Abstract
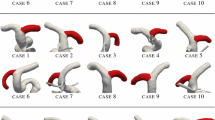
We propose a novel method to reconstruct the hypothetical geometry of the healthy vasculature prior to intracranial aneurysm (IA) formation: a Frenet frame is calculated along the skeletonization of the arterial geometry; upstream and downstream boundaries of the aneurysmal segment are expressed in terms of the local Frenet frame basis vectors; the hypothetical healthy geometry is then reconstructed by propagating a closed curve along the skeleton using the local Frenet frames so that the upstream boundary is smoothly morphed into the downstream boundary. This methodology takes into account the tortuosity of the arterial vasculature and requires minimal user subjectivity. The method is applied to 22 clinical cases depicting IAs. Computational fluid dynamic simulations of the vasculature without IA are performed and the haemodynamic stimuli in the location of IA formation are examined. We observe that locally elevated wall shear stress (WSS) and gradient oscillatory number (GON) are highly correlated (20/22 for WSS and 19/22 for GON) with regions susceptible to sidewall IA formation whilst haemodynamic indices associated with the oscillation of the WSS vectors have much lower correlations.






Similar content being viewed by others
Notes
The Circle of Willis is a circle of arteries at the base of the brain. All the principal arteries that supply blood to the cerebral hemispheres of the brain branch off from the Circle of Willis.
References
Aird, W. C. Spatial and temporal dynamics of the endothelium. J. Thrombosis Haemost. 3(7):1392–1406, 2005.
Alnaes, M. S., J. Isaksen, K. A. Mardal, B. Romner, M. K. Morgan, T. L. Ingebrigtsen. Computation of hemodynamics in the circle of willis. Stroke 38:2500–2505, 2007.
Aoki, T., and M. Nishimura. Molecular mechanism of cerebral aneurysm formation focusing on NF-κB as a key mediator of inflammation. J. Biorheol. 24(1):16–21, 2010.
Augst, A. D., B. Ariff, S. A. G. McG Thom, X. Y. Xu, and A. D. Hughes. Analysis of complex flow and the relationship between blood pressure, wall shear stress, and intima-media thickness in the human carotid artery. Am. J. Physiol. Heart Circ. Physiol. 293:H1031–H1037, 2007.
Baek, H., M. V. Jayaraman, and G. E. Karniadakis. Wall shear stress and pressure distribution on aneurysms and infundiblae in the posterior communicating artery bifurcation. Ann. Biomed. Eng. 37:2469–2487, 2009.
Brisman, J. L., J. K. Song, and D. W. Newell. Cerebral aneurysms. N. Engl. J. Med. 355:928–939, 2006.
Cebral, J. R., M. A. Castro, O. Soto, R. Lohner, and N. Alperin. Blood-flow models of the circle of willis from magnetic resonance data. J. Eng. Math. 47:369–386, 2003.
Cebral, J. R., and H. Meng. Counterpoint: realizing the clinical utility of computational fluid dynamics–closing the gap. AJNR—Am. J. Neuroradiol. 33(3):396–398, 2012.
Cebral, J. R., M. C. Putman, M. T. Alley, T. Hope, R. Bammer, and F. Calamante. Hemodynamics in normal cerebral arteries: qualitative comparison of 4d phase-contrast magnetic resonance and image-based computational fluid dynamics. J. Eng. Math. 64:367–378, 2009.
Cheng, C. P., D. Parker, and C. A. Taylor. Quantification of wall shear stress in large blood vessels using Lagrangian interpolation functions with cine phase-contrast magnetic resonance imaging. Ann. Biomed. Eng. 30(8):1020–1032, 2002.
Doenitz, C., K. M. Schebesch, R. Zoephel, and A. Brawanski. A mechanism for rapid development of intracranial aneurysms: a case study. Neurosurgery 67:1213–1221, 2010.
Dolan, J. M., H. Meng, S. Singh, R. Paluch, and J. Kolega. High fluid shear stress and spatial shear stress gradients affect endothelial proliferation, survival, and alignment. Ann. Biomed. Eng. 39:1620–1631, 2011.
Ford, M. D., Y. Hoi, M. Piccinelli, L. Antiga, and D. A. Steinman. An objective approach to digital removal of saccular aneurysms: technique and applications. Br. J. Radiol. 82:55–61, 2009.
Glor, F. P., B. Ariff, A. D. Hughes, L. A. Crowe, P. R. Verdonck, D. C. Barratt, S. A. McG Thom, D. N. Firmin, and X. Y. Xu. Image-based carotid flow reconstruction: a comparison between MRI and ultrasound. Physiol. Meas. 25:1495–1509, 2004.
Glor, F. P., Q. Long, A. D. Hughes, A. D. Augst, B. Ariff, S. A. Thom, P. R. Verdonck, and X. Y. Xu. Reproducibility study of magnetic resonance image-based computational fluid dynamics prediction of carotid bifurcation flow. Ann. Biomed. Eng. 31:142–151, 2002.
He, X., and D. N. Ku. Pulsatile flow in the human left coronary artery bifurcation: average conditions. J. Biomech. Eng. 118(1):74–82, 1996.
Hoi, Y., H. Meng, S. H. Woodward, B. R. Bendok, R. A. Hanel, L. R. Guterman, and L. N. Hopkins. Effects of arterial geometry on aneurysm growth: three-dimensional computational fluid dynamics study. J. Neurosurg. 101(4):676–81, 2004.
Humphrey, J. D., and C. A. Taylor. Intracranial and abdominal aortic aneurysms: similarities, differences, and need for a new class of computational models. Annu. Rev. Biomed. Eng. 10:221–246, 2008.
Jou, L. D., and M. E. Mawad. Hemodynamic effect of neuroform stent on intimal hyperplasia and thrombus formation in a carotid aneurysm. Med. Biol. Eng. Comput. 33:573–580, 2011.
Jou, L. D., and M. E. Mawad. Timing and size of flow impingement in a giant intracranial aneurysm at the internal carotid artery. Med. Biol. Eng. Comput. 49:891–899, 2011.
Juvela, S. Treatment options of unruptured intracranial aneurysms. Stroke 35:372–374, 2004.
Kanematsu, Y., M. Kanematsu, C. Kurihara, Y. Tada, T.-L. Tsou, N. van Rooijen, M. T. Lawton, W. L. Young, E. I. Liang, Y. Nuki, and T. Hashimoto. Critical roles of macrophages in the formation of intracranial aneurysm. Stroke 42(1):173–178, 2011.
Karmonik, C., A. Arat, G. Benndorf, S. Akpek, R. Klucznik, M. E. Mawad, and C. M. Strother. A technique for improved quantitative characterization of intracranial aneurysms. Am. J. Neuroradiol. 25:1158–1161, 2004.
Ku, D. N., D. P. Giddens, C. K. Zarins, and S. Glagov. Pulsatile flow and atherosclerosis in the human carotid bifurcation. positive correlation between plaque location and low oscillating shear stress. Arterioscler. Thrombosis Vasc. Biol. 5(3):293–302, 1985.
Kulcsár, Z., A. Ugron, M. Marosfoi, Z. Berentei, G. Paál, and I. Szikora. Hemodynamics of cerebral aneurysm initiation: the role of wall shear stress and spatial wall shear stress gradient. Am. J. Neuroradiol. 32:587–594, 2011.
Mantha, A., C. Karmonik, G. Benndorf, C. Strother, and R. Metcalfe. Hemodynamics in a cerebral artery before and after the formation of an aneurysm. Am. J. Neuroradiol. 27:1113–1118, 2006.
Meng, H., E. Metaxa, L. Gao, N. Liaw, S. K. Natarajan, D. D. Swartz, A. H. Siddiqui, J. Kolega, and J. Mocco. Progressive aneurysm development following hemodynamic insult. J. Neurosurg. 114(4):1095–1103, 2011.
Meng, H., D. D. Swart, Z. Wang, Y. Hoi, J. Kolega, E. Metaxa, and M. P. Szymanski. A model system for mapping vascular responses to complex hemodynamics at arterial bifurcations in vivo. Neurosurgery 59(5):1094–1100, 2006.
Meng, H., Z. Wang, Y. Hoi, L. Gao, E. Metaxa, D. D. Swartz, J. Kolega, and D. D. Swart. Complex hemodynamics at the apex of an arterial bifurcation induces vascular remodelling resembling cerebral aneurysm initiation. Stroke 38(6):1924–1931, 2007.
Metaxa, E., M. Tremmel, S. K. Natarajan, J. Xiang, R. A. Paluch, M. Mandelbaum, and A. H. Siddiqui. Characterization of critical hemodynamics contributing to aneurysmal remodeling at the basilar terminus in a rabbit model. Stroke 41(8):1774–1782, 2010.
Patankar, S. V. Numerical Heat Transfer and Fluid Flow. Hemisphere Series on Computational Methods in Mechanics and Thermal Science. Washington/New York: Hemisphere Pub. Corp./McGraw-Hill, 1980.
Regan, E. R., and W. C. Aird. Dynamical systems approach to endothelial heterogeneity. Circ. Res. 111(1):110–130, 2012.
Reymond, P., Y. Bohraus, F. Perren, F. Lazeyras, and N. Stergiopulos. Validation of a patient-specific one-dimensional model of the systemic arterial tree. AJP 301:1173–1182, 2011.
Reymond, P., F. Merenda, F. Perren, D. Rufenacht, and N. Stergiopulos. Validation of a one-dimensional model of the systemic arterial tree. AJP 297:208–222, 2009.
Rinkel, G. J., M. Djibuti, A. Algra, and J. V. Gijn. Prevalence and risk of rupture of intracranial aneurysms: a systematic review. Stroke 29:251–256, 1998.
Robertson, A. M., and P. N. Watton. Computational fluid dynamics in aneurysm research: critical reflections, future directions. Am. J. Neuroradiol. 33(6):992–995, 2012.
Shimogonya, Y., T. Ishikawa, Y. Imai, N. Matsuki, and T. Yamaguchi. A realistic simulation of saccular cerebral aneurysm formation: focussing on a novel haemodynamic index, the gradient oscillatory number. Int. J. Comput. Fluid Dyn. 23:583–593, 2009.
Shimogonya, Y., T. Ishikawa, Y. Imai, N. Matsuki, and T. Yamaguchi. Can temporal fluctuation in spatial wall shear stress gradient initiate a cerebral aneurysm? A proposed novel hemodynamic index, the gradient oscillatory number (GON). J. Biomech. 42:550–554, 2009.
Shimogonya, Y., H. Kumamaru, and K. Itoh. Sensitivity of the gradient oscillatory number to flow input waveform shapes. J. Biomech. 45:985–989, 2012.
Singh, P. K., A. Marzo, B. Howard, D. A. Rufenacht, P. Bijlenga, A. F. Frangi, P. V. Lawford, S. C. Coley, D. R. Hose, and U. J. Patel. Effects of smoking and hypertension on wall shear stress and oscillatory shear index at the site of intracranial aneurysm formation. Clin. Neurol. Neurosurg. 112(4):306–313, 2010.
Tremmel, M., J. Xiang, Y. Hoi, J. Kolega, A. H. Siddiqui, J. Mocco, and H. Meng. Mapping vascular response to in vivo hemodynamics: application to increased flow at the basilar terminus. Biomech. Model. Mechanobiol. 9:421–434, 2010.
Villa-Uriol, M. C., G. Berti, D. R. Hose, A. Marzo, A. Chiarini, J. Penrose, J. Pozo, J. G. Schmidt, P. Singh, R. Lycett, I. Larrabide, and A. F. Frangi. @neurist complex information processing toolchain for the integrated management of cerebral aneurysms. Interface Focus 1:308–319, 2011.
Vlak, M. H., A. Algra, R. Brandenburg, and G. J. Rinkel. Prevalence of unruptured intracranial aneurysms, with emphasis on sex, age, comorbidity, country, and time period: a systematic review and meta-analysis. Lancet Neurol. 10(7):626–636, 2011.
Watton, P. N., H. Huang, and Y. Ventikos. Multi-scale modelling of vascular disease: abdominal aortic aneurysm evolution. In: Computational Modeling in Tissue Engineering, volume 10 of Studies in Mechanobiology, Tissue Engineering and Biomaterials, edited by L. Geris. Springer, Berlin, 2013, pp. 309–339.
Watton, P. N., Y. Ventikos, and G. A. Holzapfel. Modelling cerebral aneurysm evolution. In: Biomechanics and Mechanobiology of Aneurysms, Vol. 7 of Studies in Mechanobiology, Tissue Engineering and Biomaterials, chapter 12, edited by T. McGloughlin. Heidelberg: Springer, pp. 307-322, 2011.
Wolfe, S. Q., M. K. Bakaya, R. C. Heros, R. P. Tummala. Cerebral aneurysms: learning fom the past and looking toward the future. Clin. Neurosurg. 53:157–178, 2006.
Zeng, Z., D. F. Kallmes, M. J. Durka, Y. Ding, D. A. Lewis, R. Kadirvel, and A. M. Robertson. Hemodynamics and anatomy of elastase-induced rabbit aneurysm modelssimilarity to human cerebral aneurysms? Am. J. Neuroradiol. 32:595–601, 2011.
Acknowledgments
Haoyu Chen is funded by the Qualcomm scholarship provided by Qualcomm Inc. (Qualcomm Inc., San Diego, CA). Alisa Selimovic is funded by the Robert Menzies Memorial Scholarship in Engineering. Paul N. Watton holds a University Research Lectureship funded by the Centre of Excellence in Personalized Healthcare (funded by the Wellcome Trust and EPSRC, grant number WT 088877/Z/09/Z). This support is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Ender A. Finol oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Chen, H., Selimovic, A., Thompson, H. et al. Investigating the Influence of Haemodynamic Stimuli on Intracranial Aneurysm Inception. Ann Biomed Eng 41, 1492–1504 (2013). https://doi.org/10.1007/s10439-013-0794-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-013-0794-6