Abstract
BACKGROUND AND PURPOSE: Brain AVM, cerebral abscess, and ischemic stroke are among the well known neurologic manifestations of hereditary hemorrhagic telangiectasia. However, recently reported data suggest an additional association with malformations of cortical development. The purpose of this study was to determine the prevalence of malformations of cortical development in a population of pediatric patients with hereditary hemorrhagic telangiectasia.
MATERIALS AND METHODS: A retrospective review of brain MRIs from 116 pediatric patients was performed. Each patient was referred from our institution's Hereditary Hemorrhagic Telangiectasia Clinic. Each MRI included a 3D sequence, most frequently MPRAGE. The 3D sequence was evaluated by a neuroradiology fellow, with specific attention to the presence or absence of malformations of cortical development. Positive studies were subsequently reviewed by 2 attending neuroradiologists, who rendered a final diagnosis.
RESULTS: Fourteen of 116 (12.1%) patients were found to have a malformation of cortical development. Among these 14, there were 12 cases of polymicrogyria and 2 cases of bifrontal periventricular nodular heterotopia.
CONCLUSIONS: Pediatric patients with hereditary hemorrhagic telangiectasia have a relatively high prevalence of malformations of cortical development, typically perisylvian polymicrogyria.
ABBREVIATIONS:
- HHT
- hereditary hemorrhagic telangiectasia
- MCD
- malformation of cortical development
- PMG
- polymicrogyria
Hereditary hemorrhagic telangiectasia (HHT) is an autosomal dominant disorder with a geography-dependent prevalence that ranges from approximately 1/5000 to 1/8000 in Japan to 1/10,000 in the United Kingdom.1,2 HHT is diagnosed clinically based on the Curacao criteria, which include: 1) spontaneous and recurrent epistaxis, 2) mucocutaneous telangiectasias, 3) visceral AVMs, and 4) a first-degree relative who has the disease. If 3 or 4 of the criteria are present, a patient is considered to have “definite” HHT. “Possible” HHT requires that 2 criteria be present, whereas patients with fewer than 2 positive criteria are considered “unlikely” to have the disease. HHT results from various mutations in genes involved in signaling in the transcription growth factor–β pathway in vascular endothelial cells. Known mutations include endoglin (ENG), activin receptor-like kinase (ALK1), and SMAD4.3⇓⇓–6 Cerebral manifestations of HHT include brain AVMs, which are estimated to occur in 10%–20% of patients.7 In addition, pulmonary AVMs are present in approximately 30% of patients with HHT. The presence of a right-to-left pulmonary shunt predisposes patients with HHT to cerebral abscess and embolic stroke.8,9
Malformations of cortical development (MCDs) encompass a broad array of disorders seen in patients with developmental delay and epilepsy. Included in this category is polymicrogyria (PMG), a term that refers to cerebral cortex that contains unusually small convolutions.10 MRI features of PMG include small gyri separated by thin, shallow sulci. This often results in the appearance of a thickened cortex with irregularity of the cortical surface and gray-white junction.11 Heterotopia is another MCD, resulting from abnormal neuronal migration and most commonly manifesting in a nodular periventricular form.12 A variety of syndromes and genetic mutations have been shown to be associated with PMG.10,12 Data reported at the 10th International Scientific HHT Conference in 2013 suggested an 8% prevalence of MCDs in the HHT population.13 However, to date, there have been no publications in the scientific literature describing an association between MCDs and HHT. Our study aims to determine the prevalence of PMG and other MCDs in pediatric patients with HHT.
Materials and Methods
We identified 116 pediatric patients—referred to our institution's HHT clinic between 2005 and 2015—who underwent a brain MRI that included at least 1 high-resolution 3D sequence. At our institution, children being evaluated for HHT are routinely screened with brain MRI. In most cases, the 3D sequence was T1 MPRAGE, which is routinely performed at our institution for pediatric brain MRI. All MRI examinations were performed at either 1.5T or 3T. Each MRI was retrospectively evaluated with specific attention to the presence or absence of MCDs by a neuroradiology fellow (G.J.P.). For each case, the radiology report was consulted with attention to documentation of MCDs. Potentially positive cases were then confirmed by 2 board-certified attending neuroradiologists (M.S.G., A.S.), with 2 and 10 years of experience, respectively. For each of the 116 patients, the following information was acquired from the electronic medical record and a pediatric HHT data base: sex, age, diagnosis by Curacao criteria, genetic testing (if available), and the presence or absence of pulmonary and brain AVMs. For each patient who screened positive, the medical record was reviewed to document history of neurologic symptoms with particular attention to the presence or absence of epilepsy. Descriptive statistics were computed, and associations were tested with the Fisher exact test. The criterion for statistical significance was P < .05.
Results
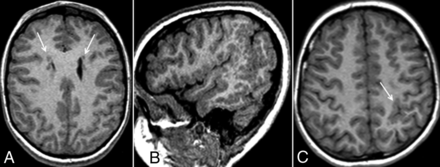
Demographic information, genetic mutation status, and other clinical data for the 116 patients are detailed in Table 1. Because our institution is an HHT Center of Excellence for adults and children, most patients included in our pediatric HHT data base are the children of adults with known HHT, thus accounting for the higher proportion of children meeting Curacao criteria for definite HHT. Fourteen of 116 (12.1%) patients were found to have an MCD (Table 2). The positive studies included 12 cases of PMG (12/116, 10.3%) and 2 cases of bifrontal periventricular nodular heterotopia (2/116, 1.7%). Two sets of siblings were positive for MCD. This included a set of 3 boys, 2 of whom had perisylvian PMG, with the other sibling having periventricular nodular heterotopia (Fig 1). An additional set of 2 sisters each had perisylvian PMG, but in opposite cerebral hemispheres. The siblings of 2 other positive cases were negative for MCD.
Summary of 116 patients
Description of 14 patients with MCD
Three siblings with HHT and malformations of cortical development. Axial T1 MPRAGE demonstrates bilateral periventricular nodular heterotopia (A, arrows) in a 9-year-old boy. Sagittal T1 MPRAGE demonstrates extensive perisylvian PMG (B) in a 12-year-old boy. Axial T1 MPRAGE demonstrates thickened and excessively convoluted cortex in the left posterior frontoparietal region (C, arrow), consistent with PMG.
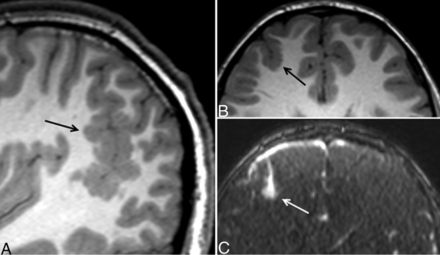
PMG was seen unilaterally in all 12 instances. Multifocal PMG was seen in 4/12 cases. In 1 case, PMG was seen adjacent to a previously resected AVM. In 2 patients, focal PMG was seen immediately adjacent to a small AVM (Fig 2). In another case, PMG was noted to line a large porencephalic cyst. Clinically, this patient had both epilepsy and hemiplegia and was the only patient to exhibit either of those manifestations among the cases of MCD. Additional neurologic symptoms included 6 patients with headaches, 1 with a gait disorder, and 1 with a prior stroke (Table 2).
Eleven-year-old boy with unilateral, multifocal PMG and an AVM. Sagittal T1 MPRAGE shows extensive PMG in the right parietotemporal region (A, arrow). An additional, smaller focus of PMG is seen in the right frontal lobe (B, arrow). Immediately adjacent to the right frontal PMG, postcontrast MRA demonstrates a small enhancing nidus (C, arrow) with cortical venous drainage, consistent with an AVM.
AVMs were seen more frequently in patients with MCD compared with those without MCD. Eight of 13 (61.5%) patients with MCD who underwent testing had a pulmonary AVM; 30/100 (30.0%) of patients without MCD who underwent testing had a pulmonary AVM (P = .029). Five of 14 (35.7%) patients with MCD also had a brain AVM, compared with 13/102 (12.7%) patients without MCD (P = .042).
Discussion
This study demonstrated a relatively high prevalence (12.1%) of MCD within a pediatric HHT population, consistent with the results reported in the conference abstract by Bergerot et al13 in 2013. Most cases of MCD were PMG, seen predominantly in a perisylvian distribution, which is the most common location for PMG in general.14 The prevalence of PMG in the general population is unknown, but is likely quite rare. Large series of brain MRIs of healthy, asymptomatic patients reported no cases of PMG.15,16 A study looking at incidental brain MRI findings in 120 healthy Japanese children, which included MPRAGE sequences, also reported no cases of PMG.17 Many possible causes for PMG have been identified, including various in utero neurologic insults. Prenatal infection with cytomegalovirus has been associated with the development of PMG.18 In utero ischemic insults have also been implicated as a cause of both layered and unlayered PMG.19 PMG has been described in association with several genetic mutations and metabolic diseases.10,12
Although the cause for MCD in HHT is unclear, the distribution of PMG in our cases supports a regional rather than global pathogenesis. Twelve of 12 cases were unilateral, which is notable for the fact that a prior series of 71 cases of PMG showed unilaterality in only 42% of cases.14 In another series of 328 patients with PMG, the unilateral perisylvian pattern was seen only 9% of the time.20 The genetic basis for HHT is abnormal vascular development related to various mutations in the transcription growth factor-β pathway. Three of our cases provide circumstantial support of abnormal vascular development being associated with PMG by virtue of spatial proximity. This includes 2 cases of a small AVM directly adjacent to focal PMG and another case with PMG lining an AVM resection cavity.
Notably, the prevalence of both brain AVMs and pulmonary AVMs was significantly higher in our patients with MCD compared with those without MCD. In 1 of our cases, PMG was seen adjacent to a porencephalic cyst, an association that has been described previously and attributed to prenatal ischemia.21 The pathogenesis of brain AVMs and vascular dysplasia has been recently investigated in mouse models of HHT. Given the results of our study, it would be informative to investigate for cortical malformations in these mouse models.
Genetic testing was not available for most of our cases, but at least 5 of 12 cases with PMG were very likely in patients with ENG mutations. Endoglin mRNA expression peaks throughout the brain during the latter half of gestation and the perinatal period and, in the brain, spatiotemporally correlates with the expression of other genes involved with transcription growth factor-β–mediated angiogenesis, such as ALK1, another gene implicated in HHT.22
The clinical manifestations of PMG are widely variable, including developmental delay, cognitive abnormalities, and epilepsy. The incidence of epilepsy in the setting of PMG has been reported to be as high as 78%.20 However, in our study, only 1 patient, who had a large porencephalic cyst in the left cerebral hemisphere, had documented epilepsy. A review of 87 patients with PMG and epilepsy found that a more extensive distribution of PMG correlated with an earlier age of seizure onset.23 More favorable clinical outcomes are seen when PMG is unilateral, localized, and not associated with other structural malformations.10 Our findings suggest that PMG in the HHT population may be subclinical, possibly because of its unilaterality and lack of association with other major structural abnormalities. Nevertheless, it is likely prudent to consider the possibility of PMG as a cause for seizures in patients with HHT and epilepsy, in addition to the more well-known causes such as brain AVM, abscess, and infarct.
Our study is limited by its retrospective nature and variability in MR imaging protocols that may alter the sensitivity for cortical malformations or brain AVMs. At present, PMG is likely underdiagnosed in the HHT population, as evidenced by the fact that several of the cases with PMG were only identified in this retrospective review. Our results suggest that a high spatial resolution sequence in brain MRIs performed for HHT evaluation may be useful.
Conclusions
In summary, our study confirms a high prevalence of MCDs in the pediatric HHT population, particularly perisylvian PMG. Our data suggest that PMG in this setting is typically subclinical, but can be associated with epilepsy and often coexists with other manifestations of HHT. Moreover, though the underlying pathogenesis of MCDs in HHT remains unknown, our findings suggest an association between vascular anomalies and PMG.
Footnotes
M.S. Goyal and A.J. White are senior authors who contributed equally to this work.
Disclosures: Manu Goyal—UNRELATED: Grants/Grants Pending: ASNR/RSNA Scholar Grant,* McDonnell Center for Systems Neuroscience,* Comments: Scholar Grant 2015–2017, McDonnell Center Grant to obtain lifespan MRA data. *Money paid to the institution.
Paper previously presented at: American Society of Neuroradiology Annual Meeting 2016, May 21–26, 2016; Washington, D.C.
Indicates open access to non-subscribers at www.ajnr.org
References
- 1.
- 2.
- 3.
- 4.
- 5.
- 6.
- 7.
- 8.
- 9.
- 10.
- 11.
- 12.
- 13.
- 14.
- 15.
- 16.
- 17.
- 18.
- 19.
- 20.
- 21.
- 22.
- 23.
- Received June 22, 2016.
- Accepted after revision August 29, 2016.
- © 2017 by American Journal of Neuroradiology