Abstract
SUMMARY: Sudden sensorineural hearing loss is defined as acute hearing loss of the sensorineural type of at least 30 dB over 3 contiguous frequencies that occurs within a 72-hour period. Although many different causative factors have been proposed, sudden sensorineural hearing loss is still considered “idiopathic” in 71%–85% of cases, and treatments are empiric, not based on etiology. MR imaging implemented with a 3D FLAIR sequence has provided new insights into the etiology of sudden sensorineural hearing loss. Herein, we review the current management trends for patients with sudden sensorineural hearing loss, from the initial clinical diagnosis to therapeutic strategies and diagnostic work-up. We focused primarily on MR imaging assessment and discuss the relevance that MR imaging findings might have for patient management, pointing out different perspectives for future clinical research.
ABBREVIATION:
- SSHL
- sudden sensorineural hearing loss
According to the guidelines of the Committee on Hearing and Equilibrium of the American Academy of Otolaryngology, sudden sensorineural hearing loss (SSHL) is defined as acute hearing loss of the sensorineural type of at least 30 dB over 3 contiguous frequencies occurring within a 72-hour period.1 SSHL occurs most often in the fourth decade of life, with an annual incidence that varies between 5 and 20 cases among 100,000 people. The severity of hearing loss is variable and is usually unilateral, though bilateral involvement has been reported in up to 4% of patients.2
Although SSHL has been attributed to many different causative factors,3 there are still no audiologic means of accurately assessing its etiology,4 so it is still considered “idiopathic” in 71%–85% of cases.5 Consequently, there is not a treatment that targets the etiology, which partly explains the inconsistent results of the various empiric drug protocols proposed, which include systemic steroids, antiviral medications, vasodilators, and carbogen or hyperbaric oxygen alone or in combination, none of which has been proved to be superior to the others.6⇓–8
Over the past decade, MR imaging has provided new insights about SSHL etiology because of high-resolution sequences that are able to detect subtle changes in the inner ear.9⇓⇓⇓⇓⇓–15 However, for many reasons, the use of this imaging technique for SSHL is still not widespread. First, many radiologists are not yet aware of the possibilities offered by advanced MR imaging studies of the inner ear and/or of the possible MR imaging findings in SSHL. Second, it is still unclear and is not yet addressed in the literature how MR imaging could change therapeutic strategies.
In this paper, we review the current management trends of patients with SSHL, from the initial clinical diagnosis to the therapeutic strategies, through the diagnostic work-up. Our work focuses particularly on MR imaging assessment. We discussed how MR imaging should be performed and the relevance that the possible MR imaging findings in the inner ear might have. Finally, we addressed the perspectives of possible changes in the therapeutic management of SSHL based on MR imaging findings, which could impact the patient's prognosis.
Current Diagnostic Work-Up of SSHL
SSHL is suspected in patients with a sudden onset of generally unilateral decrease or loss of hearing, occurring instantaneously or rapidly developing over a period of hours or days.3 In other instances, primary care or emergency department physicians refer patients to an audiologist.16,17
The evaluation usually begins with a careful history and physical examination to look for potential causes such as infections, systemic diseases, and exposure to known ototoxic medications. Otomicroscopy is generally negative for external and middle ear pathologies. Pure-tone audiometry is the main diagnostic tool used to differentiate between conductive and sensorineural hearing loss, which have very different management strategies. The physician can differentiate SSHL (symptoms occur within 3 days) from progressive or fluctuating SSHL. The presence of bilateral sudden hearing loss, recurrent episodes of sudden hearing loss, or focal neurologic findings suggests systemic disorders, autoimmune or metabolic disorders, bilateral Menière disease, or primary neurologic disorders.
If SSHL is diagnosed, an empiric treatment is started while the diagnostic work-up continues. A complete audiovestibular evaluation, including speech audiometry, speech in noise test, tympanometry, acoustic reflexes, and otoacoustics emissions, is performed in an attempt to investigate whether the SSHL is cochlear or retrocochlear.8
Brain MR imaging with and without gadolinium is often used to exclude a vestibular schwannoma (reported in up to 10%–20% of patients with SSHL) as well as rarer causes of retrocochlear hearing loss such as other cerebellopontine tumors, brain stem infarctions, and demyelinating disease.3,8,18⇓⇓–21 A number of studies have advocated the use of MR imaging without gadolinium as the more appropriate means of screening patients with asymmetric SSHL suspected to be retrocochlear.22
In cases of suspected cochlear SSHL, different studies have proposed a tailored temporal bone MR imaging with 3D-FLAIR sequence to exclude abnormalities in the inner ear structures.9⇓⇓⇓⇓⇓–15 MR imaging of the temporal bone can be negative, and the SSHL is therefore defined as idiopathic, or the MR imaging can show abnormalities in the inner ear structures, suggesting a specific etiopathogenesis (vascular or inflammatory).14 In both cases, because no trial has yet investigated different medical protocols guided by MR imaging findings, physicians continue with empiric therapy, so temporal bone MR imaging with a 3D-FLAIR sequence has not had any effect on the therapeutic management of cochlear SSHL in clinical practice. Nevertheless, in our opinion, it is important that radiologists perform temporal bone MR imaging with a 3D-FLAIR sequence to clarify the cochlear origin of SSHL, suggesting the probable pathogenesis, and provide prognostic information to physicians.
How We Perform MR Imaging in Patients with SSHL
MR imaging of the temporal bone is challenging because of the complexity and small dimensions of the anatomic structures. Thus, MR imaging should preferably be performed on a 3T scanner, which provides high-resolution images with a higher signal-to-noise ratio compared with a 1.5T scanner. The basic MR imaging protocol should include an axial precontrast 3D steady-state free procession sequence, a pre- and postcontrast T1-weighted FSE sequence, and a pre- and postcontrast 3D-FLAIR sequence.4⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓–15 The On-line Table summarizes the MR imaging scan parameters used in our department. It is recommended to use both postcontrast 3D-FLAIR and postcontrast T1-weighted FSE sequences because the former are more sensitive than a T1-weighted sequence in detecting intralabyrinthine contrast enhancement,11,14,23 and the latter can clarify the presence of a small schwannoma.
Postcontrast sequences should be acquired approximately 10 minutes after administration of the contrast agent.13,15 To cover part of the 10 minutes needed before the acquisition of postcontrast sequences, it is advantageous to inject a contrast agent before the acquisition of the 3D steady-state free procession sequence. Although the contrast enhancement of some structures can be seen on the 3D steady-state free procession sequence,24 the assessment of the inner ear anatomic structures and internal auditory canal on this sequence is usually not hindered by the presence of the contrast agent.
What Can MR Imaging Detect?
Neuroradiologists should review MRIs and look for asymmetry of the signal between the affected and unaffected sides. In the literature, abnormalities on MR imaging are reported in 27%–53% of SSHL cases.10,14
Two patterns can be recognized based on the MR imaging signal of the inner ear on precontrast T1-weighted and 3D-FLAIR images:
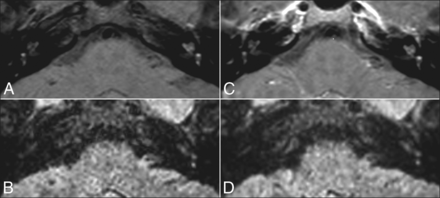
The vascular pattern shows hyperintensity on precontrast T1-weighted and 3D-FLAIR images because of the presence of methemoglobin in the inner ear (Fig 1); and
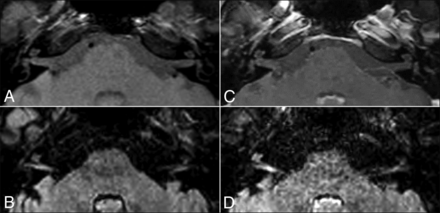
The inflammatory pattern shows hyperintensity only on 3D-FLAIR images because of the presence of proteinaceous exudate in the inner ear (Fig 2).
Vascular pattern in a 20-year-old woman with left SSHL. Precontrast T1-weighted (A) and precontrast 3D-FLAIR (B) sequences show a high signal in the middle and upper turns of the left cochlea without enhancement on postcontrast T1-weighted (C) and 3D-FLAIR images (D).
Inflammatory pattern in a 35-year-old man with right SSHL. The precontrast T1-weighted sequence (A) shows no signal abnormalities. The precontrast 3D-FLAIR sequence (B) shows a high signal in the right cranial nerves VII and VIII and in the middle and upper turns of the left cochlea. A postcontrast T1-weighted sequence (C) does not show enhancement, whereas a postcontrast 3D-FLAIR sequence (D) shows the cochlea and cranial nerves VII and VIII as markedly enhanced on the right side.
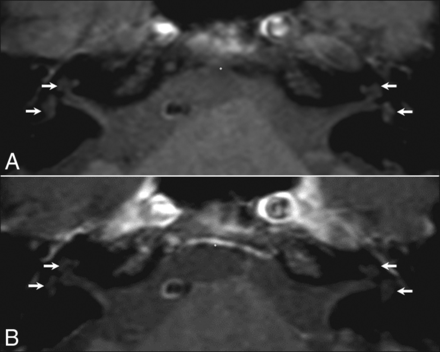
Regarding the vascular pattern, radiologists should be aware that pseudohyperintensity of the intralabyrinthine fluid can normally be detected on fat-suppressed T1-weighted images, which could hamper the diagnosis. This pseudohyperintensity has previously been described as an artifact25 and can be differentiated from methemoglobin because it is symmetrical and less evident on postcontrast fat-suppressed T1-weighted images (see Fig 3 for a detailed explanation).
Normal MR imaging findings in a 59-year-old man with right SSHL. The precontrast T1-weighted sequence (A) shows spontaneous hyperintense intralabyrinthine fluid in both inner ear structures (arrows), which is symmetric and less evident on the same sequence after contrast injection (B). This hyperintensity is an artifact caused by the altered dynamic range when the fat signal is subtracted, but also reflects an alteration in the visual appearance of signal intensity as the ambient contrast is changed (checker-shadow illusion).24
Recent studies have investigated the relationship between precontrast 3D-FLAIR signals and clinical findings regardless of the hyperintensity on precontrast T1-weighted sequences.9⇓–11,23 The rate of abnormalities on precontrast 3D-FLAIR images correlated with the level of hearing loss at onset, resulting in fewer abnormalities in patients with mild to moderate hearing loss compared with patients with profound hearing loss.10,14,15 However, Yoshida et al11 did not observe similar results. Inflammatory diseases can also affect the vestibule, the semicircular canals, or cranial nerve VIII, and precontrast 3D-FLAIR images can detect signal abnormalities in these structures (Fig 3).10⇓⇓⇓–14,23 Hyperintensity on precontrast 3D-FLAIR images of the vestibule or the semicircular canals has been associated with vertigo,11,14 which is reported in approximately 30% of SSHL cases.9
The 2 MR imaging patterns are not always associated with inner ear enhancement on postcontrast 3D-FLAIR, which is consistent with blood-labyrinth barrier breakdown.14 The advantage of postcontrast 3D-FLAIR images is that they identify patients with more severe blood-labyrinth barrier breakdown when signal abnormalities are subtle on precontrast 3D-FLAIR images.23 However, the significance of the inner ear enhancement is still unclear. Viral infection, immune-mediated inner ear disease, and perilymphatic fistulas have been suggested as possible causes of cochlear enhancement.26,27
Usually, the 3D T2-weighted steady-state free procession sequence does not show any pathologic findings23; nevertheless, it is essential to investigate the morphology of the inner ear structures, cranial nerve VIII, internal auditory canal, and cerebellopontine angle.
Prognostic Value of MR Imaging
To the best of our knowledge, no published studies have investigated the difference in prognosis and outcome between the 2 MR imaging patterns mentioned above, though a vascular pattern has been associated with a poor prognosis.28 On the contrary, the published studies have concentrated on the prognostic value of 3D-FLAIR abnormalities and reported conflicting results. Two studies showed that a high signal in the affected inner ear on precontrast 3D-FLAIR was associated with a poor prognosis.11,15 In 2 other studies, the hearing outcome was worse in patients with multiple-location hyperintensities on precontrast 3D-FLAIR (cochlea plus vestibule) than in patients with a single-subsite hyperintensity (cochlea only).12,29 A recent study by Liao et al23 showed that the more asymmetric FLAIR signal between the affected ear and the normal one and presence of a high signal beyond the cochlea indicated a poorer prognosis. Lee et al10 demonstrated that precontrast 3D-FLAIR abnormalities do not affect the prognosis when the initial hearing loss is mild to moderate, whereas such abnormalities represent a negative prognostic factor in patients with initial profound hearing loss. However, Berrettini et al14 failed to find a correlation between the severity of precontrast 3D-FLAIR abnormalities and hearing improvement. Lee et al9 reported that a high signal on precontrast 3D-FLAIR did not significantly affect the final hearing ability.
These inconsistent results are partially explained by methodologic differences, including the time span between SSHL onset and MR imaging, method of MR imaging assessment, pharmacologic protocols of drug administration during follow-up, length of follow-up, and assessment criteria used to determine hearing improvement.30 A meta-analysis by Gao and Chi30 that included studies of patients without primary treatment before temporal bone MR imaging concluded that precontrast 3D-FLAIR hyperintensity in the inner ear is associated with more severe initial hearing loss and a lower chance of recovery.
Current Management of SSHL and Perspectives in MR Imaging Research
The results of SSHL treatment are still largely unpredictable; a very large variability in responses has been reported, ranging from no response to total recovery.7,31 Moreover, the high rate of spontaneous recovery, which varies from 45%–65%,21 should be considered.
Although cochlear vascular microthrombosis has been hypothesized as the main pathogenic mechanism,32⇓–34 because there is no objective test that can detect the occlusion of microvessels, the pharmacologic treatment remains highly empirical, and its overall efficacy is controversial because of the absence of prospective double-blind studies.3 Pharmacologic treatment includes many drugs that, without a certain etiology of SSHL, are often prescribed in combination: oral and/or intratympanic corticosteroids, hyperbaric oxygen therapy, antivirals drugs, vasodilators, and vasoactive substances.
For decades, the “gold standard” treatment for SSHL has been the oral administration of corticosteroids.35,36 The exact mechanism by which steroids improve hearing is still unknown, though some of the major hypotheses are that they modulate cochlear function, decrease inflammation and edema, improve cochlear blood flow, and protect against cochlear ischemia. Although the evidence concerning the use of oral corticosteroids remains contradictory,37 it seems reasonable to offer them because of the potentially devastating disability caused by SSHL and the relatively low morbidity of the treatment.3 Intratympanic steroid treatment allows high steroid concentrations to be reached in the perilymph while avoiding the common side effects of systemic steroids. A recent meta-analysis has shown the benefits of intratympanic steroid treatment in combination with oral corticosteroids as the first-line therapy and salvage monotherapy in idiopathic SSHL.38 Hyperbaric oxygen treatment is also used as primary, adjuvant, or salvage therapy because it is thought to protect hair cells from ischemic damage by increasing oxygenation.39,40 However, cost/benefit issues, limited availability, and the absence of strong evidence because of the difficulties of organizing controlled randomized studies have halted its widespread application. Although there is no clear evidence supporting their use, antiviral drugs are often used in pharmacologic protocols for the treatment of SSHL because various viruses have been implicated in its etiology.41 Vasodilators and vasoactive substances are sometimes used because obstructed vascular flow to the cochlea is a theoretic cause of SSHL, but there is currently insufficient evidence to support their routine use.3
Future research should investigate the role of MR imaging with a 3D-FLAIR sequence in the therapeutic management of SSHL (On-line Figure). Before future trials can investigate different medical protocols guided by MR imaging findings, longitudinal studies should investigate the effects of early steroid treatment and the time interval between disease onset and scanning on the sensitivity of MR imaging. In our opinion, its optimal timing would be upon admission to the emergency department or on the same day as a clinical assessment by an audiologist or otolaryngologist. We hypothesize that precontrast and contrast-enhanced MR imaging abnormalities are more easily detected before steroid treatment is started, so MR imaging should be performed as soon as possible, and preferably before the masking effect of steroids becomes apparent.28 In line with this suggestion, Berrettini et al14 have reported that the time interval between SSHL onset and MR imaging tended to be shorter in patients with 3D-FLAIR abnormalities than in those without 3D-FLAIR abnormalities (P = .06). However, this has not been confirmed by other authors.
Conclusions
MR imaging with 3D-FLAIR sequences provides new insights into SSHL etiology and may change current clinical and therapeutic practices. Radiologists should therefore be trained to perform tailored temporal bone MR imaging in the case of SSHL and recognize the common findings and pitfalls of the technique to provide clinicians with information useful for patient management.
REFERENCES
- 1.
- 2.
- 3.
- 4.
- 5.
- 6.
- 7.
- 8.
- 9.
- 10.
- 11.
- 12.
- 13.
- 14.
- 15.
- 16.
- 17.
- 18.
- 19.
- 20.
- 21.
- 22.
- 23.
- 24.
- 25.
- 26.
- 27.
- 28.
- 29.
- 30.
- 31.
- 32.
- 33.
- 34.
- 35.
- 36.
- 37.
- 38.
- 39.
- 40.
- 41.
- Received January 5, 2017.
- Accepted after revision March 20, 2017.
- © 2017 by American Journal of Neuroradiology