Abstract
BACKGROUND AND PURPOSE: Transradial access for neurointerventional procedures has been proved a safer and more comfortable alternative to femoral artery access. We present our experience with transradial (distal radial/anatomic snuffbox and radial artery) access for treatment of intracranial aneurysms using all 3 FDA-approved flow diverters.
MATERIALS AND METHODS: This was a high-volume, dual-center, retrospective analysis of each institution’s data base between June 2018 and June 2020 and a collection of all patients treated with flow diversion via transradial access. Patient demographic information and procedural and radiographic data were obtained.
RESULTS: Seventy-four patients were identified (64 female patients) with a mean age of 57.5 years with a total of 86 aneurysms. Most aneurysms were located in the anterior circulation (93%) and within the intracranial ICA (67.4%). The mean aneurysm size was 5.5 mm. Flow diverters placed included the Pipeline Embolization Device (Flex) (PED, n = 65), the Surpass Streamline Flow Diverter (n = 8), and the Flow-Redirection Endoluminal Device (FRED, n = 1). Transradial access was successful in all cases, but femoral crossover was required in 3 cases (4.1%) due to tortuous anatomy and inadequate support of the catheters in 2 cases and an inability to navigate to the target vessel in a patient with an aberrant right subclavian artery. All 71 other interventions were successfully performed via the transradial approach (95.9%). No access site complications were encountered. Asymptomatic radial artery occlusion was encountered in 1 case (3.7%).
CONCLUSIONS: Flow diverters can be successfully placed via the transradial approach with high technical success, low access site complications, and a low femoral crossover rate.
ABBREVIATIONS:
- CCA
- common carotid artery
- dRA
- distal radial artery
- FD
- flow diverter
- RA
- radial artery
- TRA
- transradial access
The transradial access (TRA), including distal radial artery (dRA) access in the anatomic snuffbox and radial artery (RA) access at the palmar surface of the wrist, is being increasingly used as primary vascular access for neurointerventional procedures. In prior years, large randomized trials in the field of interventional cardiology and more recent articles in neurointerventional surgery have shown higher patient preference for the TRA, cost reduction, as well as lower morbidity and mortality compared with the traditional transfemoral access (TFA).1⇓⇓⇓⇓⇓⇓⇓⇓⇓-11 Reduction in access site complications has been a particular advantage of wrist over femoral access and is an important consideration for vascular access choice in the treatment of intracranial aneurysms using flow diversion. Patients undergoing flow diversion are required to take dual-antiplatelet agents and receive heparin during the procedure, all of which increase the risk of bleeding from the access site.12 Also, flow diverters (FDs) may require large-bore catheter assemblies for delivery and deployment, which may increase the risk of radial artery occlusion, access site bleeding, or vascular injury.13,14
To date, only a limited number of case reports and case series have described the safety and feasibility of TRA for the treatment of intracranial aneurysms using flow diverters.15⇓⇓⇓⇓⇓⇓-22
Recently, a large, retrospective multicenter study reported the safety of TRA for flow diversion, showing a lower access site (P = .039) and overall complication rate (P = .035).12 This study, however, did not cover catheter systems, patient functional outcome, and aneurysm occlusion. Here, we report our experience with TRA (dRA [anatomic snuffbox] and RA) for the treatment of intracranial aneurysms using all 3 FDA-approved flow diverters, including technical feasibility, procedural safety, patient outcome, and aneurysm occlusion on follow-up. Additionally, we reviewed the current literature on use of flow diverters via TRA.
MATERIALS AND METHODS
Study Design
Between June 2018 and June 2020, we retrospectively reviewed the data bases from 2 high-volume neurointerventional centers that routinely perform radial access in the United States and collected all patients treated with flow diversion via TRA. Patient demographic information and procedural and radiographic data were also obtained. The institutional review boards at the University of Massachusetts (H00001860_10) and Christiana Health System (CCC number: 34154 and DDD number: 602798) have approved the study.
Procedural Protocol and Technique
All patients received dual-antiplatelet therapy for at least 5 days before the elective procedure. The therapeutic antiplatelet effect was ensured by the VerifyNow P2Y12 assay (Accumetrics) (defined per our practice as showing at least 50% platelet inhibition or a P2Y12 reaction unit of <200 with the platelet aggregation assay) before each procedure. Dual-antiplatelet therapy was continued for a minimum of 6 months, and aspirin will be continued for life.
With the exception of 1 left RA access for treatment of a left vertebral artery dissecting aneurysm, all interventions were performed either via the right dRA in the anatomic snuffbox or right RA at the palmar surface of the wrist. After local subcutaneous anesthesia and vasodilation of the local periarterial tissue using 1 mL of lidocaine and 200–400 µg of nitroglycerine, the dRA or RA was accessed using a micropuncture needle under sonographic guidance. A 6F radial sheath was placed, and a radial cocktail consisting of verapamil, 2.5–5 mg; nitroglycerine, 100–200 µg; and heparin, 5000 IU, was slowly administered intra-arterially. In sheathless access cases, the guide catheter was directly inserted over its dilator. In patients with taut skin, a small incision with a scalpel at the access site was occasionally needed. Another dose of spasmolytic agents was administered through the radial sheath before catheter exchanges to prevent vasospasm. Activated clotting time was measured at baseline and throughout the procedure with activated clotting time goals of 250–300 seconds. Additional IV doses of heparin were administered as needed. A radial artery roadmap was obtained in all cases.
In most cases, the flow diverters were delivered and deployed via a triaxial catheter system. Most Pipeline Embolization Devices (PED; Medtronic) were deployed using a Benchmark guide catheter (Penumbra) or a Phenom Plus intermediate catheter or Phenom 27 microcatheter (Medtronic). The Surpass Streamline Flow Diverter (Stryker) was placed using either an AXS Infinity (Stryker) or Fubuki (Asahi Intecc) guide catheter and a AXS Catalyst 5 intermediate catheter (Stryker) as well as an AXS Offset Delivery Assist catheter (Stryker). The Flow-Redirection Endoluminal Device (FRED; MicroVention) was implanted using a Cerebase DA Guide Sheath (Cerenovus), a Sofia EX Intracranial Support Catheter (MicroVention), and a Headway 27 microcatheter (MicroVention). On completion of the procedure, a radial compression device was used to achieve patent hemostasis at the vascular access site.
RESULTS
Between June 2018 and June 2020, a total of 144 flow-diverter procedures were performed. We identified a total of 74 flow-diverter interventions performed via the anatomic snuffbox or radial artery access (51.4%). Sixty-four patients were women (86.5%). The mean patient age was 57.5 years. Mean distal radial and radial artery diameters were 2.1 and 2.6 mm, respectively.
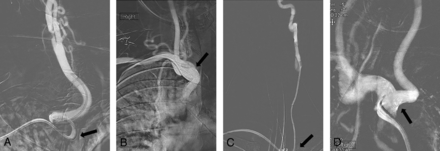
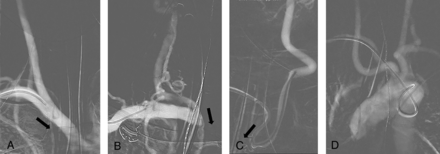
Transradial access was achieved in all cases with anatomic snuffbox access in 42 cases (56.8%) and radial artery access in 32 cases (43.2%). However, 3 cases (4.1%) required conversion to femoral access due to tortuous anatomy and limited support of the catheters in 2 cases and an inability to navigate the catheters to the target vessel in a patient with an aberrant right subclavian artery. All of the other 71 interventions were successfully performed via a transradial approach. We did not observe any access-related complications, and no patients required a transfusion or vascular surgical repair for an access site injury. Figure 1 shows arches amenable to transradial intervention, and Fig 2 shows arch types that are difficult to navigate via transradial access.
Frontal roadmap images show a curved origin of the right CCA at the junction of the innominate artery and right subclavian artery origin (arrows, A and B). This anatomy offers a “shelf” for right transradial catheters, which provides good stability, even with a tortuous course of the right CCA (A). Frontal roadmap image demonstrates a straight origin of the left CCA from the aortic arch (C, arrow), which allows good navigation of a right transradial catheter. The transverse portion of the aortic arch may act as a shelf for transradial catheters during intervention, providing good stability for an intervention. Frontal roadmap image shows a bovine-type aortic arch with horizontal origin of the left CCA (D, arrow). Access of the left CCA would be easy from a right transradial approach because the catheter can easily move across midline and into the vessel. There would be no risk of catheter herniation.
Frontal roadmap image reveals very straight anatomy of the innominate, right subclavian, and common carotid arteries (A). Catheter navigation from a right transradial access is not easy, given that the vector force will push the catheter into the right innominate artery (A, arrow). Frontal roadmap image shows a fairly horizontal course of the right subclavian artery and a straight continuation of the right innominate artery into the right CCA (B). Here, the vector force will push a right transradially navigated catheter into the innominate artery (B, arrow), making catheterization of the right intracranial vasculature difficult. Frontal roadmap image demonstrates a medially directed origin of the left CCA from the aortic arch. This makes access from a right radial approach difficult because the catheter will be pushed into the ascending aorta (C, arrow). Additional tortuosity along the course of the left CCA increases the difficulty of the approach. Frontal roadmap image shows an aberrant origin of the right subclavian artery (D). Navigation of a right transradial catheter is difficult, given that the catheter is directed toward the descending aorta. Larger catheter systems will not have sufficient stability, and catheter herniation into the descending aorta is to be expected.
A total of 86 aneurysms were treated, with most aneurysms located within the intracranial ICA (67.4%). One aneurysm was ruptured, and 4 were previously coiled but showed neck residual/recanalization. The mean aneurysm size was 5.5 mm. A summary of patient demographics and aneurysm characteristics is presented in the Table.
Summary of patient demographics and aneurysm characteristics
A PED was used in 65 cases, a Surpass Streamline Flow Diverter was placed in 8 patients, and 1 FRED was implanted. Additional coiling and placement of a Neuroform EZ stent (Stryker) for proper FD wall apposition were performed in 1 case for treatment of a large, 16-mm ICA aneurysm.
We experienced 3 intraprocedural complications (4.2%). Two patients showed a small amount of SAH on postprocedural head CT (2.8%). One of these patients had mild headache but was neurologically intact. The other patient showed new right-arm weakness on awakening from anesthesia and was brought back to the angiography suite where hyperacute platelet aggregation within the stent was identified and successfully treated with intra-arterial eptifibatide. The patient fully recovered within 6 months with an mRS of 0. In another case, we encountered acute clot formation during placement of the flow diverter, which was immediately treated with intra-arterial eptifibatide.
Of our 71 patients with transradial access, 1 patient died due to an unrelated cause, 3 patients moved out of state, and 1 patient requested follow-up at an outside institution. Ten patients were supposed to have follow-up, but this has been delayed due to institutional restrictions during the coronavirus disease 2019 (COVID-19) pandemic, and 14 patients have not yet reached their follow-up time point. Six patients were lost to follow-up.
Two- to 6-month follow-up angiograms and CTAs were available in 29 and 7 patients, respectively. Twenty-seven of the 29 angiogram follow-ups were performed transradially. Follow-up diagnostic angiograms were performed via a 5F radial sheath. Spasmolytic cocktails used for transradial access were the same as for the procedure. There were no conversions from initial anatomic snuffbox access to volar radial artery access for follow-up. In 3 cases, radial (volar) access was used for the intervention, but the follow-up angiogram was performed via anatomic snuffbox access. Asymptomatic radial artery occlusion was encountered in 1 case (3.7%). The initial procedure in this patient was performed via the radial artery (volar access) with a spasmolytic cocktail consisting of 2.5 mg of verapamil and nitroglycerin, 100 mcg. Follow-up angiography in this patient was performed via the ulnar artery. One patient requested femoral access for the follow-up angiography. There was otherwise no contraindication to wrist access in this patient.
Complete and near-complete occlusion was seen in 27 of 43 aneurysms (62.8%). One-year follow-up (n = 5) showed progression to complete occlusion in 2 cases. Additional procedural information and follow-up data are summarized in the Online Supplemental Data.
DISCUSSION
The TRA approach for interventional cardiology procedures has been used since 1989.23,24 Several large cardiology studies provided strong evidence in favor of wrist over femoral access, including decreased morbidity and mortality, reduced cost, better control of access site hemostasis, reduction in vascular complications, as well as higher patient satisfaction due to less postprocedural discomfort and faster recovery.6,25⇓⇓⇓⇓-30 In the neurointerventional field, TRA is now beginning to gain popularity, and some reports on the feasibility and safety of this approach for diagnostic angiographies31⇓-33 and a variety of neurointerventions are available.10,11,21,34⇓⇓-37 The first case reports on the use of TRA for flow diversion published between 2013 and 2017 were mostly on patients with difficult arch anatomy.15⇓-17 In 2019, the first case series on treatment of consecutive patients with aneurysms exclusively evaluated the use of the PED via TRA.18,19,22 With a total of 71 successful interventions, our study is currently the largest series to evaluate TRA for intracranial aneurysm treatment with all FDA-approved flow diverters and also includes information on catheter systems, patient outcome, and aneurysm occlusion. In addition, our access conversion rate of only 4.1% is the lowest reported in the literature of transradial access for flow diversion to date. Previously reported conversion rates ranged between 5.7% and 20.4%.12,18,19,22
A stable catheter assembly is crucial to successfully deliver and deploy the flow diverters because their braided configuration and stiffness require a substantial forward-loading force. Lack of catheter support and radial artery vasospasm are the most commonly described reasons for TRA failure. Prior case series primarily reported failed TRA interventions for left-sided interventions,18,19,22 but we experienced an inability to achieve a stable catheter position during catheterization of the right common carotid artery (CCA) and ICA in a patient with a type III arch. Left-sided interventions are generally regarded as more challenging; however, we believe that with certain anatomic configurations, right-sided interventions may be just as challenging. The course of the right subclavian artery, its angle of origin from the innominate artery, and the angle of origin of the innominate artery itself from the aortic arch are important for catheter navigation and steerability. The more tortuosity and steep angles that are present, the less one-to-one motion and catheter control are experienced.
Aortic arch configuration, great vessel takeoff angles, and proximal CCA tortuosity influence catheter stability. For left-sided interventions, we agree with Waqas et al,22 who stated that a parallel configuration of the innominate artery and left CCA is challenging. However, we would argue that the distance between the origins of the innominate artery and the left CCA, their relationship to the ascending and descending aorta, the arch configuration, and its capaciousness are also important factors to consider. For right-sided interventions, a steep origin of the innominate artery from the arch, which continues into a straight right CCA, as well as a straight, wide-angle origin of the right subclavian artery represent a challenging anatomic configuration. We are presently still trying to understand what anatomy or combination of anatomic configurations can truly be considered unfavorable and what catheter systems are going to be successful in which kind of situations.
Chen et al19 reported 2 cases of radial artery vasospasm in their study, which used biaxial, triaxial, and even quadriaxial catheter systems. Biaxial and triaxial systems were used in the studies reported by Sweid et al18 and Waqas et al,22 who did not encounter any case of radial artery vasospasm. In our large case series, we also did not encounter any radial artery vasospasm requiring access conversion despite using triaxial systems, even with large-bore catheters. We strongly believe that additional doses of antispasmolytic agents during catheter exchanges are crucial to minimize the occurrence of radial artery vasospasm. We did not observe any access site complications, persistent neurologic deficits, or procedure-related deaths. Two patients developed SAH (2.8%), with one of these patients also experiencing stroke symptoms, which completely resolved within 6 months postprocedure. Waqas et al22 reported 1 death (3%) in their study. Major ipsilateral stroke or neurologic death was reported in 5.6% of patients undergoing PED deployment via traditional femoral artery access in the Pipeline Embolization Device for Uncoilable or Failed Aneurysms (PUFS) trial.38 Headache or intracerebral hemorrhage was seen in 4.7% each, and ischemic stroke, in 3.7%.38
Routine sonographic follow-up evaluation of the RA was not performed, but there was no symptomatic RA occlusion. On follow-up, 1 of 27 patients was found to have an asymptomatic RA occlusion (3.7%). RA occlusion can be seen in 0.04%–10% of cases after radial access.13,19,39⇓⇓⇓⇓⇓⇓-46 Most RA occlusions are asymptomatic due to collateral circulation from the ulnar artery. Another very rare complication of TRA is forearm compartment syndrome, which was reported in 1 of 9681 cases in an interventional cardiology study.45 This complication can be avoided by accessing the radial artery more proximally, not deep to the brachioradialis muscle, or by puncturing the dRA, which lies beyond the forearm compartment.46
Limitations of a 6F Benchmark triaxial system include limited ability to obtain angiograms and roadmaps after the microcatheter is advanced secondary to being able to inject only the intermediate catheter. Using a 5F Sofia 115-cm Intermediate Catheter (MicroVention) allows slightly larger inner diameter and better angiograms than a Phenom Plus. Additionally, jailing and coil-assisted flow-diverting stent placement preclude the use of an intermediate catheter, which can greatly affect column strength during stent delivery.
This study has limitations. Given its dual-center nature, different clinical settings, patient-selection bias, and technical/procedural variations were inevitable. However, patients benefited from TRA for flow diversion despite this heterogeneity. The study is further limited by the retrospective review of each center’s prospective data base, and although our sample size is relatively large, more data (specifically long-term follow-up) are needed.
Nevertheless, our experience is promising and shows that TRA is a feasible and safe approach to treat a wide range of intracranial aneurysms with flow diversion. The improved TRA safety profile is undeniable and will benefit a large variety of patients, especially those with a large body habitus, previous iliofemoral artery stent placement or bypass, aortic dissections, heavily calcified pelvic vasculature, and femoral artery occlusions.
CONCLUSIONS
This study demonstrates the feasibility and safety of TRA for flow diversion to treat a broad range of anterior and posterior circulation intracranial aneurysms. Currently available catheters were originally designed for traditional femoral artery access interventions, but dedicated catheter systems for TRA are being developed. Catheters specifically tailored to traditional femoral artery access will facilitate vascular access and permit navigation of standard as well as challenging anatomy. Future technical advances, including dedicated guide catheters and smaller stent delivery systems, will inevitably decrease femoral conversion rates. Future research focusing on favorable and unfavorable anatomy will also be important to further aid in patient selection and decrease the need for access conversion.
Footnotes
Disclosures: Sudhakar R. Satti—RELATED: Consulting Fee or Honorarium: Medtronic; Other: Medtronic, Pipeline proctor.* Matthew J. Gounis—UNRELATED: Consultancy: Astrocyte Pharmaceuticals, Cerenovous, Imperative Care, Medtronic, MIVI Neurosciences, phenox, Q’Apel, Route 92 Medical, Stryker Neurovascular, Consultancy: Wallaby Medical, Comments: fee-per-hour consulting; Grants/Grants Pending: National Institutes of Health, Israel Binational Science Foundation, Anaconda, ApicBio, Arsenal Medical, Axovant, Cerenovus, Ceretrieve, Cook Medical, Galaxy Therapeutics, Gentuity, Imperative Care, InNeuroCo, Insera, Magneto, MicroVention, Medtronic, MIVI Neurosciences, Naglreiter MDDO, Neurogami, Omniox, Philips Healthcare, Progressive Medical, Pulse Medical, Rapid Medical, Route 92 Medical, Stryker Neurovascular, Syntheon, ThrombX Medical, the Wyss Institute, Xtract Medical*; Stock/Stock Options: Galaxy Therapeutics, Imperative Care, InNeuroCo, Neurogami. Ajit Puri—RELATED: Consulting Fee or Honorarium: proctor for Stryker, Cerenovus, Medtronic, and MicroVention; UNRELATED: Consultancy: consultant for Q’Apel, Merit Medical, Arsenal medical; Grants/Grants Pending: small business innovation research, National Institutes of Health*; Payment for Lectures Including Service on Speakers Bureaus: Merit Medical, Cerenovus, Q’Apel; Stock/Stock Options: InNeuroCo, Galaxy therapeutics, Neurotherapeutics Inc., Agile Medical, Perfuze. *Money paid to the institution.
References
- Received August 13, 2020.
- Accepted after revision October 8, 2020.
- © 2021 by American Journal of Neuroradiology