bolus,” and an “infusion” (Table 1). All perfusion scans were acquired as multiple blocks of single-shot echo- planar images with a 22-cm field of view and 128 × 64 acquisition, reconstructed to 128 × 128.4,5 A T1-weighted fluid-attenuated inversion recovery with matching section thickness and orientation provided anatomy. Baseline image sets with at least 10 images were acquired before contrast arrival. The clinical bolus, used by University of Wisconsin Hospital neuroradiologists for many years for patient care, is oriented axially. The quantitative bolus, developed to optimize quantitation of signal intensity change for MTT measurements,1 and the infusion acquisition,4 developed specifically for CBV measurements, were oriented obliquely, with the third section parallel to the inferior surface of the medial frontal lobe, to minimize middle cerebral artery susceptibility artifact and provide a more accurate arterial input function. The total gadolinium dose for the quantitative bolus and infusion pair was 185 μmol/kg, less than a “double-dose” scan, and the total session dose remained within Food and Drug Administration guidelines. Perfusion data were analyzed by using programs written in MatLab (Mathworks, Natick, Mass). Four regions of interest each in cortical gray, deep gray, and white matter, and their associated arteries, were defined by using a semiautomated approach as described elsewhere4,5 to avoid venous contamination common with fully automated arterial input function finding procedures.
Comparison of perfusion imaging protocols used in this study
Signal intensity change was converted to relative contrast concentration, Crel, by using Equation (1): 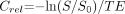 1) where S is signal intensity and S0 is the mean baseline signal intensity. The mean region of interest relative concentration was calculated for each time point from all selected pixels.
1) where S is signal intensity and S0 is the mean baseline signal intensity. The mean region of interest relative concentration was calculated for each time point from all selected pixels.
Details of our CBV methods have been published elsewhere.4 Calculation of CBV by the parametric area under the curve (AUC) method was based on Equation (2): 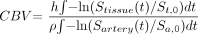 2) where St,0, Sa,0, Stissue(t), and Sartery(t) were tissue or arterial signals at baseline or time t, ρ, the attenuation of brain tissue (1.04 g/mL),6 and h corrects for the hematocrit difference in large vessels and brain microvasculature (0.73).7 Integration involved fitting of the arterial concentration–time curve to a gamma variate function and the tissue concentration–time curve to a 5-parameter lagged normal attenuation function8 after 5-fold nonlinear interpolation (INTERP) to improve the Simplex nonlinear fitting routines.
2) where St,0, Sa,0, Stissue(t), and Sartery(t) were tissue or arterial signals at baseline or time t, ρ, the attenuation of brain tissue (1.04 g/mL),6 and h corrects for the hematocrit difference in large vessels and brain microvasculature (0.73).7 Integration involved fitting of the arterial concentration–time curve to a gamma variate function and the tissue concentration–time curve to a 5-parameter lagged normal attenuation function8 after 5-fold nonlinear interpolation (INTERP) to improve the Simplex nonlinear fitting routines.
Calculation of CBV by the infusion (INF) method was based on Equation (3): 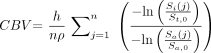 3) where n is the number of points measured within the plateau of the infusion curve and St(j) and Sa(j) are the jth signal intensity measurements during that stable portion in the tissue region of interest and artery, respectively, as described elsewhere.4,5
3) where n is the number of points measured within the plateau of the infusion curve and St(j) and Sa(j) are the jth signal intensity measurements during that stable portion in the tissue region of interest and artery, respectively, as described elsewhere.4,5
All reported MTT values were calculated by the singular value decomposition deconvolution method using the bolus acquisition data.9 Note that, with this method, MTT is obtained directly from the integration of the concentration-time curve normalized by the maximum value of the curve. It is not necessary, or desirable, to extract CBV and CBF separately from the data and then calculate their ratio to obtain MTT by a reversal of the central volume theorem.2
CBF was calculated from the central volume theorem,10 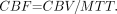 4) Statistical analyses were performed with programs written with MatLab except for repeated measures ANOVA, which was performed by using R.11
4) Statistical analyses were performed with programs written with MatLab except for repeated measures ANOVA, which was performed by using R.11
Results
CBV values from AUC analysis of the clinical bolus are unacceptably large (Table 2). Quantitative bolus acquisitions improved the range of CBV considerably, but the values were still larger than other techniques.4 Only the infusion method yielded CBV values within the expected range. CBV values from the clinical and quantitative bolus AUC were 3.6 and 1.5 times those from infusion, respectively (P = .0015). Infusions also reduced subject to subject variability dramatically (Table 2).
Summary of results
MTT measured from the clinical and quantitative boluses were essentially equivalent (P = .25), though variability was less for the quantitative bolus.
Values of CBF obtained by using clinical bolus data were unreasonably large (Table 2). CBF obtained by using quantitative bolus data were closer to but consistently higher than values obtained by using other techniques. Only results calculated by using infusion CBV and quantitative bolus MTT produced CBF values similar to those available in the literature (P = .0003 for differences among the 3 methods). Mean subject infusion CBV varied from 0.025 to 0.030 mL/g, quantitative bolus MTT from 3.43 to 4.18 seconds, and CBF from 43.8 to 55.5 mL/100 g/min, very close to those obtained with PET, SPECT, and xenon CT.
Discussion
This study demonstrates that it is possible to use dynamic contrast T2*-weighted MR imaging to measure CBV, MTT, and CBF without any normalization procedure. The advantages derive from the separation of CBV and MTT measurements, which permits optimization of each acquisition and analysis independently. Reasonable values of CBF are obtained only when CBV is measured by infusion, supporting earlier reports.2,4 Infusion CBV also improves contrast-to-noise ratio, subject to subject variability and sensitivity to pharmacologic3 or pathologic4 differences among subjects.
The quantitative bolus differs from the clinical bolus by using a thinner section, oblique section angulation, shorter TR and TE, larger flip angle, and lower contrast dosage injected over a slightly shorter time.1 The large CBV values resulting even from the optimized quantitative bolus, however, demonstrate the difficulty of obtaining accurate CBF by using only bolus data. Only infusion CBV provides the basis for CBF estimates that compare with literature values from other methods. The potential explanations for these quantitative differences have been discussed recently.5
It is feasible to design a single acquisition sequence to measure MTT and CBV, but the lack of programmable MR imaging infusion pumps prevents implementation at present. Studies suggest an optimal delay of 7 seconds between bolus and infusion. Furthermore, it should be possible to extract permeability data from the infusion data curve with the appropriate analysis.
References
- Received October 25, 2005.
- Accepted after revision February 22, 2006.
- Copyright © American Society of Neuroradiology

