Abstract
BACKGROUND AND PURPOSE: Traditional cardiovascular risk factors have been associated with white matter disease. Because hypertension results in vascular stiffness and impaired cerebral perfusion, we hypothesized that it would be the most relevant risk factor for microstructural white matter disruption in apparently healthy middle-aged individuals with a family history of early-onset coronary artery disease.
MATERIALS AND METHODS: This was a cross-sectional analysis of participants in the Genetic Study of Atherosclerosis Risk with DTI. Regional fractional anisotropy of 181 segmented brain regions was measured using Eve WM Atlas. Risk factors were examined using univariate analysis for 48 regions representing deep WM structures. Minimal multivariable linear regression models adjusting for age, sex, and race and maximal linear regression models adjusting for cardiovascular risk factors were performed for regions meeting the Bonferroni threshold in the initial analysis.
RESULTS: Included were 116 subjects (mean age, 49 ± 11 years; 57% men) with a moderate load of cardiovascular risk factors. Subjects with hypertension had significantly lower regional fractional anisotropy in the right cingulum and left stria terminalis in the minimal and maximal regression models. Additionally, there was lower regional fractional anisotropy in the left fornix in the maximal model and right sagittal stratum in the minimal model. Systolic blood pressure values were significantly associated with regional fractional anisotropy in the left superior longitudinal fasciculus in the maximal model. There were no significant differences among regional fractional anisotropy values for other cardiovascular risk factors.
CONCLUSIONS: In middle-aged apparently healthy individuals with susceptibility to vascular disease, among all known cardiovascular risk factors, hypertension was associated with microstructural WM disruption.
ABBREVIATIONS:
- BMI
- body mass index
- BP
- blood pressure
- CSVD
- cerebral small vessel disease
- FA
- fractional anisotropy
- LDL
- low-density lipoprotein
- rFA
- regional fractional anisotropy
White matter hyperintensities, frequently incidentally observed on brain MR imaging, have been attributed to cerebral small vessel disease (CSVD).1 Histopathologically, WM hyperintensity areas show nonspecific changes of gliosis, myelin and axonal loss, tissue rarefaction, and lipohyalinosis.2 The exact mechanisms leading to CSVD remain undefined.3 It has been hypothesized that interactions between genetics and environmental factors lead to small-vessel changes causing white matter damage secondary to vasculopathy and arteriosclerosis, resulting in hypoxia and hypoperfusion. These events are influenced by altered cerebrovascular autoregulation as well as endothelial and blood-brain barrier dysfunction.4 Among traditional cardiovascular risk factors, hypertension has been consistently found to be related to CSVD.5 Rat models of hypertension develop diffuse WM disease.6 Human studies have linked hypertension directly to arterial stiffness, leading to WM changes related to CSVD.7⇓–9 Other traditional atherogenic risk factors such as diabetes, smoking, and hyperlipidemia have been less consistently linked to CSVD compared with hypertension.10,11 DTI has emerged as a valuable tool for assessment of WM in health and disease through measurement of signal attenuation in the fiber tracts.12 DTI has an advantage over conventional T2 or FLAIR sequences due to improved mapping and 3D diffusion characterization incorporating spatial location. This property can be used to describe the magnitude, degree, and orientation of diffusion anisotropy.12
Fractional anisotropy (FA) is a sensitive biomarker for WM integrity, reflecting axonal loss or demyelination.12 Decreased FA has been found in cases of demyelination, edema, or inflammation.12 In patients with CSVD, impaired WM microstructure has been detected by DTI studies.13 In Nyquist et al,14 our group demonstrated that WM hyperintensity contributes to subclinical psychomotor impairment in apparently healthy subjects. In the current work, we aimed to investigate the relationship of various atherogenic vascular risk factors with the impairment of WM microstructure as measured by DTI. We hypothesized that early changes in regional WM can be detected by DTI in apparently healthy middle-aged individuals and that these changes are primarily associated most strongly with hypertension.
Materials and Methods
Sample and Recruitment
We selected healthy participants who underwent DTI as part of the Genetic Study of Atherosclerosis Risk (GeneSTAR) silent stroke study. GeneSTAR is an ongoing prospective study of vascular disease risk factors, occult coronary artery disease and cerebrovascular disease, and incident coronary artery disease and strokes in 3533 initially healthy family members ascertained from probands hospitalized with documented coronary artery disease younger than 60 years of age.14 Of these initially healthy family members, 808 were screened with cranial MR imaging for WM hyperintensity; of those, 116 healthy subjects had DTI sequences.
The 116 participants included apparently healthy asymptomatic siblings, their offspring, and the offspring of the probands who were 30–72 years of age. None had a known history of coronary artery disease, stroke, or transient ischemic attacks and represented 97 families. Exclusion criteria included the following: chronic corticosteroids, life-threatening diseases, neurologic diseases impairing accurate MR imaging interpretation, implanted metals prohibiting MR imaging, atrial fibrillation, and symptomatic cardiovascular or cerebrovascular disease. The study was approved by the Johns Hopkins Medicine institutional review board (NA_00002856). All participants provided written informed consent before screening.
Participant Screening and Data Collection
Each participant was examined by a physician. All participants were screened for coronary artery disease and stroke risk factors, including hypertension, diabetes, total cholesterol, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein cholesterol, body mass index (BMI), and cigarette smoking.15 Race was self-reported. Height and weight were measured and BMI was calculated.16 Current cigarette smoking was defined by self-report of any smoking within the past month and/or 2 expired carbon monoxide levels of ≥8 ppm. Blood pressure (BP) was measured 3 times during 1 day, and the average systolic and diastolic BP was used to characterize BP according to guidelines of the American Heart Association.17 Hypertension was defined as an average measured BP ≥ 140 mm Hg systolic or 90 mm Hg diastolic and/or use of antihypertensive drugs. Blood for measurements of cholesterol and glucose levels was collected following a 12-hour fast overnight. Type 2 diabetes was defined as a physician-diagnosed history, a fasting glucose level of ≥126 mg/dL, and/or use of hypoglycemic antidiabetic medications. Total cholesterol, high-density lipoprotein, and triglyceride levels were measured according to the United States Centers for Disease Control standardized methods.18 LDL cholesterol was estimated using the Friedewald formula.19
MR Imaging Acquisition
MR imaging was acquired on a 3T Achieva imaging unit (Philips Healthcare, Best, the Netherlands) according to standardized protocols. We acquired the following series: 1) axial T1-weighted MPRAGE—TR, 10 ms; TE, 6 ms; voxel size, 0.75 × 0.75 × 1 mm3; contiguous slices; FOV, 240 × 240 mm; matrix, 320 × 320 mm; 2) axial spin-echo T2 images—TR, 4685 ms; TE, 78 ms; voxel size, 0.47 × 0.47 × 3 mm3; contiguous slices; FOV, 240 × 240 mm; matrix, 512 × 512; 3) DTI—TR, 7043 ms; TE, 71 ms; voxel size, 0.83 × 0.83 × 2.2 mm3; contiguous slices; FOV, 212 × 212 mm; matrix, 256 × 256; 32 directions; b-value = 700 s/mm2.
DTI Processing and Analysis
DTI was preprocessed and analyzed using an MR imaging studio package (DTIstudio, DiffeoMap, ROIEditor) publicly available at www.mristudio.org through Johns Hopkins University. First, all images (T1, T2, DWI) were reviewed by a board-certified physician in neurology and neurocritical care (Y.H.) for the presence of any rotation, motion artifacts, or eddy current artifacts in the DTI studio image viewer application, which was followed by tensor calculation according to the standardized steps in DTIstudio.20 3D FA, mean diffusivity, and radial diffusivity maps were created following tensor calculation.
WM segmentation was performed in the subject's native space according to the Eve WM Atlas in the DifeoMap and ROIEditor package software.21 First, FA and mean B0 maps were skull-stripped in ROIEditor. Following skull-stripping, a dual-channel large deformation diffeomorphic mapping algorithm was used in DifeoMap to perform linear and nonlinear registration to the Eve WM Atlas JHU-MNI-SS-SS template in the Montreal Neurological Institute space. This step allowed automated segmentation of the FA subject map in native space using the inverse registration matrix into 181 regions (On-line Figure A–C). The mean regional FA value (rFA) was calculated in each of the regions by choosing a voxel FA threshold of ≥0.2 to exclude voxels that included CSF and gray matter. Then, 48 regions of the Atlas representing the deep white matter regions primarily affected by CSVD were selected for the subsequent statistical analysis (On-line Table 1 and On-line Figure D–F).
Statistical Analysis
Statistical analysis was performed using SAS software (Version 9.2; SAS Institute, Cary, North Carolina). Univariable analysis comparing the mean rFA between participants with and without current smoking, hypertension, and diabetes was completed using t tests. Spearman rank order correlations were used to test the associations among continuous variables, including: systolic BP, diastolic BP; total cholesterol, triglyceride, high-density lipoprotein cholesterol, LDL cholesterol, and blood glucose levels; and BMI with a mean rFA. The significance threshold was defined using the Bonferroni method for adjustment for multiple comparisons as P ≤ .001 (.05/48 rFA regions). A minimal linear regression model predicting rFA and adjusting for age, sex, and race was performed for the regions that met the Bonferroni significance threshold. Then, to understand the effect of the combination of atherogenic risk factors on rFA, we performed a maximal linear regression model predicting rFA and adjusting for age, sex, race, hypertension, diabetes, total cholesterol level, BMI, and smoking for these regions.
Results
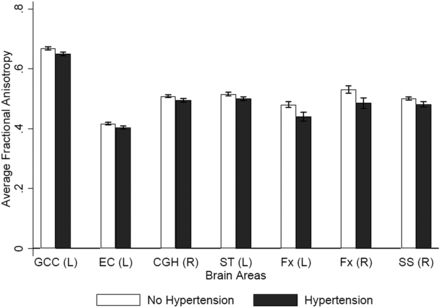
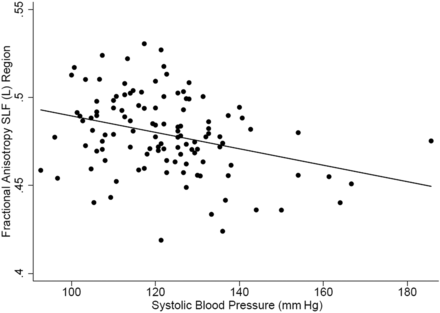
The study sample consisted of 116 middle-aged participants (mean age, 49.6 ± 11 years; 49.1% men; 40.5% African American), well-educated with moderate vascular risk factors (Table 1). Review of T2 sequences did not reveal any stroke or localized WM hyperintensity. WM hyperintensity involving the periventricular and deep WM areas was largely symmetric without predilection to any specific areas of the selected WM regions. Participants with hypertension showed widespread reduced rFA compared with participants with normal BP meeting the Bonferroni threshold (≤0.001) in the left genu of the corpus callosum, left external capsule, right cingulum, left stria terminalis, bilateral fornices, and right sagittal stratum (On-line Table 2 and Figs 1 and 2). This relationship remained significant in the minimal regression model, after adjusting for age, sex, and race, in the right cingulum; left stria terminalis; and right sagittal stratum (Table 2). Additionally, the relationship remained significantly independent of other atherogenic vascular risk factors in the maximal linear regression model in the right cingulum, left stria terminalis, and left fornix (Table 2). The mean rFA correlated with the average systolic BP in the left superior longitudinal fasciculus (On-line Table 3 and Figs 2 and 3). There was a trend toward significance of the relationship of the rFA of this region with hypertension in the minimal regression model (P = .08). However, this relationship was significant in the maximal regression model following adjustment for other vascular risk factors (Table 3).
Baseline characteristics of the study cohort (N = 116)
Bar chart of the regions that showed a significant difference in rFA between subjects with and without hypertension. CGH (R) indicates right cingulum; EC (L), left external capsule; Fx (L), left fornix; Fx (R), right fornix; GCC (L), left genu of the corpus callosum; SS (R), right sagittal stratum; ST (L), left stria terminalis.
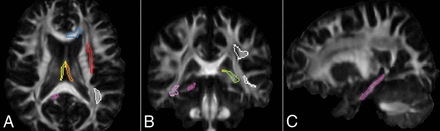
Axial, coronal, and sagittal sections of the FA map, showing areas, in distinct colors, that demonstrate a significant difference of rFA between patients with and without hypertension and areas that show correlation of rFA with systolic blood pressure. Magenta indicates right cingulum; red, left external capsule; orange, left fornix; yellow, right fornix; blue, left genu of the corpus callosum; pink, right sagittal stratum; green, left stria terminalis; white, left superior longitudinal fasciculus.
Minimal and maximal regression models predicting mean rFA of the WM regions that were significantly associated with hypertension in the univariate analysis
Scatterplot showing the relationship of rFA of the left superior longitudinal fasciculus and systolic blood pressure. SLF (L) indicates left superior longitudinal fasciculus.
Minimal and maximal regression models predicting mean rFA of the WM region that were significantly associated with systolic blood pressure in the univariate analysis
Notably, rFA was not significantly associated with diabetes or current smoking status in any of the regions (On-line Tables 4 and 5). Additionally, rFA was not significantly correlated with blood glucose, LDL, high-density lipoprotein, total cholesterol, and triglyceride levels; diastolic BP; and BMI in any of the regions (On-line Tables 6–12).
Discussion
In this cross-sectional study, we show that decreased rFA, reflecting microstructural WM disruption, is strongly associated with hypertension in middle-aged apparently healthy subjects at increased risk for vascular disease in the deep WM regions of the brain. We did not observe additional associations between rFA and other traditional atherogenic vascular risk factors such as diabetes, total cholesterol level, LDL, BMI, or smoking. The role of DTI in detecting WM disruption in subjects with vascular risk factors has been addressed previously.22⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓⇓–39
Hypertension has been consistently associated with decreased FA and elevated mean diffusivity in different WM regions, including normal-appearing WM.24,25,27⇓⇓⇓–31 In addition, increased BP values linearly correlated with decreased FA in the right anterior corpus callosum, inferior fronto-occipital fasciculus, and the fibers that project from the thalamus to the superior frontal gyrus in 1 study.22 Most of these studies have assessed elderly subjects with an average age of 65.8–83 years25,27⇓–29 or investigated the effect of hypertension in middle life on the development of WM disruption at a later age.31 Two previous studies investigated the relationship of systolic BP and WM microstructure at a younger age (39.2–48 years).22,24 One of them linked adiposity as measured by waist circumference with BP and WM microstructure.24 The second article analyzed the systolic BP relationship with regional DTI metrics by measuring the number of voxels within each WM region that correlated with the systolic BP value.22 Our study used a different approach by analyzing the average rFA of a well-defined WM atlas in relation to vascular risk factors. Given the healthy nature of our relatively younger cohort compared with cohorts in the literature, we believe that we captured the disease at an early stage when it had not progressed yet to involve all the subcortical WM in a symmetric fashion. Indeed, our results reflect that by showing a lower rFA of selected WM regions.
Brain DTI of patients with diabetes showed reduced FA using Tract-Based Spatial Statistics (TBSS; http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/TBSS) in patients with mild cognitive impairment compared with those without it.39 A similar analytic method showed a significant decrease in the WM tract DTI metrics in closely related areas to the default mode network in patients with type 2 diabetes compared with healthy controls.38 Finally, the Atherosclerosis Risk in Communities (ARIC) neurocognitive study found a relationship between midlife and late-life elevated blood glucose levels and worse WM microstructural integrity in late life.31 We did not observe a relationship between diabetes or glucose levels and rFA in our cohort. This is likely secondary to the healthy nature of our subjects, who are cognitively intact, younger in age, and with fewer atherogenic risk factors.
In contrast to diabetes and hypertension, the relationship between WM microstructures and other atherogenic vascular risk factors is modest and inconsistent. The ARIC study did not observe a relationship between lipid levels at midlife and late life with late-life DTI metrics.31 However, another study found an association between lipids, especially LDL levels, and WM microstructure integrity in 125 generally healthy older adults (mean age, 68 years).32 BMI was found to be associated with WM microstructure integrity in selected ROIs in a sample of older healthy community participants (mean age, 71.3 years).23 The combination of atherogenic vascular risk factors (≥2) was associated with a longitudinal drop in FA in WM regions compared with no vascular risk factors in elderly subjects (mean age, 73.9 years).25 Finally, smoking was associated with increased FA in WM regions in adolescent/young adult smokers in a recent systematic review.35 Another study of chronic smokers showed decreased FA of the hippocampus in association with decreased memory performance compared with nonsmokers.34
In summary, our study cohort is unique because it tests a relatively young and healthy group of subjects who are relatives of patients with coronary artery disease compared with most previous literature that evaluated elderly subjects, patients who have advanced vascular risk factors, or subjects with cognitive impairments. Our findings indicate hypertension as being the most important risk factor in otherwise healthy subjects at early stages of the disease. The identified WM regions in our study likely play important roles in cognition and memory. The stria terminalis and fornix are critical for normal cognitive functioning and memory.40 Fornix pathology can be found in Alzheimer disease. The cingulum is known to play a role in executive control, emotion, and episodic memory, and it has been implicated in multiple psychiatric diseases and Alzheimer disease.41 The superior longitudinal fasciculus abnormalities on DTI have been attributed to age-related language decline.42 Finally, the sagittal stratum carries multiple functional fibers implicated in language and visual function.43
Despite available animal and human data suggesting a mechanistic relationship of hypertension to CSVD, the underlying processes leading to these events are yet to be established.4 Arterial stiffness has been closely linked to hypertension and aging. Arterial stiffness results from remodeling of the arterial wall secondary to degradation of elastin and accumulation of collagen and calcium deposition.7 Measures of arterial stiffness such as the ankle-brachial pressure index have been associated with the development of CSVD findings.8,9 Additionally it is well-known that hypertension is associated with arteriosclerosis, which is the main pathologic feature of CSVD, characterized by fibrohyaline material, narrowing of the lumen, and thickening of the vessel wall.2 Moreover, hypertension leads to impaired cerebral autoregulation, which results in cerebral hypoxia and WM injury.44 It is plausible that hypertension plays an important role in early WM damage compared with other atherogenic risk factors in middle-aged apparently healthy subjects.45 Recently, an alternative pathway involving disruption of the blood-brain barrier leading to CSVD has been heavily investigated.4,44 The exact relationship of hypertension to BBB dysfunction needs to be thoroughly investigated in the future.
Our study has certain limitations, and it should be interpreted within its context. First, given the cross-sectional nature of the study, a mechanistic relationship between the atherogenic vascular risk factors and WM microstructure disruption cannot be ascertained in our sample. Second, more regions of deep WM structures could have been involved in this disease; however, they were not detected given the relatively small sample size. We decided to choose a conservative method for adjustment for multiple comparisons (Bonferroni method) despite the distributed nature of the CSVD in the brain because we aimed to avoid any possibility of finding false-positive results. In addition, an interaction analysis among vascular risk factors and associated WM damage could not be performed due to the relatively small sample size. Finally, we did not examine the severity of hypertension and blood pressure control at baseline to assess its relationship to the severity of WM disease.
Conclusions
In apparently healthy middle-aged subjects at increased risk for vascular disease, among traditional risk factors, hypertension is most closely associated with microstructural WM impairment. These findings have implications for patient care by early selection of subjects who are at risk for future cognitive impairment through identification of these impaired tracts. This may even prove to be of further value because novel therapies are being developed for patients with CSVD.
Footnotes
Disclosures: Lisa R. Yanek—RELATED: Grant: National Institutes of Health, Comments: NS062059 from the National Institutes of Health/National Institute of Neurological Disorders and Stroke to Dr Paul Nyquist. Dhananjay Vaidya—RELATED: Grant: National Institutes of Health.* Paul Nyquist—RELATED: Grant: National Institutes of Health.* *Money paid to the institution.
This work was supported by the National Institutes of Health/National Institute of Neurological Disorders and Stroke grant NS062059; and the National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health, to the Johns Hopkins Institute for Clinical and Translational Research, grant UL1RR025005.
Paper previously presented at: International Stroke Conference, February 22–24, 2017; Houston, Texas.
Indicates open access to non-subscribers at www.ajnr.org
References
- Received February 26, 2018.
- Accepted after revision September 21, 2018.
- © 2018 by American Journal of Neuroradiology