Abstract
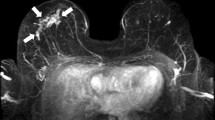
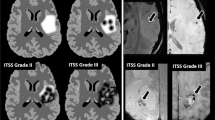
Radiation to the brain and adjuvant chemotherapy may produce late delayed changes from several months to years after treatment of intracranial malignancies with a reported prevalence of 5–24%. The pattern of treatment-related injury may vary from diffuse periventricular white matter lesions to focal or multifocal lesions. Differentiation of treatment-related injury from tumor progression/recurrence may be difficult with conventional MR imaging (MRI). With both disease processes, the characteristic but nonspecific imaging features are vasogenic edema, contrast enhancement, and mass effect. This pictorial essay presents MRI spectra of late therapy-induced injuries in the brain with a particular emphasis on radiation necrosis, the most common and severe form. Novel MRI techniques, such as diffusion-weighted imaging (DWI), proton MR spectroscopy (MRS), and perfusion MRI, improve the possibilities of better characterization of treatment-related changes. Advanced MRI techniques allow for the assessment of metabolism and physiology and may increase specificity for therapy-induced changes.







Similar content being viewed by others
References
Sheline GE, Wara WM, Smith V (1980) Therapeutic irradiation and brain injury. Int J Radiat Oncol Biol Phys 6:1215–1228
Ruben JD, Dally M, Bailey M, Smith R, McLean CA et al (2006) Cerebral radiation necrosis: incidence, outcomes, and risk factors with emphasis on radiation parameters and chemotherapy. Int J Radiation Oncology Biol Phys 65:499–508
Johannesen TB, Lien HH, Hole KH, Lote K (2003) Radiological and clinical assessment of long-term brain tumour survivors after radiotherapy. Radiother Oncol 69:169–176
Marks JE, Baglan RJ, Prassad SC, Blank WF (1981) Cerebral radionecrosis: incidence and risk in relation to dose, time, fractionation and volume. Int J Radiat Oncol Biol Phys 7:243–252
Perry A, Schmidt RE (2006) Cancer therapy-associated CNS neuropathology: an update and review of the literature. Acta Neuropathol 111:197–212
Langleben DD, Segall GM (2000) PET in differentiation of recurrent brain tumor from radiation injury. J Nucl Med 41:1861–1867
Spaeth N, Wyss MT, Weber B, Scheidegger S et al (2004) Uptake of 18F-fluorocholine, 18F-fluoroethyl-L-tyrosine, and 18F-FDG in acute cerebral radiation injury in the rat: implications for separation of radiation necrosis from tumor recurrence. J Nucl Med 45:1931–1938
Sugahara T, Korogi Y, Tomiguchi S, Shigematsu Y, Ikushima I et al (2000) Posttherapeutic intraaxial brain tumor: the value of perfusion-sensitive contrast-enhanced MR imaging for differentiating tumor recurrence from nonneoplastic contrast-enhancing tissue. AJNR Am J Neuroradiol 21:901–909
Morris JG, Grattan-Smith P, Panegyres PK, O’Neill P, Soo YS, Langlands AO (1994) Delayed cerebral radiation necrosis. Q J Med 87:119–129
Burger PC, Mahley MS Jr, Dudka L, Vogel FS (1979) The morphologic effects of radiation administered therapeutically for intracranial gliomas: a postmortem study of 25 cases. Cancer 44:1256–1272
Kumar AJ, Leeds NE, Fuller GN, Tassel PV, Maor MH et al (2000) Malignant gliomas: MR imaging spectrum of radiation therapy- and chemotherapy-induced necrosis of the brain after treatment. Radiology 217:377–384
Schultheiss TE, Kun LE, Ang KK, Stephens LC (1995) Radiation response of the central nervous system. Int J Radiat Oncol Biol Phys 31:1093–1112
Burger PC, Boyko OB (1991) The pathology of central nervous system radiation injury. In: Gutin PH, Leibel SA, Sheline GE (eds) Radiation injury to the nervous system. Raven, New York, pp 191–208
Burger PC, Scheithauer BW (2007) Tumors of the central nervous system. American Registry of Pathology, Washington
Ironside JW, Moss TH, Louis DN, Lowe JS, Weller RO (2002) Diagnostic pathology of nervous system tumours. Churchill Livingstone, New York
Belka C, Budach W, Kortmann RD, Bamberg M (2001) Radiation induced CNS toxicity-molecular and cellular mechanisms. Br J Cancer 85:1233–1239
Lampert PW, Davis RL (1964) Delayed effects of radiation on the human central nervous system: “early” and “late” delayed reactions. Neurology 14:912–917
Sugahara T, Korogi Y, Kochi M, Ikushima I, Shigematu Y, Hirai T et al (1999) Usefulness of diffusion-weighted MRI with echoplanar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 9:53–60
Asao CH, Korogi Y, Kitajima M, Hirai T, Baba Y et al (2005) Diffusion-weighted imaging of radiation-induced brain injury for differentiation from tumor recurrence. AJNR Am J Neuroradiol 26:1455–1460
Kono K, Inoue Y, Nakayama K et al (2001) The role of diffusion-weighted imaging in patients with brain tumors. AJNR Am J Neuroradiol 22:1081–1088
Hayashida Y, Hirai T, Morishita S, Kitajima M, Murakami R et al (2006) Diffusion-weighted imaging of metastatic brain tumors: comparison with histologic type and tumor cellularity. AJNR Am J Neuroradiol 27:1419–1425
Hein PA, Eskey CJ, Dunn JF, Hug EB (2004) Diffusion-weighted imaging in the follow-up of treated high-grade gliomas: tumor recurrence versus radiation injury. AJNR Am J Neuroradiol 25:201–209
Zeng QS, Li CF, Liu H, Zhen JH, Feng DC (2007) Distinction between recurrent glioma and radiation injury using magnetic resonance spectroscopy in combination with diffusion-weighted imaging. Int J Radiation Oncology Biol Phys 68:151–158
Tung GA, Evangelista P, Rogg JM, Duncan JA (2001) Diffusion-weighted MR imaging of rim-enhancing brain masses: is markedly decreased water diffusion specific for brain abscess? AJR Am J Roentgenol 177:709–712
Le Bihan D, Douek P, Argyropoulou M (1993) Diffusion and perfusion magnetic resonance imaging in brain tumors. Top Magn Reson Imaging 5:25–31
Moritani T, Ekholm S, Westesson P-L (2004) Diffusion-weighted MR imaging of the brain. Springer Verlag, Berlin Heidelberg New York
Ebisu T, Tanaka C, Umeda M et al (1996) Discrimination of brain abscess from necrotic or cystic tumors by echo planar imaging. Magn Reson Imaging 14:1113–1116
Fredman DP, Goldman HW, Flanders AE (1997) MR imaging of stereotaxic palidotomy and thalamotomy. AJR Am J Roentgenol 169:894–896
Tien RD, Flesberg GJ, Friedman H, Brown M, MacFall J (1994) MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echoplanar pulse sequences. AJR Am J Roentgenol 162:671–677
Hakyemez B, Erdogan C, Yildirim N, Parlak M (2005) Glioblastoma multiforme with atypical diffusion-weighted MR findings. Br J Radiol 78:989–992
Aronen HJ, Gazit IE, Louis DN et al (1994) Cerebral blood volume maps of gliomas: comparison with tumor grade and histologic findings. Radiology 191:41–51
Covarrubias DJ, Rosen BR, Lev MH (2004) Dynamic magnetic resonance perfusion imaging of brain tumors. Oncologist 9:528–537
Cha S, Knopp EA, Johnson G, Wetzel SG, Litt AW, Zagzag D (2002) Intracranial mass lesions: dynamic contrast-enhanced susceptibility-weighted echo-planar perfusion MR imaging. Radiology 223:11–29
Cotton F, Hermier M (2006) The advantage of high relaxivity contrast agents in brain perfusion. Eur Radio1 Suppl 16(Suppl 7):M 16–M 26
Wong JC, Provenzale JM, Petrella JR (2000) Perfusion MR imaging of brain neoplasms. AJR 174:1147–1157
Sugahara T, Korogi Y, Kochi M et al (1998) Correlation of MR imaging-determined cerebral blood volume maps with histologic and angiographic determination of vascularity of gliomas. AJR Am J Roentgenol 171:1479–1486
Aksoy FG, Lev MH (2000) Dynamic contrast- enhanced brain perfusion imaging: technique and clinical applications. Semin Ultrasound CT MR 21:462–477
Sorensen AG, Reimer P (2000) Cerebral MR perfusion imaging. Thieme, Stuttgart
Kimura T, Sako K, Tohyama Y, Aizawa S, Yoshida H et al (2003) Diagnosis and treatment of progressive space-occupying radiation necrosis following stereotactic radiosurgery for brain metastasis: value of proton magnetic resonance spectroscopy. Acta Neurochir (Wien) 145:557–564
Ge Y, Law M, Johnson G et al (2005) Dynamic susceptibility contrast perfusion MR imaging of multiple sclerosis lesions: characterizing hemodynamic impairment and inflammatory activity. AJNR Am J Neuroradiol 26:1539–1547
Chong VF-H, Rumpel H, Fan Y-F, Mukherji SK (2001) Temporal lobe changes following radiation therapy: imaging and proton MR spectroscopic findings. Eur Radiol 11:317–324
Edelman RR, Hesselink JR, Zlatkin MB, Crues JV (2006) Clinical magnetic resonance imaging. Saunders, Philadelphia
Hollingworth W, Medina LS, Lenkinski RE, Shibata DK, Bernal B et al (2006) A systematic literature review of magnetic resonance spectroscopy for the characterization of brain tumors. AJNR 27:1404–1411
Lichy MP, Plathow CH, Schulz-Ertner D, Kauczor HU, Schlemmer H-P (2005) Follow-up gliomas after radiotherapy: 1H MR spectroscopic imaging for increasing diagnostic accuracy. Neuroradiology 47:826–834
Kimura T, Sako K, Tanaka K, Gotoh T, Yoshida H et al (2004) Evaluation of the response of metastatic brain tumors to stereotactic radiosurgery by proton magnetic resonance spectroscopy, 201TICI single-photon emission computerized tomography, and gadolinium-enhanced magnetic resonance imaging. J Neurosurg 100:835–841
Lichy MP, Henze M, Plathow C et al (2004) Metabolic imaging to follow stereotactic radiation of gliomas—the role of 1H MR spectroscopy in comparison to FDG-PET and IMT-SPECT. Rofo 176:1114–1121
Ando K, Ishikura R, Nagami et al (2004) Usefulness of Cho/Cr ratio in proton MR spectroscopy for differentiating residual/recurrent glioma from non-neoplastic lesions. Nippon Igaku Hoshasen Gakkai Zasshi 64:121–126
Schlemmer HP, Bachert P, Herfarth K, Zuna I, Debus J et al (2001) Proton MR spectroscopic evaluation of suspicious brain lesions after stereotactic readiotherapy. AJNR Am J Neuroradiol 22:1316–1324
Sundgren PC, Mukherji SK (2004) MRS applications in oncology. In: Imaging Economics. http://www.imagingeconomics.com/issues/articles/2004-09_06.asp. Accessed 1 Aug 2008
Plotkin M, Eisenacher J, Brunh H et al (2004) 123I-IMT SPECT and 1H MR-spectroscopy at 3.0 T in the differential diagnosis of recurrent or residual gliomas: a comparative study. Neurooncol 70:49–58
Kimura T, Sako K, Tanaka K, Gotoh T, Tanaka T (2001) In vivo single-voxel proton MR spectroscopy in brain lesions with ring-like enhancement. NMR in Biomed 14:339–349
Traber F, Block W, Flacke S et al (2002) 1H MR spectroscopy of brain tumors in the course of radiation therapy: use of fast spectroscopic imaging and single-voxel spectroscopy for diagnosing recurrence. Rofo 174:33–42
Rabinov JD, Lee PL, Barker FG, Louis DN, Harsh GR et al (2002) In vivo 3-T MR spectroscopy in the distinction of recurrent glioma versus radiation effects: initial experience. Radiology 225:871–879
Rock JP, Scarpace L, Hearshen D, Gutierrez J et al (2004) Associations among magnetic resonance spectroscopy, apparent diffusion coefficients, and image guided histopathology with special attention to radiation necrosis. Neurosurgery 54:1111–1119
Acknowledgment
Supported by Slovak Scientific Grant Agency VEGA (No. 1/3430/06)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pružincová, Ľ., Šteňo, J., Srbecký, M. et al. MR imaging of late radiation therapy- and chemotherapy-induced injury: a pictorial essay. Eur Radiol 19, 2716–2727 (2009). https://doi.org/10.1007/s00330-009-1449-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-009-1449-8