Abstract
BACKGROUND AND PURPOSE: The safety and efficacy of tirofiban during endovascular therapy in patients undergoing intravenous thrombolysis with recombinant IV tPA remain unclear. This study aimed to investigate the safety and efficacy of intra-arterial tirofiban use during endovascular therapy in patients treated with IV tPA.
MATERIALS AND METHODS: Using a multicenter registry, we enrolled patients with acute ischemic stroke who underwent endovascular therapy. Safety outcomes included postprocedural parenchymal hematoma type 2 and/or thick subarachnoid hemorrhage, intraventricular hemorrhage, and 3-month mortality. Efficacy outcomes included the successful reperfusion rate, postprocedural reocclusion, and good outcomes at 3 months (mRS scores of 0–2). The tirofiban effect on the outcomes was evaluated using a multivariable analysis while adjusting for potential confounders.
RESULTS: Among enrolled patients, we identified 314 patients with stroke (279 and 35 patients in the no tirofiban and tirofiban groups, respectively) due to an intracranial artery occlusion who underwent endovascular therapy with intravenous thrombolysis. A multivariable analysis revealed no association of intra-arterial tirofiban with postprocedural parenchymal hematoma type and/or thick subarachnoid hemorrhage (adjusted OR, 1.07; 95% CI, 0.20–4.10; P = .918), intraventricular hemorrhage (adjusted OR, 0.43; 95% CI, 0.02–2.85; P = .467), and 3-month mortality (adjusted OR, 0.38; 95% CI, 0.04–1.87; P = .299). Intra-arterial tirofiban was not associated with good outcome (adjusted OR, 2.22; 95% CI, 0.89 –6.12; P = .099).
CONCLUSIONS: Using intra-arterial tirofiban during endovascular therapy after IV tPA could be safe.
ABBREVIATIONS:
- aOR
- adjusted OR
- EVT
- endovascular therapy
- IA
- intra-arterial
- ICAS-O
- intracranial atherosclerotic stenosis–related occlusion
- IVT
- intravenous thrombolysis
- LVO
- large-vessel occlusion
- mTICI
- modified TICI
Given the positive findings of randomized controlled trials of endovascular therapy (EVT) with newer devices,1⇓⇓⇓-5 EVT has become a standard therapy for anterior circulation ischemic stroke.2,4 Although it remains unclear whether EVT combined with intravenous thrombolysis (IVT) with tPA is better than EVT alone, the American Stroke Association/American Heart Association guidelines recommends IVT for eligible patients with large-vessel occlusion (LVO).6
IV tPA improves outcomes in patients with acute ischemic stroke.7 However, given that IV tPA increases the risk of intracranial hemorrhage, it limits additional procedural techniques during EVT. A large pivotal study on EVT reported that 29% of patients lacked successful reperfusion (modified TICI [mTICI] ≧ 2b).8 Additionally, during EVT, endothelial damage can occur with resulting platelet activation, which causes reocclusion.9 This often requires rescue treatment, including balloon angioplasty, stent placement, or adjuvant thrombolytic infusion. Although antiplatelet agents or thrombolytic infusion has benefits in cases involving stent deployment or ongoing thrombus formation, these treatments may increase the risk of bleeding complications.
Tirofiban is the most commonly used rescue thrombolytic.10 However, its safety and efficacy in EVT among patients with acute ischemic stroke remain unclear.11⇓⇓⇓⇓⇓-17 Additionally, although studies of EVT have reported that 83% of patients were treated with IV tPA before EVT,8 there is no evidence regarding the use of tirofiban during EVT in patients treated with IV tPA.
Therefore, this study aimed to investigate the safety and efficacy of intra-arterial (IA) tirofiban during EVT in patients treated with IV tPA.
MATERIALS AND METHODS
Patients
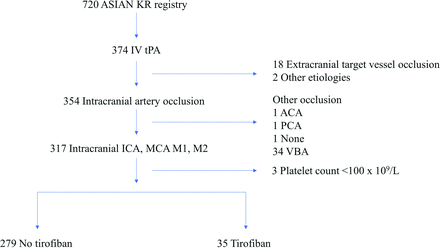
This study used the Intracranial Atherosclerotic Occlusion and Neurointervention-Korean Retrospective registry. Briefly, this study collected data from patients with acute ischemic stroke who underwent EVT between 2011 and 2016 in 3 comprehensive centers in Korea. The inclusion criteria of the present study were the following: 1) treated with IV tPA for acute ischemic stroke within 4.5 hours of symptom onset, and 2) having LVO in the ICA, MCA M1, and MCA M2. The exclusion criteria were the following: 1) missing 3-month mRS scores; 2) extracranial and/or tandem intracranial large-artery occlusions; 3) known thrombocytopenia at presentation or a thrombocyte count of ≤ 100 × 109/L; and 4) having other stroke etiologies, including vasculitis, arterial dissection, or Moyamoya disease. This study was approved by the local institutional review board (Keimyung University Dongsan Hospital IRB: 2016–01–038-009; Ajou University Hospital IRB: AJIRB-MED–OBS–15-483, AJIRB-MED–OBS–17-094; Kyungpook National University Hospital IRB: 2016–01–020-006), which waived the requirement for written informed consent given the retrospective and registry-based design. The data sets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
IVT and EVT Procedures
On the basis of the international guidelines, IV tPA, 0.9 mg/kg, was administered within 4.5 hours of symptom onset in all centers.6 Angiography was usually performed with the patient under local anesthesia. The EVT procedure was chosen at the discretion of neurointerventionalists. Stent retrieval and contact aspiration methods were routinely selected. In case of failed EVT for LVO, rescue treatments, including emergency stent placement, balloon angioplasty, or tirofiban infusion, were administered.
IA Tirofiban Treatment
The choice of using tirofiban was similarly at the discretion of the interventionalists in the following situations: 1) when the interventionalist detected thrombus embolization causing distal arterial occlusion and thrombus was likely to be distally embolized and risky; 2) when reocclusion reoccurred due to intracranial atherosclerosis; 3) when the target artery remained occluded or rescue treatment with emergency stent placement or balloon angioplasty due to postthrombectomy residual stenosis was required; and 4) when patients had a high possibility of reocclusion (high-grade intracranial stenosis or progression of stenosis, and so forth) after occluded arteries were recanalized during EVT. Treatment involved 0.5 mg (2 mL) of tirofiban diluted with 8 mL of normal saline injected with an infusion rate of 1 mL/min. Alternately, if 1 mg (4 mL) of tirofiban was required at first, treatment involved 1 mg (4 mL) of tirofiban diluted with 6 mL of normal saline injected at a rate of 1 mL/min. On follow-up angiography immediately and 10 minutes after infusion of IA, if additional tirofiban was needed, the same protocol was used. Total IA tirofiban infusion ranged from 0.5 to 2.0 mg.
Data Acquisition and Outcomes
We analyzed clinical characteristics, including age, sex, NIHSS score on admission, and baseline mRS scores. The LVO etiology was determined using angiographic diagnosis as previously reported.19 Brain CT was performed to evaluate hemorrhagic complications immediately and 12–24 hours after EVT. The safety outcomes included adverse hemorrhagic complications, intraventricular hemorrhage, and mortality. Intracranial hemorrhages were classified on the basis of the European Cooperative Acute Stroke Study.20 Serious hemorrhagic complications were defined as parenchymal hematoma type 2 and/or thick SAH with or without intraventricular hemorrhage (modified Fisher grade 3 or 4 of SAH). The efficacy outcomes included successful reperfusion, postprocedural reocclusion, and 3-month mRS scores. On the basis of the final angiography, successful reperfusion was defined as an mTICI grade of 2b or 3. Postprocedrual reocclusion was evaluated using angiography performed until discharge by comparing it final angiography performed during EVT. Good outcomes at 3 months were defined as mRS scores of 0–2.
Statistical Analyses
Descriptive statistics were used for between-group comparisons of patient characteristics and outcomes. Categoric variables were analyzed using χ2 tests or Fisher exact tests and were presented as percentages. Continuous variables were analyzed using the Student t test or Mann-Whitney U test and were presented as median and interquartile range.
Multivariable logistic regression analysis was performed to evaluate the efficacy and safety outcomes. Variables with P < .15 in the bivariate analysis were included in the multivariable logistic regression analysis. All statistical analyses were performed using R statistical and computing software (http://www.r-project.org/). For all analysis, P < .05 was considered statistically significant.
RESULTS
Patients
We analyzed a total of 314 patients who underwent EVT combined with IV tPA. Among them, 35 patients were treated with IA tirofiban (tirofiban group), while 279 patients were not (no tirofiban group) (Fig 1). Table 1 shows the baseline characteristics of the recruited participants. Compared with the no tirofiban group, the tirofiban group had significantly more cases of intracranial atherosclerotic stenosis–related occlusion (ICAS-O) (10.0% versus 51.4%, P < .001), higher rates of intracranial stent placement (3.2% versus 17.1%, P = .001), and higher rates of intraprocedural reocclusion (3.6% versus 31.4%, P < .001). There was no significant between-group difference in the age, baseline NIHSS scores, and ASPECTS.
Study flow chart. The Acute Stroke due to Intracranial Atherosclerotic occlusion and Neurointervention-Korean Retrospective (ASIAN KR) registry. ACA indicates anterior cerebral artery; PCA, posterior cerebral artery; VBA, vertebrobasilar artery.
Baseline characteristics of patients
Patient Outcomes: Safety and Efficacy
The Table 2 present the safety and efficacy outcomes. Serious hemorrhage was observed in 11.1% and 8.6% of patients in the no tirofiban and tirofiban groups, respectively (P = .867). Intraventricular hemorrhage was observed in 6.8% and 2.9% of the patients in the no tirofiban and tirofiban groups, respectively (P = .592). The 3-month mortality rate was 12.5% and 5.7% in the no tirofiban and tirofiban groups, respectively (P = .366). Multivariable analysis revealed no association of IA tirofiban with serious hemorrhage (adjusted OR [aOR], 1.07; 95% CI, 0.20–4.10; P = .918), intraventricular hemorrhage (aOR, 0.43; 95% CI, 0.02–2.85; P = .467), and death at 3 months (aOR, 0.38; 95% CI, 0.04–1.87; P = .299). There was successful reperfusion in 75.3% and 80.0% of patients in the no tirofiban and tirofiban groups, respectively (P = .684). After adjustment, IA tirofiban was not associated with successful reperfusion (aOR, 1.56; 95% CI, 0.5–5.34; P = .440).
Patient safety and efficacy outcomes
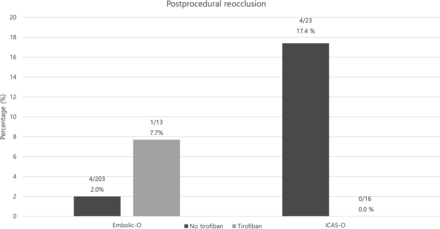
At 3 months, 53.8% and 57.1% of patients in the no tirofiban and tirofiban groups, respectively, showed good outcome (P = .843). Postprocedural reocclusion until discharge was observed in 4.4% and 2.9% of the patients in the no tirofiban and tirofiban groups, respectively (P = .688). We analyzed 255 patients in whom postprocedural reocclusion among patients with mTICI ≥ 2a was assessed for subgroup analysis. Four (17.4%) patients in the no tirofiban group and none (0%) in the tirofiban group had postprocedural reocclusion until discharge in ICAS-O, but the difference was not statistically significant (4/23, 17.4% versus 0/16, 0%, P = .221). In embolic occlusion, postprocedural reocclusion was not found to be different between the no tirofiban and the tirofiban groups (4/203, 2.0% versus 1/13, 7.7%, P = .705) (Fig 2).
Subgroup analysis for postprocedural reocclusion in patients with mTICI ≥2a. In Embolic-O, postprocedrual reocclusion is 2.0% in patients without IA tirofiban and 7.7% in those with IA tirofiban. Patients with ICAS-O with IA tirofiban tend to have less postprocedural reocclusion than those without IA tirofiban (no tirofiban, 17.4%, versus tirofiban, 0.0%). Embolic-O indicates embolic occlusion.
DISCUSSION
In clinical practice, the efficacy of antiplatelet administration when performing EVT in patients treated with IV tPA remains unclear. There is limited available evidence regarding optimal antiplatelet administration during EVT, with antiplatelets being recommended during stent deployment for EVT;21 however, the 2018 American Heart Association/American Stroke Ass-ociation guidelines indicate that aspirin administration should generally be delayed until after 24 hours for those treated with IV tPA.6 Tirofiban may be an alternative rescue treatment for neurointerventionalists. Related literature is limited to case series and retrospective reviews of a single-center data base;22,23 moreover, these studies were performed without considering the use of IV tPA. Therefore, the safety and efficacy of tirofiban during EVT after IV tPA remain unclear. In this study, IA injection of tirofiban during EVT in patients treated with IV tPA did not increase the risk of adverse intracranial hemorrhage and mortality compared with patients without tirofiban injections.
Tirofiban is a reversible fibrinogen antagonist that binds to the glycoprotein IIb/IIIa receptor on platelets.24 Tirofiban inhibits platelet aggregation in a dose-dependent manner, which exerts an antiplatelet effect after a 30-minute loading dose. It has a short half-life, with platelet function being normalized after 4 hours.25,26 Given these pharmacokinetics, tirofiban may be beneficial when hemorrhagic complications occur. The inconsistent results of tirofiban as an adjunct therapy during EVT may be attributed to differences in the administration method. Kellert et al12 suggested that IV tirofiban increased fatal intracranial hemorrhage and poor functional outcomes in patients treated with EVT. In contrast to IV tirofiban, previous small-scale studies have shown that IA tirofiban during EVT could be safe.18,27,28 We revealed that IA tirofiban did not increase the bleeding risk in patients who underwent EVT with IV tPA. These results are consistent with previous studies on tirofiban that did not consider using IV tPA.16,29 The safety of tirofiban may be attributed to several advantages when administering tirofiban via IA. IA tirofiban can be administered at a lower dose than IV tirofiban. In the study by Kellert et al, tirofiban was administered IV, infused at 0.4 μg/kg/min during 30 minutes, followed by a continuous infusion of 0.1 μg/kg/min for 48 hours. The total tirofiban dose is 18 mg for a 60-kg adult. Our dose (ranging from 0.5 to 2.0 mg) is smaller than the IV tirofiban dose; moreover, tirofiban can be administered in a target artery. We speculated that the dose was a important factor and that low-dose tirofiban was feasible during EVT in patients treated with IV tPA.
Reocclusion could require additional rescue treatment and eventually delay the final reperfusion time, which results in a poor outcome.30⇓-33 A recent observational study reported more frequent reocclusion in ICAS-O than in embolic occlusion. Additionally, inflammatory reactions and platelet aggregation can result in more ischemic events in patients with ICAS-O. In this study, the tirofiban group showed a high frequency of the ICAS-O subtype. However, our study showed similar good outcomes at 3 months between the 2 groups, even though there was more intraprocedural reocclusion in the tirofiban group. A previous study suggested that ICAS-O had good collaterals.34 Good collaterals are related to hemodynamic factors, including increased distribution of thrombolytics to the clot surface, potentially making the clot more susceptible to thrombolysis. This feature may affect clinical outcomes in patients treated with IA tirofiban. Additionally, we analyzed this subgroup to investigate the effect of IA tirofiban on vessel patency in the group with ICAS-O. Patients with ICAS-O with IA tirofiban tend to have less postprocedural reocclusion than those without IA tirofiban. While we did not investigate long-term vessel patency after IA tirofiban, these findings suggested that IA tirofiban may help maintain vessel patency in ICAS-O. Further studies of long-term vessel patency will be needed prove this possibility.
Rescue treatment, including tirofiban, is often required during EVT. However, numerous neurointerventionalists have expressed concerns regarding the use of tirofiban after IV tPA due to the hemorrhage risk. Among patients without EVT, tirofiban after IV tPA has been reported as safe in previous study comparing the safety and preliminary efficacy between patients undergoing IV tPA with tirofiban and those undergoing IV tPA without it;35 this finding is consistent with ours. Our findings regarding safety might inform neurointerventionalists when making decisions regarding the use of tirofiban when rescue treatment is needed during EVT with IV tPA. However, we did not determine the patients eligible for tirofiban administration. Previous studies have demonstrated that tirofiban should be administered to patients with a small core volume.36 Further studies are required to elucidate specific patients eligible for safe tirofiban administration.
This study has several limitations. First, the study is limited in its sample size. Specifically, only 35 patients received IA tirofiban in addition to IV tPA. Second, this was a retrospective study without a prespecified infusion speed and dose of IA tirofiban. However, there were no extensive variances in the dose and infusion speed. All participating centers had a protocol, with the total dose and infusion speed of tirofiban being 0.5-2.0 mg and 0.05–0.1 mg/min, respectively. Third, given that the use of IA tirofiban was at the discretion of the neurointerventionalist, there might be a selection bias. Specifically, the neurointerventionalists might have decided to use IA tirofiban only when they considered it safe.
CONCLUSIONS
Using IA tirofiban during EVT after IV tPA could be safe.
Footnotes
Disclosures: Sung-Il Sohn—UNRELATED: Employment: Keimyung University.
References
- 1.
- 2.
- 3.
- 4.
- 5.
- 6.
- 7.
- 8.
- 9.
- 10.
- 11.
- 12.
- 13.
- 14.
- 15.
- 16.
- 17.
- 18.
- 19.
- 20.
- 21.
- 22.
- 23.
- 24.
- 25.
- 26.
- 27.
- 28.
- 29.
- 30.
- 31.
- 32.
- 33.
- 34.
- 35.
- 36.
- Received December 31, 2020.
- Accepted after revision April 7, 2021.
- © 2021 by American Journal of Neuroradiology