Abstract
BACKGROUND AND PURPOSE: Intravoxel incoherent motion imaging can measure both microvascular and parenchymal abnormalities simultaneously. The contamination of CSF signal can be suppressed using inversion recovery preparation. The clinical feasibility of inversion recovery–intravoxel incoherent motion imaging was investigated in patients with cerebrovascular disease by studying its reproducibility.
MATERIALS AND METHODS: Sixteen patients with cerebrovascular disease (66 ± 8 years of age) underwent inversion recovery–intravoxel incoherent motion imaging twice. The reproducibility of the perfusion volume fraction and parenchymal diffusivity was calculated with the coefficient of variation, intraclass correlation coefficient, and the repeatability coefficient. ROIs included the normal-appearing white matter, cortex, deep gray matter, white matter hyperintensities, and vascular lesions.
RESULTS: Values for the perfusion volume fraction ranged from 2.42 to 3.97 ×10−2 and for parenchymal diffusivity from 7.20 to 9.11 × 10−4 mm2/s, with higher values found in the white matter hyperintensities and vascular lesions. Coefficients of variation were <3.70% in normal-appearing tissue and <9.15% for lesions. Intraclass correlation coefficients were good to excellent, showing values ranging from 0.82 to 0.99 in all ROIs, except the deep gray matter and cortex, with intraclass correlation coefficients of 0.66 and 0.54, respectively. The repeatability coefficients ranged from 0.15 to 0.96 × 10−2 and 0.10 to 0.37 × 10−4 mm2/s for perfusion volume fraction and parenchymal diffusivity, respectively.
CONCLUSIONS: Good reproducibility of inversion recovery–intravoxel incoherent motion imaging was observed with low coefficients of variation and high intraclass correlation coefficients in normal-appearing tissue and lesion areas in cerebrovascular disease. Good reproducibility of inversion recovery–intravoxel incoherent motion imaging in cerebrovascular disease is feasible in monitoring disease progression or treatment responses in the clinic.
ABBREVIATIONS:
- CV
- coefficient of variation
- D
- parenchymal diffusivity
- f
- perfusion volume fraction
- ICC
- intraclass correlation coefficient
- IR
- inversion recovery
- IVIM
- intravoxel incoherent motion
- NAWM
- normal-appearing white matter
- RC
- repeatability coefficient
Perfusion imaging using an exogenous contrast agent is commonly used in the clinic in patients with cerebrovascular diseases.1,2 This technique is sensitive to the microcirculatory condition and provides insight into the cerebral hemodynamics in, for example, patients with ischemic stroke. Because this technique is contraindicated in patients with impaired renal function, there is interest in techniques that avoid contrast agents. For example, by labeling endogenous blood as a tracer, arterial spin-labeling has been used to measure perfusion. However, this technique is associated with low signal-to-noise ratios and is dependent on the arterial transit time. Changes in flow conditions due to disease (eg, stroke) can complicate selecting the correct arterial transit time.3 Another technique without the use of a contrast agent that has received attention is intravoxel incoherent motion (IVIM) imaging. IVIM is a DWI technique that measures the diffusion properties of water molecules in static tissue and in blood flowing into the capillary networks. Therefore, it can measure perfusion and diffusion properties in the brain simultaneously.4 The resulting perfusion volume fraction (f) and parenchymal diffusivity (D) provide information on the microvasculature and parenchymal microstructure, respectively, and have shown promising results in assisting in the assessment of stroke.5,6 Moreover, with this technique, the affected microvasculature and parenchyma in patients with cerebral small-vessel disease have been demonstrated previously.7
Unfortunately, the IVIM signal and the resulting measures can be affected by partial volume effects of CSF. The contamination of CSF has been previously demonstrated to be relevant for DWI, and an improved accuracy of diffusion measures was shown when suppressing the CSF signal.8,9 Recently, this suppression has been applied to IVIM.10 This so-called inversion recovery–based IVIM technique (ie, IR-IVIM) can be particularly helpful for patients with cerebrovascular disease because these patients often have brain atrophy,11 which can increase the CSF contamination.
The reproducibility of IR-IVIM has not yet been fully studied. Several groups have investigated the reproducibility of conventional IVIM and have shown good results (eg, low coefficients of variation [CVs]) for both the perfusion volume fraction and parenchymal diffusivity.12⇓⇓–15 Thus far, only 1 reproducibility study has been performed on IR-IVIM. This study examined only f and healthy participants.10
We aimed to assess the clinical feasibility of IR-IVIM using the reproducibility of the perfusion volume fraction and parenchymal diffusivity.
Materials and Methods
Study Population
For this study, we enrolled patients with varying manifestations of cerebrovascular disease (n = 17): 1) cerebral small vessel disease (n = 11), 2) cortical stroke (n = 5), and 3) intracerebral hemorrhage (n = 1). Patients were recruited in either the transient ischemic attack outpatient service or the stroke unit of the department of neurology of the hospital. A more detailed description of the study population has been published previously.16
All patients were scanned twice on separate days. The patient with intracerebral hemorrhage did not complete both sessions and was excluded from analysis. This exclusion resulted in 16 patients (14 men and 2 women; mean age, 66 ± 8 years; 1.9 ± 2.2 mean days between the 2 sessions) suitable for image analysis.
Standard Protocol Approvals, Registrations, and Patient Consents
The medical ethics committee of our institution approved this study. Written informed consent was obtained from patients before participation.
Imaging Protocol
Participants underwent brain imaging on an MR imaging system (3T, Achieva TX; Philips Healthcare, Best, the Netherlands) using a 32-element head coil suitable for parallel imaging. For IVIM imaging, a Stejskal-Tanner DWI single-shot spin-echo EPI pulse sequence was used (TR/TE, 6800/84 ms; FOV, 221 × 269 × 139 mm3; 2.4-mm cubic voxel size). To suppress the CSF signal, an inversion pulse (TI, 2230 ms) was given before the DWI sequence.10 The total scan time of IVIM imaging using the inversion pulse was 14 minutes. Fifteen DWIs were acquired in 3 orthogonal directions using multiple diffusion-sensitive b-values (0, 5, 7, 10, 15, 20, 30, 40, 50, 60, 100, 200, 400, 700, and 1000 s/mm2). IVIM images were aligned with the corpus callosum (ie, the line connecting the center of the genu and splenium of the corpus callosum). The number of signal averages for the highest 2 b-values was 2 and 3, instead of 1, respectively, to increase the signal-to-noise ratio at high b-values.
For anatomic reference, a T1-weighted sequence (TR/TI/TE, 8.3/800/3.8 ms; FOV, 256 × 256 × 160 mm3; cubic voxel, 1.0 mm3) and T2-weighted FLAIR sequence (TR/TI/TE, 4800/1650/299 ms; FOV, 250 × 256 × 180 mm3; cubic voxel, 1.0 mm3) were performed.
Image Analysis
Brain Segmentation.
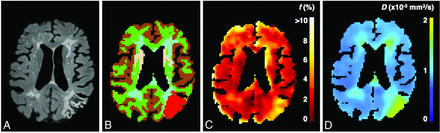
T1-weighted images were segmented into white matter, cortex, and deep gray matter using FreeSurfer software (http://surfer.nmr.mgh.harvard.edu)17 and FSL (Version 5.0; http://www.fmrib.ox.ac.uk/fsl).18 Subsequently, the normal-appearing white matter (NAWM) was separated from the white matter hyperintensities on FLAIR using a semiautomated segmentation algorithm19 followed by visual checks. Visible vascular lesions were identified (n = 4) by a neuroradiologist (>20 years of experience). Because a range of cerebrovascular diseases was included (ie, cerebral small-vessel disease and cortical infarction), we explored various ROIs: NAWM, cortex, deep gray matter, white matter hyperintensities, and vascular lesions (Fig 1A, -B). For patients (n = 5) with an acute infarct, only the contralateral hemisphere was considered for analysis.
A, FLAIR image of a patient with a cortical infarct. Cerebral tissue was segmented (B) into normal-appearing white matter (green), cortex (brown), deep gray matter (pink), white matter hyperintensities (light blue), and vascular lesion (red). C and D, The calculated perfusion volume fraction map and parenchymal diffusivity map are shown, respectively.
IVIM Analysis.
IVIM images were corrected for distortions and head displacements by registering to a corresponding T1-weighted image, using ExploreDTI Version 4.8.3 (http://exploredti.com/).20 Subsequently, trace images were obtained by calculating the geometric mean of the DWIs measured in 3 orthogonal directions. Henceforth, spatial smoothing was performed on the trace images with a 3-mm (full width at half maximum) Gaussian kernel. The signal-to-noise ratio21 of cerebral tissue of the trace images was 60; and of the images obtained in 1 direction, it was 45 with a b-value of 1000 s/mm2, which was higher than the recommended value (ie, 30) for reliable IVIM estimation.22
Model Fitting.
We used a 2-compartment model representing a vascular and nonvascular component, which also accounts for CSF contamination and differences in the relaxation time of blood and tissue.7 The IVIM signal was fitted biexponentially using a 2-step approach, which has been described previously7,23,24: 1) The parenchymal diffusivity was obtained by fitting a monoexponential decay to the signal attenuation curve, obtained with b-values of >200 s/mm2; and 2) a biexponential decay was fitted to the signal attenuation curve, using all b-values and a fixed D, to obtain the perfusion volume fraction. This fitting was performed in a voxelwise manner; and for each ROI, the average IVIM measure was calculated. This calculation was performed with in-house software in Matlab R2012a (MathWorks, Natick, Massachusetts).
Reproducibility Assessment and Statistical Analysis
All IVIM measures obtained with the trace images were calculated for 2 sessions and compared between sessions to evaluate the reproducibility using the coefficient of variation (CV), the repeatability coefficient (RC), and the intraclass correlation coefficient (ICC).25 The CV expresses the relative variation within the subject from 1 session to the second session, whereas the ICC provides information on the effect of this variation with respect to the biologic variation of the study population. Furthermore, the RC indicates the minimal difference needed between the 2 measurements obtained from the same method to be considered an actual change rather than a test-retest error.26
CV was calculated to express the relative within-subject variation and is defined as
 where σws is the population SD during 2 sessions within the same subject and μws the mean value during 2 sessions within the same subject.
where σws is the population SD during 2 sessions within the same subject and μws the mean value during 2 sessions within the same subject.
The repeatability coefficient was calculated as

Both the CV and RC were calculated per participant and averaged over all subjects to represent the overall CV and RC. Low CV and RCs represented good test-retest reproducibility.
Third, the intraclass correlation coefficient was used, defined as
 where σbs2 denotes the biologic variance, which was obtained by calculating the variance over the average values of both sessions in all patients. The ICC expresses the effect of the biologic variance on the total variance rather than the test-retest error (σws2). ICC values toward 1 represent good reproducibility. The ICC was categorized as follows: poor, ICC < 0.40; moderate, 0.40 ≤ ICC < 0.60; good, 0.60 ≤ ICC < 0.75; and excellent, 0.75 ≤ ICC ≤ 1.00.27
where σbs2 denotes the biologic variance, which was obtained by calculating the variance over the average values of both sessions in all patients. The ICC expresses the effect of the biologic variance on the total variance rather than the test-retest error (σws2). ICC values toward 1 represent good reproducibility. The ICC was categorized as follows: poor, ICC < 0.40; moderate, 0.40 ≤ ICC < 0.60; good, 0.60 ≤ ICC < 0.75; and excellent, 0.75 ≤ ICC ≤ 1.00.27
Effect of Smoothing
To analyze the effect of smoothing on the reproducibility, we smoothed DWIs with a Gaussian kernel with increasing full width at half maximum ranging from 0 to 6 mm. Subsequently, CV values were calculated and used for comparing the reproducibility for increased smoothing. This calculation was performed for both f and D and for the ROIs in the white and gray matter.
Results
The Table shows the results of the reproducibility of the IVIM parameter perfusion volume fraction and parenchymal diffusivity. Figure 1C, -D shows examples of f and D maps. The measured f (2.42–2.90 × 10−2) and D (7.20–7.44 × 10−4 mm2/s) seen in patients with cerebrovascular disease are in the range of expected values in NAWM and gray matter. White matter hyperintensities and vascular lesions had values for f and D ranging from 3.22–3.97 × 10−2 and 8.83–9.11 × 10−4 mm2/s, respectively. CVs of ≤3.7% for f and D were observed for the white and gray matter. Vascular lesions showed relatively higher CVs for f and D of 9.15 and 1.30%, respectively. Excellent ICCs (0.82–0.97) were observed for both IVIM parameters, except for f in the deep gray matter and D in the cortex showing moderate-to-good ICCs. Furthermore, RCs ranged from 0.15–0.96 × 10−2 for f and from 0.09–0.37 × 10−4 mm2/s for D.
Reproducibility values of the perfusion fraction and parenchymal diffusivity
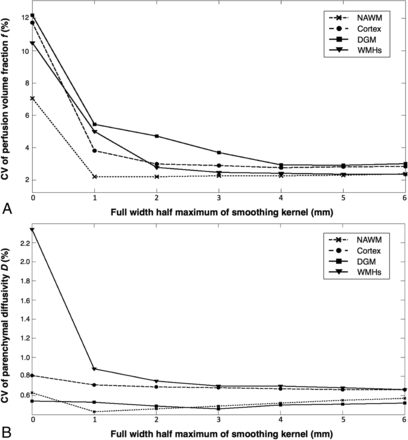
When comparing our CVs obtained with smoothing (ie, full width at half maximum = 3 mm) with CVs without smoothing (ie, full width at half maximum = 0 mm), we observed lower CVs for f (CV, 2.27%–3.70%) when smoothing was applied than when no smoothing was applied (CV, 7.05%–12.20%) in the white and gray matter. For D, lower CVs were also found when smoothing was applied (CV, 0.49%–0.70%) than without smoothing (CV, 0.54%–2.34%) in the white and gray matter. In Fig 2, the CVs for an increasing size of the smoothing kernel are shown. For f, CVs without smoothing ranged from 7.05%–12.20%, and with smoothing, from 2.20%–5.45% in the white and gray matter. For D, CVs without smoothing ranged from 0.54%–2.34%, and with smoothing, from 0.43%–0.88% in the white and gray matter.
Coefficients of variation as a function of the full width at half maximum of the smoothing kernels ranging from 0 to 6 mm for the perfusion volume fraction (A) and parenchymal diffusivity (B). Higher CVs are present when no smoothing was performed. CVs decreased up to 3 times when smoothing was applied.
Discussion
In this study, we assessed the reproducibility of IR-IVIM. Low CVs and moderate-to-excellent ICCs were observed for both normal-appearing tissue and lesion areas. Furthermore, low values for RCs were observed for all regions.
Several groups have evaluated the reproducibility of conventional IVIM in the brain.12⇓⇓–15 In patients with gliomas, CVs of 6.5% and 0.6% in the NAWM outside the tumor mass for f and D were reported, respectively.12 Furthermore, Peterson et al15 investigated healthy participants and reported moderate-to-excellent ICCs for f in the deep gray matter. In a multicenter study14 also in healthy participants, CVs of f were 5.8% and 11.1%, and CVs of D were 1.7% and 2.1% in the white matter and gray matter, respectively. In comparison with these results, we reported somewhat lower CVs of f and D for the white and gray matter, not exceeding 3.70% and 0.70%, respectively, and comparable ICCs. An explanation for the lower CV values might be the effect of smoothing. We have shown that our CVs with smoothing (ie, full width at half maximum = 3 mm) were generally 3 times lower than when no smoothing was applied. When one assesses CVs with no smoothing (f, CV ≤ 12.20%; and D, CV ≤ 2.34%), it can be observed that CVs are in the same range as in the other studies (ie, f, 5.8%–11.1%; and D, 0.6%–2.1%12,15,14). This finding indicates that the reproducibility of IR-IVIM is like that of conventional IVIM. Nevertheless, differences exist between our study and other studies that can influence the accurate comparison between IR-IVIM and IVIM, including the study population (ie, healthy controls14,15 versus patients with disease and cerebrovascular disease versus brain gliomas12); the exact method used (eg, IVIM versus IR-IVIM and selection of ROIs to analyze the reproducibility); and image-acquisition settings, which can contribute to a better signal-to-noise ratio (eg, the number of averages and the number of diffusion-sensitizing directions).
A study that used a similar IR-IVIM technique also explored the reproducibility of f in 5 healthy adults at 1.5T in the gray matter.10 They reported high-repeatability indices (dissimilar to the repeatability coefficient but comparable with CV) ranging from 27.3% to 65.2%, with a smoothing window full width at half maximum of 0–6 mm. The seemingly higher reproducibility we show from our CVs (ie, full width at half maximum, 0–6 mm: CVs, 2.77%–12.20%) may be explained by several factors: First, our study was performed at a higher field strength (ie, 3T versus 1.5T). At a higher field strength, a better signal-to-noise ratio and/or a higher spatial resolution can be acquired, which may contribute to better reproducibility. Second, the methods used to calculate the reproducibility values were different. Whereas we have calculated reproducibility values from the mean IVIM measure taken over an ROI (ie, “mean method”), they calculated the reproducibility values in a voxelwise manner. An earlier study has shown that CVs calculated in a voxelwise manner are approximately twice as high as CVs obtained with the mean method.25 On the basis of this observation, our CV values of f for the voxelwise method can be roughly estimated to be approximately ≤7.40% for the white and gray matter, which is lower than that reported by Hales and Clark (27.3%–65.2%)10 and is indicative of good reproducibility.
In our study, we applied spatial smoothing, which is beneficial because it improves the signal-to-noise ratio and corrects for small movement or spatial registration errors. We observed that smoothing might lead to improved CVs (f, 2.20%–5.45%; D, 0.43%–0.88%), which were generally up to 3 times lower compared with CVs without smoothing (f, 7.05%–12.20%; D, 0.54%–2.34%). This finding is in accordance with a previous study that reported better reproducibility with an increasing full width at half maximum of the smoothing kernel.10
Lower ICCs in the cortex and DGM can be observed, which indicate lower reproducibility in those areas. We argue that partial volume effects in general can still contribute to the lower reproducibility values. Even though CSF is suppressed and contamination of CSF is reduced, other tissue (eg, white matter) can still contribute to partial volume effects leading to lower reproducibility values.
The clinical contribution of IVIM in neuroimaging is an active research topic. IR-IVIM shows good reproducibility in both normal-appearing tissue and lesion area, which is required when monitoring disease progression or treatment responses. The clinical applicability of our reproducibility results can be demonstrated by the following: A difference in f and D in the stroke region (f, 2.6% ± 1.9%; D, 3.9 ± 0.79 × 10−4 mm2/s) compared with the contralateral side (f, 5.6% ± 2.5%; D, versus 7.5% ± 0.86 × 10−4 mm2/s) has been reported.6 Our RC values in the vascular lesions of 1.0 and 0.4 for f and D, respectively, show that this difference can be easily detected with IR-IVIM. Furthermore, another benefit of IR-IVIM, as opposed to contrast-enhanced techniques, is that it can be performed multiple times to assess both diffusion and perfusion properties without the concern of contrast deposition in the brain. In addition, the advantage of DWI in ischemic stroke has already been widely reported,2 and the IR-IVIM–derived D was corrected for microvascular contributions; this process resulted in a more accurate D (ie, less biased).
The strength of this study is that it was performed in a patient group in which both normal-appearing tissue and lesion areas (ie, white matter hyperintensities and vascular lesions) were investigated. On the contrary, in this study, only 2 sessions were performed, whereas multiple scan sessions could have improved the reproducibility evaluation. Furthermore, IVIM imaging without suppression of the CSF would have provided a more accurate comparison of reproducibility between IVIM with and without CSF suppression. However, IVIM without CSF suppression was not acquired because there was limited scan time. In addition, only a few patients (n = 4) had an acute infarct that we could use to investigate the vascular lesion area. However, we were able to provide some preliminary results. Last, in this study, we assumed a random microvascular network and hence an isotropic f. However, in a recent study, f was shown to be anisotropic in the gray matter.28 Further investigation is needed to study the effect on the reproducibility of fitting the anisotropic f.
Conclusions
IR-IVIM shows good reproducibility in its clinical feasibility in patients with cerebrovascular disease. Good reproducibility is a primary requirement in monitoring disease progression or treatment responses in the clinical setting.
Acknowledgments
The authors greatly appreciate the effort and help of Professor P. Hofman in evaluating MR images on vascular lesions.
Footnotes
Disclosures: Sau May Wong—RELATED: Other: Stichting de Weijerhorst, Comments: Salary. Walter H. Backes—RELATED: Grant: Stichting de Weijerhorst*. *Money paid to the institution.
This project was funded by the Netherlands Organization for Scientific Research, (grant: 017.009.048), Hersenstichting (grant: 2013(1)-195), and Stichting de Weijerhorst Foundation.
Indicates open access to non-subscribers at www.ajnr.org
References
- 1.
- 2.
- 3.
- 4.
- 5.
- 6.
- 7.
- 8.
- 9.
- 10.
- 11.
- 12.
- 13.
- 14.
- 15.
- 16.
- 17.
- 18.
- 19.
- 20.
- 21.
- 22.
- 23.
- 24.
- 25.
- 26.
- 27.
- 28.
- Received June 4, 2017.
- Accepted after revision October 3, 2017.
- © 2018 by American Journal of Neuroradiology