Abstract
Background
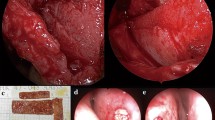
Septal cartilage still is the most appropriate graft material used in rhinoplasty. In traumatic or revision cases, septal cartilage can be insufficient, and the need for an implant material emerges. In this study, the safety and efficacy of polytetrafluoroethylene (Gore-Tex) used as an implant material in nasal dorsal augmentation were assessed.
Methods
This study retrospectively reviewed 74 patients who underwent nasal dorsal augmentation with Gore-Tex. Of the 74 patients who underwent rhinoplasty with Gore-Tex for dorsal augmentation from February 1999 to January 2006, 46 (62.2%) represented primary cases and 28 (37.8%) represented revision cases. The patients were followed from 5 to 62 months (average, 28 months) and questioned about cosmetic and functional outcomes. The results were assessed according to patients’ charts as well as preoperative and postoperative photographic documentation.
Results
There were no complications such as infections, foreign body reaction, extrusions, resorption, or migration, and all the patients were satisfied with their results. Only one case of soft tissue reaction was observed, which lasted for 3 weeks postoperatively.
Conclusions
Biocompatibility, ease of use, lack of extrusion or resorption, and low rate of infection make Gore-Tex a good alternative to autogenous tissue. However, long-term success and complication rates still are lacking, and large numbers of patients with longer follow-up periods are required.


Similar content being viewed by others
References
Beekhuis GJ: Polyamide mesh used in facial plastic surgery. Arch Otolaryngol 106:642–644, 1980
Brown BL, Neel HB III, Kern EB: Implants of Supramid, Proplast, Plasti-Pore, and Silastic. Arch Otolaryngol 105:605–609, 1979
Conrad K, Gillman G: A 6-year experience with the use of expanded polytetrafluoroethylene in rhinoplasty. Plast Reconstr Surg 101:1675–1684, 1998
Davis PK, Jones SM: The complications of silastic implants: Experience with 137 consecutive cases. Br J Plast Surg 24:405–411, 1971
Godin MS, Waldman SR, Johnson CM Jr: Nasal augmentation using Gore-Tex: A 10-year experience. Arch Facial Plast Surg 1:118–121, 1999
Gore-Tex Soft Tissue Patch: Technical Bulletin. WL Gore & Assoc Inc.: Flagstaff, AZ, August 1994
Maas CS, Merwin GE, Wilson J, et al: The use of expanded polytetrafluoroethylene (Gore-Tex) in rhinoplasty: A 6-year experience. Arch Otolaryngol Head Neck Surg 121:113–116, 1995
Neel HB III: Implants of Gore-Tex. Arch Otolaryngol 109:427–433, 1983
Owsley TG, Taylor CO: The use of Gore-Tex for nasal augmentation: A retrospective analysis of 106 patients. Plast Reconstr Surg 94:241–250, 1994
Schuller DE, Bardach J, Krause CJ: Irradiated homologous costal cartilage for facial contour restoration. Arch Otolaryngol 103:12–15, 1977
Singh S, Baker JL Jr: Use of expanded polytetrafluoroethylene in aesthetic surgery of the face. Clin Plast Surg 27:579–593, 2000
Soyer T, Lempinen M, Cooper P, et al: A new venous prosthesis. Surgery 72:864–872, 1972
Stucker FJ: Use of implantation in facial deformities. Laryngoscope 87:1523–1527, 1977
Welling DB, Maves MD, Schuller DE, Bardach J: Irradiated homologous cartilage grafts: Long-term results. Arch Otolaryngol Head Neck Surg 114:291–295, 1988
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Inanli, S., Sari, M. & Baylancicek, S. The Use of Expanded Polytetrafluoroethylene (Gore-Tex) in Rhinoplasty. Aesth Plast Surg 31, 345–348 (2007). https://doi.org/10.1007/s00266-007-0037-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-007-0037-z