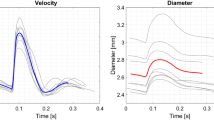
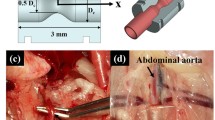
Abstract
High resolution in vivo velocity measurements within the cardiovascular system are essential for accurate calculation of vessel wall shear stress, a highly influential factor for the progression of arterial disease. Unfortunately, currently available techniques for in vivo imaging are unable to provide the temporal resolution required for velocity measurement at physiological flow rates. Advances in technology and improvements in imaging systems are allowing a relatively new technique, X-ray velocimetry, to become a viable tool for such measurements. This study investigates the haemodynamics of pulsatile blood flow in an optically opaque in vitro model at physiological flow rates using X-ray velocimetry. The in vitro model, an asymmetric stenosis, is designed as a 3:1 femoral artery with the diameter and flow rate replicating vasculature of a mouse. Velocity measurements are obtained over multiple cycles of the periodic flow at high temporal and spatial resolution (1 ms and 29 μm, respectively) allowing accurate measurement of the velocity gradients and calculation of the wall shear stress. This study clearly illustrates the capability of in vitro X-ray velocimetry, suggesting it as a possible measurement technique for future in vivo vascular wall shear stress measurement.









Similar content being viewed by others
References
Adrian, R. J. Particle-imaging techniques for experimental fluid-Mechanics. Annu. Rev. Fluid Mech. 23:261–304, 1991.
Augst, A. D., D. C. Barratt, A. D. Hughes, F. P. Glor, S. A. M. Thom and X. Y. Xu. Accuracy and reproducibility of CFD predicted wall shear stress using 3D ultrasound images. J. Biomech. Eng. Trans. ASME 125:218–222, 2003.
Baek H., M. V. Jayaraman, and G. E. Karniadakis. Wall shear stress and pressure distribution on aneurysms and infundibulae in the posterior communicating artery bifurcation. Ann. Biomed. Eng. 37(12):2469–2487, 2009.
Benveniste, H., and S. Blackband. MR microscopy and high resolution small animal MRI: applications in neuroscience research. Prog. Neurobiol. 67:393–420, 2002.
Cheng, C., D. Tempel, R. van Haperen, A. van der Bann, F. Grosveld, M. J. A. P. Daemen, R. Krams, and R. de Crom. Atherosclerotic lesion size and vulnerability are determined by patterns of fluid shear stress. Circulation 113:2744–2753, 2006.
Chytilova, E., J. Malik, Z. Kasalova, R. Dolezalova, T. Stulc, and R. Ceska. Lower wall shear rate of the common carotid artery in treated type 2 diabetes mellitus with metabolic syndrome. Physiol. Res. 58:185–191, 2009.
Davis, T. J., D. Gao, T. E. Gureyev, A. W. Stevenson, and S. W. Wilkins. Phase-contrast imaging of weakly absorbing materials using hard X-rays. Nature 373:595–598, 1995.
Dubsky, S., R. A. Jamison, S. C. Irvine, K. K. W. Siu, K. Hourigan, and A. Fouras. Computed tomographic X-ray velocimetry. Appl. Phys. Lett. 96:023702, 2010.
Dubsky, S., R. A. Jamison, S. P. A. Higgins, K. K. W. Siu, K. Hourigan, and A. Fouras. Computed tomographic X-ray velocimetry for simultaneous 3D measurement of velocity and geometry in opaque vessels. Exp. Fluids, 2010. doi:10.1007/s00348-010-1006-x
Ford, M. D., H. N. Nikolov, J. S. Milner, S. P. Lownie, E. M. Demont, W. Kalata, F. Loth, D. W. Holdsworth, and D. A. Steinman. PIV-measured versus CFD-predicted flow dynamics in anatomically realistic cerebral aneurysm models. J. Biomech. Eng. Trans. ASME 130(2):021015, 2008.
Forouhar, A. S., M. Liebling, A. Hickerson, A. Nasiraei-Moghaddam, H. Tsai, J. R. Hove, S. E. Fraser, M. E. Dickinson, and M. Gharib. The embryonic vertebrate heart tube is a dynamic suction pump. Science 312:751–753, 2006.
Fouras, A., J. Dusting, R. Lewis, and K. Hourigan. Three-dimensional synchrotron X-ray particle image velocimetry. J. Appl. Phys. 102:064916, 2007.
Fouras, A., M. J. Kitchen, S. Dubsky, R. A. Lewis, S. B. Hooper, and K. Hourigan. The past, present, and future of X-ray technology for in vivo imaging of function and form. J. Appl. Phys. 105:102009, 2009.
Fouras, A., D. Lo Jacono, and K. Hourigan. Target-free stereo PIV: a novel technique with inherent error estimation and improved accuracy. Exp. Fluids 44(2):317–329, 2008.
Fouras, A., and J. Soria. Accuracy of out-of-plane vorticity measurements derived from in-plane velocity field data. Exp. Fluids 25:409–430, 1998.
Griffith, M. D., T. Leweke, M. C. Thompson, and K. Hourigan. Pulsatile flow in stenotic geometries: flow behaviour and stability. J. Fluid Mech. 622:291–320, 2009.
Hoi, Y., S. H. Woodward, M. Kim, D. B. Taulbee, and H. Meng. Validation of CFD simulations of cerebral aneurysms with implication of geometric variations. J. Biomech. Eng. Trans. ASME 128:844–851, 2006.
Hove, J. R., R. W. Koster, A. S. Forouhar, G. Acevedo-Bolton, S. E. Fraser, and M. Gharib. Intracardiac fluid forces are an essential epigenetic factor for embryonic cardiogenesis. Nature 421:172–177, 2003.
Irvine, S. C., D. M. Paganin, S. Dubsky, R. A. Lewis, and A. Fouras. Phase retrieval for improved three-dimensional velocimetry of dynamic X-ray blood speckle. Appl. Phys. Lett. 93:153901, 2008.
Irvine, S. C., D. M. Paganin, A. Jamison, S. Dubsky, and A. Fouras. Vector tomographic X-ray phase contrast velocimetry utilizing dynamic blood speckle. Opt. Exp. 18:2368–2379, 2010.
Kim, G. B., and S. J. Lee. X-ray PIV measurements of blood flows without tracer particles. Exp. Fluids 41:195–200, 2006.
Kim, G. B, N. Y. Lim, and S. J. Lee. Hollow microcapsules for sensing micro-scale flow motion in X-ray imaging method. Microfluid. Nanofluid. 6:419–424, 2009.
Kim, M., D. B. Taulbee, M. Tremmel, and H. Meng. Comparison of two stents in modifying cerebral aneurysm hemodynamics. Ann. Biomed. Eng. 36(5):726–741, 2008.
Ku, J. P., M. T. Draney, F. R. Arko, A. Lee, F. P. Chan, N. Pelc, C. K. Zarins, and C. A. Taylor. In vivo validation of numerical prediction of blood flow in arterial bypass grafts. Ann. Biomed. Eng. 30:743–752, 2002.
Lee, J. Y., H. S. Ji, and S. J. Lee. Micro-PIV measurements of blood flow in extraembryonic blood vessels of chicken embryos. Physiol. Meas. 28:1149–1162, 2007.
Lee, S. J., S. Y. Jung, and S. Ahn. Flow tracing microparticle sensors designed for enhanced X-ray contrast. Biosens. Bioelectron. 25:1571–1578, 2010.
Lee, S. J., and G. B. Kim. X-ray particle image velocimetry for measuring quantitative flow information inside opaque objects. J. Appl. Phys. 94:3620–3623, 2003.
Lee, S. J., and G. B. Kim. Synchrotron microimaging technique for measuring the velocity fields of real blood flows. J. Appl. Phys. 97:064701, 2005
Li, M. X., J. J. Beech-Brandt, L. R. John, P. R. Hoskins, and W. J. Easso. Numerical analysis of pulsatile blood flow and vessel wall mechanics in different degrees of stenoses. J. Biomech. 40:3715–3724, 2007.
Morgan, K. S., S. C. Irvine, Y. Suzuki, K. Uesugi, A. Takeuchi, D. M. Paganin, and K. K. W. Siu. Measurement of hard X-ray coherence in the presence of a rotating random-phase-screen diffuser. Opt. Commun. 283:216–225, 2010.
Mouse Genome Sequencing Consortium. Initial sequencing and comparative analysis of the mouse genome. Nature 420:520–562, 2002.
Moyle, K. R., L. Antiga, and D. A. Steinman. Inlet conditions for image-based CFD models of the carotid bifurcation: Is it reasonable to assume fully developed flow? J. Biomech. Eng. Trans. ASME 128:371–379, 2006.
Nesbitt, W. S., E. Westein, F. J. Tovar-Lopez, E. Tolouei, A. Mitchell, J. Fu, J. Carberry, A. Fouras, and S. P. Jackson. A shear gradient-dependent platelet aggregation mechanism drives thrombus formation. Nat. Med. 15:665–673, 2009.
Poelma, C., V. Heiden, B. P. Hiereck, R. E. Poelmann, and J. Westerweel. Measurements of the wall shear stress distribution in the outflow tract of an embryonic chicken heart. J. R. Soc. Interface 7:91–103, 2010.
Poelma, C., J. M. Mari, N. Foin, M. X. Tang, R. Krams, C. G. Caro, P. D. Weinberg, and J. Westerweel. 3D Flow reconstruction using ultrasound PIV. Exp. Fluids, 2009. doi:10.1007/s00348-009-0781-8.
Poelma, C., P. Vennemann, R. Lindken, and J. Westerweel. In vivo blood flow and wall shear stress measurements in the vitelline network. Exp. Fluids 45:703–713, 2008
Reneman, R. S., T. Arts, and A. P. G. Hoeks. Wall shear stress–an important determinant of endothelial cell function and structure—in the arterial system in vivo. J. Vasc. Res. 43:251–269, 2006.
Rosencranz, R., and S. A. Bogen. Clinical laboratory measurement of serum, plasma, and blood viscosity. Pathol. Patterns Rev. 125:S78–S86, 2006.
Ross R. Cell biology of atherosclerosis. Annu. Rev. Physiol. 57:791–804, 1995.
Sheard, G. J. Flow dynamics and wall shear-stress variation in a fusiform aneurysm. J. Eng. Math. 64:379–390, 2009.
Smith, M. L., D. S. Long, E. R. Damiano, and K. Ley. Near-wall μ-PIV reveals a hydrodynamically relevant endothelial surface layer in venules in vivo. Biophys. J. 85:637–645, 2003.
Sugii, Y., S. Nishio, and K. Okamoto. In vivo PIV measurement of red blood cell velocity field in microvessels considering mesentery motion. Physiol. Meas. 23:403–416, 2002.
Texon M. Hemodynamic concept of atherosclerosis. Am. J. Cardiol. 5:291–294, 1960.
Theunissen, R., F. Scarano, and M. L. Riethmuller. Spatially adaptive PIV interrogation based on data ensemble. Exp. Fluids 48:875–887, 2010.
Thomas S. M. The current role of catheter angiography. Imaging 13:366–375, 2001.
Vennemann, P., K. T. Kiger, R. Lindken, B. C. Groenendijk, S. Stekelenburg-de Vos, T. L. ten Hagen, N. T. Ursem, R. E. Poelmann, J. Westerweel, and B. P. Hierck. In vivo micro particle image velocimetry measurements of blood-plasma in the embryonic avian heart. J. Biomech. 39:1191–1200, 2006.
Vennemann, P., R. Lindken, and J. Westerweel. In vivo whole-field blood velocity measurement techniques. Exp. Fluids 42:495–511, 2007.
Wang, Y., X. Liu, K. Im, W. Lee, J. Wang, K. Fezzaa, D. L. S. Hung, and J. R. Winkelman. Ultrafast X-ray study of dense-liquid-jet flow dynamics using structure-tracking velocimetry. Nat. Phys. 4:305–309, 2008.
Wen, C. Y., A. S. Yang, L. Y. Tseng, and J. W. Chai. Investigation of pulsatile flowfield in healthy thoracic aorta models. Ann. Biomed. Eng. 38(2):391–402, 2010.
Acknowledgments
The authors gratefully acknowledge the support of the Japan Synchrotron Radiation Research Institute (JASRI) (under Proposal No. SP2009B1910). The authors would like to thank Yoshio Suzuki, Akihisa Takeuchi and Kentaro Uesugi (SPring-8/JASRI) for their assistance with the experiments. Support from the Australian Research Council (Grant Nos. DP0877327, DP0987643) is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Konstantinos Konstantopoulos oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Jamison, R.A., Dubsky, S., Siu, K.K.W. et al. X-ray Velocimetry and Haemodynamic Forces Within a Stenosed Femoral Model at Physiological Flow Rates. Ann Biomed Eng 39, 1643–1653 (2011). https://doi.org/10.1007/s10439-011-0260-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-011-0260-2