Abstract
Objective
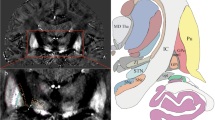
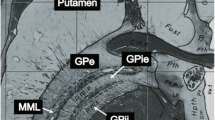
Reliable identification of the subthalamic nucleus (STN) and globus pallidus interna (GPi) is critical for deep brain stimulation (DBS) of these structures. The purpose of this study was to compare the visibility of the STN and GPi with various MRI techniques and to assess the suitability of each technique for direct stereotactic targeting.
Methods
MR images were acquired from nine volunteers with T2- and proton density-weighted (PD-W) fast spin echo, susceptibility-weighted imaging (SWI), phase-sensitive inversion recovery and quantitative T1, T2 and T2* mapping sequences. Contrast-to-noise ratios (CNR) for the STN and GPi were calculated for all sequences. Targeting errors on SWI were evaluated on magnetic susceptibility maps. The sequences demonstrating the best conspicuity of DBS target structures (SWI and T2*) were then applied to ten patients with movement disorders, and the CNRs for these techniques were assessed.
Results
SWI offers the highest CNR for the STN, but standard PD-W images provide the best CNR for the pallidum. Susceptibility maps indicated that the GPi margins may be shifted slightly on SWI, although no shifts were seen for the STN.
Conclusion
SWI may improve the visibility of the STN on pre-operative MRI, potentially improving the accuracy of direct stereotactic targeting.





Similar content being viewed by others
Abbreviations
- CNR:
-
contrast-to-noise ratio(s)
- DBS:
-
deep brain stimulation
- DESPOT1 :
-
driven equilibrium single-pulse observation of T1
- FSE:
-
fast spin echo
- GPi:
-
globus pallidus interna
- GRE:
-
gradient echo
- IR:
-
inversion recovery
- IR-FSPGR:
-
inversion recovery prepared fast spoiled gradient echo volume
- PSIR:
-
phase sensitive inversion recovery
- PD:
-
Parkinson’s disease
- PD-W:
-
proton density-weighted
- STN:
-
subthalamic nucleus
- SWI:
-
susceptibility-weighted imaging
- TE:
-
echo time
- TI:
-
inversion time
- TR:
-
repetition time
- T2-W:
-
T2-weighted
References
Tisch S, Zrinzo L, Limousin P et al (2007) Effect of electrode contact location on clinical efficacy of pallidal deep brain stimulation in primary generalised dystonia. J Neurol Neurosurg Psychiatry 78:1314–1319
Papavassiliou E, Rau G, Heath S et al (2008) Thalamic deep brain stimulation for essential tremor: Relation of lead location to outcome (Reprinted from Neurosurgery, vol 54, pg 1120-1130, 2004). Neurosurgery 62:884–893
Cuny E, Guehl D, Burbaud P et al (2002) Lack of agreement between direct magnetic resonance imaging and statistical determination of a subthalamic target: the role of electrophysiological guidance. J Neurosurg 97:591–597
Ashkan K, Blomstedt P, Zrinzo L et al (2007) Variability of the subthalamic nucleus: The case for direct MRI guided targeting. Br J Neurosurg 21:197–200
Hirabayashi H, Tengvar M, Hariz MI (2002) Stereotactic Imaging of the pallidal target. Mov Disord 17:S130–S134
Hariz MI, Krack P, Melvill R et al (2003) A quick and universal method for stereotactic visualization of the subthalamic nucleus before and after implantation of deep brain stimulation electrodes. Stereotact Funct Neurosurg 80:96–101
Starr PA, Vitek JL, DeLong M et al (1999) Magnetic resonance imaging-based stereotactic localization of the globus pallidus and subthalamic nucleus. Neurosurgery 44:303–313
Reich CA, Hudgins PA, Sheppard SK et al (2000) A high-resolution fast spin-echo inversion-recovery sequence for preoperative localization of the internal globus pallidus. Am J Neuroradiol 21:928–931
Ishimori T, Nakano S, Mori Y et al (2007) Preoperative identification of subthalamic nucleus for deep brain stimulation using three-dimensional phase sensitive inversion recovery technique. Magn Reson Med Sci 6:225–229
Pinsker MO, Volkmann J, Falk D et al (2008) Electrode implantation for deep brain stimulation in dystonia: A fast spin-echo inversion-recovery sequence technique for direct stereotactic targeting of the GPi. Zentralbl Neurochir 69:71–75
Guo T, Finnis KW, Deoni SCL et al (2006) Comparison of different targeting methods for subthalamic nucleus deep brain stimulation. Medical Image Computing and Computer-Assisted Intervention—Miccai 2006, Pt 1 4190:768-775
Elolf E, Bockermann V, Gringel T et al (2007) Improved visibility of the subthalamic nucleus on high-resolution stereotactic MR imaging by added susceptibility (T2*) contrast using multiple gradient echoes. Am J Neuroradiol 28:1093–1094
Haacke EM, Xu YB, Cheng YCN et al (2004) Susceptibility weighted imaging (SWI). Magn Reson Med 52:612–618
Vertinsky AT, Coenen VA, Lang DJ et al (2009) Localization of the subthalamic nucleus: Optimization with susceptibility-weighted phase MR imaging. Am J Neuroradiol Epub PMID: 19509077
O’Gorman RL, Wastling SJ, Lythgoe DJ et al (2009) Quantitative comparison of MRI methods for pre-surgical localisation of the subthalamic nucleus. Mov Disord 24(suppl 1):S204
O’Gorman RL, Footman M, Lythgoe DJ et al (2009) Quantitative comparison of MRI methods for pre-surgical localisation of the globus pallidus. Mov Disord 24(suppl 1):S205
Shmueli K, van Gelderen P, Yao B et al (2009) The dependence of tissue phase contrast on orientation can be overcome by quantitative susceptibility mapping. Proceedings of the Annual Meeting of the International Society of Magnetic Resonance Medicine. Honolulu, Hawaii, p 466
Shmueli K, de Zwart JA, van Gelderen P et al (2009) Magnetic susceptibility mapping of brain tissue in vivo using MRI phase data. Magn Reson Med 62:1510–1522
Deoni SCL, Rutt BK, Peters TM (2003) Rapid combined T-1 and T-2 mapping using gradient recalled acquisition in the steady state. Magn Reson Med 49:515–526
McRobbie DW (1997) A three-dimensional volumetric test object for geometry evaluation in magnetic resonance imaging. Med Phys 24:737–742
Richter EO, Hoque T, Halliday W et al (2004) Determining the position and size of the subthalamic nucleus based on magnetic resonance imaging results in patients with advanced Parkinson disease. J Neurosurg 100:541–546
Littlechild P, Varma TRK, Eldridge PR et al (2003) Variability in position of the subthalamic nucleus targeted by magnetic resonance imaging and microelectrode recordings as compared to atlas co-ordinates. Stereotact Funct Neurosurg 80:82–87
Yao B, Li TQ, van Gelderen P et al (2009) Susceptibility contrast in high field MRI of human brain as a function of tissue iron content. Neuroimage 44:1259–1266
Rutledge JN, Hilal SK, Silver AJ et al (1987) Study of movement disorders and brain iron by MR. Am J Roentgenol 149:365–379
Hallgren B, Sourander P (1958) The Effect of age on the non-haemin iron in the human brain. J Neurochem 3:41–51
Griffiths PD, Dobson BR, Jones GR et al (1999) Iron in the basal ganglia in Parkinson’s disease—An in vitro study using extended X-ray absorption fine structure and cryo-electron microscopy. Brain 122:667–673
Riederer P, Dirr A, Goetz M et al (1992) Distribution of iron in different brain regions and subcellular compartments in Parkinson’s disease. Ann Neurol 32:S101–S104
Dormont D, Ricciardi KG, Tande D et al (2004) Is the subthalamic nucleus hypointense on T2-weighted images? A correlation study using MR imaging and stereotactic atlas data. Am J Neuroradiol 25:1516–1523
Acknowledgements
The authors would like to thank Sean Deoni for supplying the DESPOT sequence.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
O’Gorman, R.L., Shmueli, K., Ashkan, K. et al. Optimal MRI methods for direct stereotactic targeting of the subthalamic nucleus and globus pallidus. Eur Radiol 21, 130–136 (2011). https://doi.org/10.1007/s00330-010-1885-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-010-1885-5