Abstract
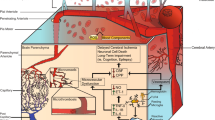
Intracerebral hemorrhagic transformation is a multifactorial phenomenon in which ischemic brain tissue converts into a hemorrhagic lesion with blood-vessel leakage, extravasation, and further brain injury. It has been estimated that up to 30–40% of all ischemic strokes undergo spontaneous hemorrhagic transformation, and this phenomenon may become even more prevalent with the increasing use of thrombolytic stroke therapy. An emerging conceptual model suggests that the loss of microvascular integrity and disruption of neurovascular homeostasis connects the experimental findings of blood-cell extravasation to brain injury after hemorrhage. In this short article, we examine mechanisms related to reperfusion injury and oxidative stress, leukocyte infiltration, vascular activation, and dysregulated extracellular proteolysis as potential triggers of hemorrhagic transformation. Perturbations in cell-cell and cell-matrix signaling within the hypothesized neurovascular unit may ultimately lead to neuroinflammation and apoptotic-like cell death in the parenchyma. Further investigations into the molecular mediators of hemorrhagic transformation may reveal new therapeutic targets for this clinically complex problem.
Similar content being viewed by others
References
Lyden P.D. and Zivin J.A. (1993) Hemorrhagic transformation after cerebral ischemia: mechanisms and incidence. Cerebrovasc. Brain Metab. Rev. 5, 1–16.
Lapchak P.A. (2002) Hemorrhagic transfromation following ischemic stroke: significance, causes, and relationship to therapy and treatment. Curr. Neurol. Neurosci. Rep. 2, 38–43.
Passero S., Ciacci G., and Reale F. (2001) Potential triggering factors of intracerebral hemorrhage. Cerebrovasc. Dis. 12, 220–227.
Gebel J.M. and Broderick J.P. (2000) Intracerebral hemorrhage. Neurol. Clin. 18, 419–438.
Andaluz N., Zuccarello M., and Wagner K.R. (2002) Experimental animal models of intracerebral hemorrhage. Neurosurg. Clin. N. Am. 3, 385–393.
Vahedi K. and Bousser M.G. (2002) Thrombolysis in stroke. Curr. Opin. Hematol. 9, 443–447.
Report of the Stroke Progress Review Group, NINDS, http://www.ninds.nih.gov/funding/neural_environment/stroke_prg
Lo E.H., Dalkara T., and Moskowitz M.A. (2003) Mechanisms, challenges and opportunities in stroke. Nat. Rev. Neurosci. 4, 399–415.
del Zoppo G.J. and Hallenbeck J.M. (2000) Advances in the vascular pathophysiology of ischemic stroke. Thromb. Res. 98, 73–81.
Petty M.A. and Wettstein J.G. (2001) Elements of cerebral microvascular ischemia. Brain Res. 36, 23–34.
Petty M.A. and Lo E.H. (2002) Junctional complexes of the blood-brain barrier:permeability changes in neuroinflammation. Prog. Neurobiol. 68, 311–323.
Wolburg H. and Lippoldt A. (2002) Tight junctions of the blood-brain barrier:development, composition and regulation. Vascul. Pharmacol. 38, 323–337.
Huber J.D., Egleton R.D., and Davis T.P. (2001) Molecular physiology and pathophysiology of tight junctions in the blood-brain barrier. Trends Neurosci. 24, 719–725.
Mark K.S. and Davis T.P. (2002) Cerebral microvascular changes in permeability and tight junction induced by hypoxia-reoxygenation. Am. J. Physiol. Heart. Circ. Physiol. 282, H1485-H1494.
Yurchenco P.D. and Schittny J.C. (1990) Molecular architecture of basement membranes. FASEB J. 4, 1577–1590.
Abrahamason D.R. (1986) Recent studies on the structure and pathology of basement membranes. J. Pathol. 149, 247–278.
Mohan P.S. and Spiro R.G. (1986) Macromolecular organization of basement membranes. J. Biol. Chem. 261, 4328–4336.
Siflinger-Birnboim A., Delvecchio P.J., Cooper J.A., Blumenstock F.A., Shepard J.M., and Malik A.B. (1987) Molecular sieving characteristics of cultured endothelial monolayer. J. Cell. Physiol. 132, 111–117.
Cserr F.H. and Bungaard M. (1986) The neuronal microenvironment: a comparative view. Ann. NY. Acad. Sci. 481, 1–6.
Fishman R.A. (1975) Brain edema. N. Engl. J. Med. 293, 706–711.
Carr F.J., McBride M.W., Carswell H.V.O., Graham D., Strahorn P., Clark J.S., et al. (2002) Genetic aspects of stroke: human and experimental studies. J. Cereb. Blood Flow Metab. 22, 767–773.
Wagner K.R. and Broderick J.P. (2001) Hemorrhagic stroke: pathophysiological mechanisms and neuroprotective treatments, Lo E.H. and Marwah J. Neuroprotection. Prominent Press, Scottsdale A2, pp. 471–508.
Mayer S.A. (2002) Intracerebral hemorrhage:natural history and rationale of ultra-early hemostatic therapy. Intensive Care Med. 28, S235-S240.
Castillo J., Davalos A., Alvarez-Sabin J., Pumar J.M., Leira R., Silva Y., et al. (2002) Molecular signatures of brain injury after intracerebral hemorrhage. Neurology 58, 624–629.
Golding E.M. (2002) Sequelae following traumatic brain injury. The cerebrovascular perspective. Brain Res. Brain Res. Rev. 38, 377–388.
Qiu J., Whalen M.J., Lowenstein P., Fiskum G., Fahy B., Darwish R., et al. (2002) Upregulation of the Fas receptor death-inducing signaling complex after traumatic brain injury in mice and humans. J. Neurosci. 22, 3504–3511.
Zhang X., Chen J., Graham S.H., Du L., Kochanek P.M., Draviam R., et al. (2002) Intranuclear localization of apoptosis-inducing factor (AIF) and large scale DNA fragmentation after traumatic brain injury in rats and in neuronal cultures exposed to peroxynitrite. J. Neurochem. 82, 181–191.
Yang G.Y., Betz A.L., Chenevert T.L., Brunberg J.A., and Hoff J.T. (1994) Experimental intracerebral hemorrhage:relationship between brain edema, blood flow, and blood-brain barrier permeability in rats. J. Neurosurg. 81, 93–102.
Rosand J., Eskey C., Chang Y., Gonzalez R.G., Greenberg S.M., and Koroshetz W.J. (2002) Dynamic single-section CT demonstrates reduced cerebral blood flow in acute intracerebral hemorrhage. Cerebrovasc. Dis. 14, 214–220.
Zazulia A.R., Diringer M.N., Videen T.O., Adams R.E., Yundt K., Aiyagari V., et al. (2001) Hypoperfusion without ischemia surrounding acute intracerebral hemorrhage. J. Cereb. Blood Flow Metab. 21, 804–810.
Zhang J., Sima B., Johns L.M., and Macdonald R.L. (1996) Time course of changes in arterial relaxation following subarachnoid hemorrhage in dogs. Neurol. Res. 18, 227–232.
Matsumoto K., Lo E.H., Pierce A.R., Halpern E.F., and Newcomb R. (1996) Secondary elevations in extracellular neurotransmitter amino acids in reperfusion phase following focal cerebral ischemia. J. Cereb. Blood Flow Metab. 16, 114–124.
Chen Y. and Swanson R.A. (2003) Astrocytes and brain injury. J. Cereb. Blood Flow Metab. 23, 137–149.
Striggow F., Riek M., Breder J., Henrich-Noack P., Reymann K.G., and Reiser G. (2000) The protease thrombin is an endogenous mediator of hippocampal neuroprotection against ischemia at low concentrations but causes degeneration at high concentrations. Proc. Natl. Acad. Sci. USA 97, 2264–2269.
Xi G., Reiser G., and Keep R.F. (2003) The role of thrombin and thrombin receptors in ischemic, hemorrhagic and traumatic brain injury: deleterious or protective? J. Neurochem. 84, 3–9.
Mayne M., Ni W., Yan H.J., Xue M., Johnston J.B., Del Bigio M.R., et al. (2001) Antisense oligodeoxynucleotide inhibition of tumor necrosis factor-alpha expression is neuroprotective after intracerebral hemorrhage. Stroke 32, 240–248.
Lenzlinger P.M., Marx A., Trentz O., Kossmann T., and Morganti-Kossmann M.C. (2002) Prolonged intrathecal release of soluble Fas following severe traumatic brain injury in humans. J. Neuroimmunol. 122, 167–174.
Regan R.F. and Panter S.S. (1993) Neurotoxicity of hemoglobin in cortical cell culture. Neurosci. Lett. 153, 219–222.
Meguro T., Chen B., Lancon J., and Zhang J.H. (2001) Oxyhemoglobin induces caspase-mediated cell death in cerebral endothelial cells. J. Neurochem. 77, 1128–1135.
Glazner G.W., Boland A., Dresse A.E., Brenneman D.E., Gozes I., and Mattson M.P. (1999) Activity-dependent neurotrophic factor peptide (ADNF9) protects neurons against oxidative stress-induced death. J. Neurochem. 73, 2341–2347.
Willmore L.J., and Rubin J.J. (1984) Effects of antiperoxidants on FeCl2-induced lipid peroxidation and focal edema in rat brain. Exp. Neurol. 83, 62–70.
Chan P.H. (1994) Oxygen radicals in focal cerebral ischemia. Brain Pathol. 4, 59–65.
Abe K., Yoshidomi M., and Kogure K. (1989) Arachidonic acid metabolism in ischemic neuronal damage. Ann. NY Acad. Sci. 559, 259–268.
Glaser K.B., Mobillo D., Channg J.Y., and Senko N. (1993) Phospholipase A2 enzymes: regulation and inhibition. Trends Pharmacol. Sci. 14, 92–98.
Asahi M., Ramahonan R., Sumii T., Wang X., Pauw R.J., Weissig V., et al. (2003) Targeted immunoliposomes reseal vascular damage and reduce hemorrhage after thrombolysis in embolic cerebral ischemia. J. Cereb. Blood Flow Metab. (in press).
Bolton S.J., Anthony D.C., and Perry V.H. (1998) Loss of the tight junction proteins occludin and zonula occludens-1 from cerebral vascular endothelium during neutrophil induced blood-brain barrier breakdown in vivo. Neuroscience. 86, 1245–1257.
Kondo T., Kinouchi H., Kawase M., and Yoshimoto T. (1996) Astroglial cells inhibit the increasing permeability of brain endothelial cell monolayer following hypoxia/reoxygenation. Neurosci. Lett. 19, 101–104.
Fischer S., Wobben M., Kleinstuck J., Renz D., and Schaper W. (2000) Effect of astroglial cells on hypoxia-induced permeability in PBMEC cells. Am. J. Physiol. Cell Physiol. 279, C935-C944.
Wei E.P., Christman C.W., Kontos H.A., and Povlishock J.T. (1985) effects of oxygen radicals on cerebral arterioles. Am. J. Physiol. 248, H157-H162.
Zweier J.L., Kuppusamy P., and Lutty G.A. (1988) Measurement of endothelial cell free radical generation: evidence for a central mechanism of free radical injury in postischemic tissue. Proc. Natl. Acad. Sci. USA 85, 4046–4050.
Franko J., Pomfy M., Novakova B., and Bens L. (1999) Stobadine protects against ischemia-reperfusion induced morphological alterations of cerebral microcirculation in dogs. Life Sci. 65, 1963–1967.
Kim G.W., Lewen A., Copin J., Watson B.D., and Chan P.H. (2001) The cytosolic antioxidant, copper/zinc superoxide dismutase, attenuates blood-brain barrier disruption and oxidative cellular injury after photothrombotic cortical ischemia in mice. Neuroscience 105, 1007–1018.
Schmid-Elsaesser R., Zausinger S., Hungerhuber E., Plesnila N., Baethmann A., Reulen H.J., et al. (1997) Superior neuroprotective efficacy of a novel antioxidant (U-101033E) with improved blood-brain barrier permeability in focal cerebral ischemia. Stroke 28, 2018–2024.
Kondo T, Reaume A.G., Huang T-T., Carlson E., Murakami K., Chen S.F., et al. (1997) Reduction of CuZn-superoxide dismutase activity exacerbates neuronal cell injury and edema formation after transient focal cerebral ischemia. J. Neurosci. 17, 4180–4189.
Lapchak P.A., Chapman D.F., and Zivin J.A. (2001) Pharmacological effects of the spin trap agents N-t-butyl-phenylnitrone (PBN) and 2,2,6,6-tetramethylpiperidine-N-oxyl (TEMPO) in a rabbit thromboembolic stroke model: combination studies with the thrombolytic tissue plasminogen activator. Stroke 32, 147–153.
Asahi M., Asahi K., Wang X., and Lo E.H. (2000) Reduction of tissue plasminogen activator-induced hemorrhage and brain injury by free radical spin trapping after embolic focal cerebral ischemia in rats. J. Cereb. Blood Flow Metab. 20, 452–457.
Haddad J.J. (2002) Redox regulation of pro-inflammatory cytokines and IkappaB-alpha/NF kappaB nuclear translocation and activation. Biochem. Biophys. Res. Commun. 296, 847–856.
Inoue N., Takeshita S., Gao D., Ishida T., Kawashima S., Akita H., et al. (2001) Lysophosphatidylcholine increases the secretion of matrix metalloproteinase 2 through the activation of NADH/NADPH oxidase in cultured aortic endothelial cells. Atherosclerosis 155, 45–52.
Giulian D., Woodward J., Young D.G., Krebs J.F., and Lachman L.B. (1988) Interleukin-1 injected into mammalian brain stimulates astrogliosis and neovascularization. J. Neurosci. 8, 2485–2490.
Holmin S. and Matheisen T. (2000) Intracerebral adminstration of interleukin-1 and induction of inflammation, apoptosis and edema. J. Neurosurg. 92, 108–120.
Shafer R.A. and Murphy S. (1997) Activated astrocytes induce nitric oxide synthase-2 in cerebral endothelium via tumor necrosis factor alpha. Glia 370–379.
Fischer S., Clauss M., Wiesnet M., Renz D., Schaper W., and Karliczek G.F. (1999) Hypoxia induces permeability in brain microvessel endothelial cells via VEGF and NO. Am. J. Physiol. 276, C812-C820.
Barone F.C. and Feuerstein G.Z. (1999) Inflammatory mediators and stroke: new opportunities for novel therapeutics. J. Cereb. Blood Flow Metab. 19, 819–834.
Stanimirovic D. and Satoh K. (2000) Inflammatory mediators of cerebral endothelium: a role in ischemic brain inflammation. Brain Pathol. 10, 113–126.
Zhang W. and Stanimirovic D. (2002) Current and future therapeutic strategies to target inflammation in stroke. Current Drug Target—Inflammation & Allergy 1, 151–166.
Janoff A. and Zeligs J.D. (1968) Vascular injury and lysis of basement membrane in vitro by neutral protease of human leukocyte. Science 161, 702–704.
Weiss S.J., Peppin G., Ortiz X., Ragsdale C., and Test S.T. (1985) Oxidative autoactivation of latent collagenase by human neutrophils. Science 227, 747–749.
Zhang R., Chopp M., Zhang Z., Jiang N., and Power C. (1998) The expression of P-and E-selectins in three models of middle cerebral artery occlusion. Brain Res. 785, 207–214.
Haraldsen G., Kvale D., Lien B., Farstad I.N., and Brandtzaeg P. Cytokine-regulated expression of E-selectin, intercellular adhesion molecule-1 (ICAM-1), and vascular cell adhesion molecule-1 (VCAM-1) in human microvascular endothelial cells. J. Immunol. 156, 2558–2565.
Gen J.G., Bevilacque M.P., Moore K.L., McIntyre T.M., and Prestcott S.M. (1990) Rapid neurophil adhesion to activated endothelium mediated by GMP-140. Nature 343, 757–760.
Wagner D.D. (1993) Weibel-Palade body: The storage granule for von Willebrand factor and P-selectin. Thromb. Haemostasis 70, 105–110.
Baggiolini M (1998) Chemokines and leukocyte traffic. Nature 392, 565–568.
Che X., Ye W., Panga L., Wu D.C., Yang G.Y. (2001) Monocyte chemoattractant protein-1 expressed in neurons and astrocytes during focal ischemia in mice. Brain Res. 902, 171–177.
Wang X., Li X., Yaish-Ohad S., Sarau H.M., Barone F.C., and Feuerstein G.Z. (1999) Molecular cloning and expression of the rat monocyte chemotactic protein-3 gene: a possible role in stroke. Brain Res. Mol. Brain Res. 71, 304–312.
Bajetto A., Bonavia R., Barbero S., and Schettini G. (2002) Characterization of chemokines and their receptors in the central nervous system: physiopathological implications. J. Neurochem. 82, 1311–1329.
Couraud P.O. (1998) Infiltration of inflammatory cells through brain endothelium. Pathol. Biol. 46, 176–180.
Rosenberg G.A. (2002) Matrix metalloproteinases in neuroinflammation. Glia 39, 279–291.
Wong D. and Dorovini-Zis K. (1992) Upregulation of intercellular adhesion molecule-1 (ICAM-1) expression in primary cultures of human brain microvessel endothelial cells by cytokines and lipopolysaccharide. J. Neuroimmunol. 39, 11–21.
Adamson P., Etienne S., Couraud P.O., Calder V., and Greenwood J. (1999) Lymphocyte migration through brain endothelial cell monolayers involves signaling through endothelial ICAM-1 via a rho-dependent pathway. J. Immunol. 162, 2964–2973.
Etienne-Manneville S., Manneville J.B., Adamson P., Wilbourn B., Greenwood J., and Couraud P. O. (2000) ICAM-I-coupled cytoskeletal rearrangements and transendothelial lymphocyte migration involve intracellular calcium signaling in brain endothelial cell lines. J. Immunol. 165, 3375–3383.
Brown R.C. and Davis T.P. (2002) Calcium modulation of adherens and tight junction function: a potential mechanism for BBB disruption after stroke. Stroke 33, 1706–1711.
Allport J.R., Ding H., Collins T., Gerritsen M.E., and Luscinkas F.W. (1997) Endothelial-dependent mechanisms regulate leukocyte transmigrationL a process involving the proteosome and disruption of vascular-endothelial -cadherin complex at endothelial cell-cell junctions. J. Exp. Med. 186, 517–527.
Zhang Z.G., Zhang L., Jiang Q., Zhang R., Davies K., Powers C., et al. (2000) VEGF enhances angiogenesis and promotes blood-brain barrier leakage in the ischemic brain. J. Clin. Investig 106, 829–838.
Zhang Z.G., Zhang L., Tsang W., Soltanian-Zadeh H., Morris D., Zhang R., et al. (2002) Correlation of VEGF and angiopoietin expression with disruption of blood-brain barrier and angiogenesis after focal cerebral ischemia. J. Cereb. Blood Flow Metab. 22, 379–392.
Marti H.J., Bernaudin M., Bellail A., Schoch H., Euler M., Petit E., et al. (2000) Hypoxia-induced vascular endothelial growth factor expression precedes neovascularization after cerebral ischemia. Am. J. Pathol. 156, 965–976.
Croll S.D. and Wiegand S.J. (2001) Vascular growth factors in cerebral ischemia. Mol. Neurobiol. 23, 121–135.
van Bruggen N., Thibodeaux H., Palmer J.T., Lee W.P., Fu L., Cairns B., et al. (1999) VEGF antagonism reduces edema formation and tissue damage after ischemia/reperfusion injury in the mouse brain. J. Clin. Investig 104, 1613–1620.
Wagner S., Tagaya M., Koziol J.A., Quaranta V., and del Zoppo G.J. (1997) Rapid disruption of an astrocyte interaction with the extracellular matrix mediated by integrin alpha 6 beta 4 during focal cerebral ischemia/reperfusion. Stroke 28, 858–865.
Tagaya M., Haring H.P., Stuiver I., Wagner S., Abumiya T., Lucero J., et al. (2001) Rapid loss of microvascular integrin expression during focal brain ischemia reflects neuron injury. J. Cereb. Blood Flow Metab. 21, 835–846.
Zhang Z.G., Bower L., Zhang R.L., Chen S., Windham J.P., and Chopp M. (1999) Three-dimensional measurement of cerebral microvascular plasma perfusion, glial fibrillary acidic protein and microtubule associated protein-2 immunoreactivity after embolic stroke in rats: a double fluorescent labeled laser-scanning confocal microscopic study. Brain Res. 844, 55–66.
Hamann G.F., Okada Y., Fitridge R., and del Zoppo G.J. (1995) Microvascular basal lamina antigens disappear during cerebral ischemia and reperfusion. Stroke 26, 2120–2126.
Hamann G.F., Liebetrau M., Martens H., Burggraf D., Kloss C.U., Bultemeier G., et al. (2002) Microvascular basal lamina injury after experimental focal cerebral ischemia and reperfusion in the rat. J. Cereb. Blood Flow Metab. 22, 526–533.
Hamann G.F., Okada Y., and del Zoppo G.J. (1996) Hemorrhagic transformation and microvascular integrity during focal cerebral ischemia/reperfusion. J. Cereb. Blood Flow Metab. 16, 1373–1378.
del Zoppo G.J., von Kummer R., and Hamann G.F. (1998) Ischaemic damage of brain microvessels: inherent risks for thrombolytic treatment in stroke. J. Neurol. Neurosurg. Psychiatry 65, 1–9.
Gasche Y., Copin J-C., Sugawara T., Fujimura K, and Chan P.H. (2001) Matrix metalloproteinase inhibition prevents oxidative stress-associated blood-brain barrier disruption after transient focal cerebral ischemia. J. Cereb. Blood Flow Metab. 21, 1393–1400.
Lo E.H., Wang X., and Cuzner M.L. (2002) Extracellular proteolysis in brain injury and inflammation:role for plasminogen activators and matrix metalloproteinases. J. Neurosci. Res. 69, 1–9.
Armao D., Kornfeld M., Estrada E.Y., Grossetete M., and Rosenberg G.A. (1997) Neutral proteases and disruption of the blood-brain barrier in rat. Brain Res. 767, 259–264.
Asahi M., Wang X., Mori T., Sumii T., Jung J-C., Moskowitz M.A., et al. (2001) Effects of matrix metalloproteinase-9 gene knock-out on the proteolysis of blood-brain barrier and white matter components after cerebral ischemia. J. Neurosci. 21, 7724–7732.
Asahi M., Asahi K., Jung J.C., del Zoppo G.J., Fini M.E., and Lo E.H. (2000) Role for matrix metalloproteinase 9 after focal cerebral ischemia:effects of gene knockout and enzyme inhibition with BB-94. J. Cereb Blood Flow Metab. 20, 1681–1689.
Fujimura M., Gasche Y., Morita-Fujimura Y., Massengale J., Kawase M., and Chan P.H. (1999) Early appearance of activated matrix metalloproteinase 9 and blood-brain barrier disruption in mice after focal cerebral ischemia and reperfusion. Brain Res. 842, 92–100.
Heo J.H., Lucero J., Abumiya T., Koizol J.A., Copeland B.R., and del Zoppo G.J. (1999) Matrix metalloproteinases increase very early during experimental focal cerebral ischemia. J. Cereb. Blood Flow Metab. 19, 624–633.
Mun-Bryce S. and Rosenberg G.A. (1998) Matrix metalloproteinases in cerebro-vascular disease. J. Cereb. Blood Flow Metab. 18, 1163–1172.
Rosenberg G.A., Navratil M., Barone F., and Feuerstein G. (1996) Proteolytic cascade enzymes increase in focal cerebral ischemia in rat. J. Cereb. Blood Flow Metab. 16, 360–366.
Anthony D.C., Miller K.M., Fearn S., Townsend M.J., Opdennaker G., Wells G.M.A., et al. (1998) Matrix metalloproteinase expression in an experimentally induced DTH model of multiple sclerosis in rat CNS. J. Neuroimmunol. 87, 62–72.
Rosenberg G.A., Estrada E.Y., and Dencoff J.E. (1998) Matrix metalloproteinases and TIMPs are associated with blood-brain barrier opening after reperfusion in rat brain. Stroke 29, 2189–2195.
Jiang X., Namura S., and Nagata I. (2001) Matrix metalloproteinase inhibitor KB-R7785 attenuates brain damage resulting from permanent focal cerebral ischemia in mice. Neurosci. Lett. 305, 41–44.
Romanic A.M., White R.F., Arleth A.J., Ohlstein E.H., and Barone F.C. (1998) Matrix metalloproteinase expression increases after cerebral focal ischemia in rats:inhibition of matrix metalloproteinase-9 reduces infarct size. Stroke 29, 1020–1030.
Lapchak P.A., Chapman D.F., and Zivin J.A. (2000) Metalloproteinase inhibition reduces thrombolytic (tissue plasminogen activator)-induced hemorrhage after thromboembolic stroke. Stroke 31, 3034–3040.
Rosenberg G.A., Estrada E.Y., Dencoff J.E., and Stetler-Stevenson W.G. (1995) Tumor necrosis factor-alpha-induced gelatinase B causes delayed opening of blood-brain barrier: an expanded therapeutic window. Brain Res. 703, 151–155.
Lee S. and Lo E.H. (2003) Induction of caspase-mediated cell death by matrix metalloproteinase after hypoxia-reoxygenation in human brain microvascular endothelial cells. Stroke 34, 250 (abstr).
Fini M.E., Cook J.R., Mohan R., and Brinckerhoff C.E. (1998) Regulation of matrix metalloproteinase gene expression, in Matrix Metalloproteinases (Parks W.C. and Mecham R.P.) Academic Press, New York, NY pp. 299–356.
Wang X., Mori T., Jung J-C., Fini M.E., and Lo E.H. (2002) Secretion of matrix metalloproteinase-2 and -9 after mechanical trauma injury in rat cortical cultures and involvement of MAP kinase. J. Neurotrauma 19, 615–625.
Morita-Fujimura Y., Fujimura M., Gasche Y., Copin J., and Chan P.H. (1999) Overexpression of copper and zinc superoxide dismutase in transgenic mice prevents the induction and activation of matrix metalloproteinases after cold injury induced brain trauma. J. Cereb. Blood Flow Metab. 20, 130–138.
Ahn M.Y., Zhang Z.G., Tsang W., and Chopp M. (1999) Endogenous plasminogen activator expression after embolic focal cerebral ischemia in mice. Brain Res. 837, 169–176.
Hosomi N., Lucero J., Heo J.H., Koziol J.A., Copeland B.R., and del Zoppo G.J. (2001) Rapid differential endogenous plasminogen activator expression after acute middle cerebral aretry occlusion. Stroke 32, 1341–1348.
Yepes M., Sankvist M., Wong M.K.K., Coleman T.A., Smith E., Cohan S.L., et al. (2000) Neuroserpin reduces cerebral infarct volume and protects neurons from ischemia-induced apoptosis. Blood 96, 569–576.
NINDS rt-PA Stroke Study Group. (1995) Tissue plasminogen activator for acute ischemic stroke. N. Engl. J. Med. 333, 1581–1587.
Larrue V., von Kummer R., del Zoppo G., and Bluhmki E. (1999) Hemorrhagic transformation in acute ischemic stroke:potential contributing factors in the ECASS study. Stroke 28, 957–960.
NINDS rt-PA Stroke Study Group. (1997) Intracerebral hemorrhage after intravenous TPA therapy for ischemic stroke. Stroke 28, 2109–2118.
Sumii T. and Lo E.H. (2002) Involvement of matrix metalloproteinase in thrombolysis-associated hemorrhagic transformation after embolic focal ischemia in rats. Stroke 33, 831–836.
Montaner J., Alvarez-Sabin J., Molina C.A., Angles A., Abilliera S., Arenillas J., et al. (2001) Matrix metalloproteinase expression is related to hemorrhagic transformation after cardioembolic stroke. Stroke 32, 2762–2767.
Maeda M., Furuichi Y., Uetama N., Moriguchi A., Satoh N., Matsuoka N., et al. (2002) A combined treatment with tacrolimus (FK506) and recombinant tissue plasminogen activator for thrombotic focal cerebral ischemia in rats: increased neuroprotective efficacy and extended therapeutic time window. J. Cereb. Blood Flow Metab. 22, 1205–1211.
Pozzi A., Moberg P.E., Miles L.A., Wagner S., Soloway P., and Gardner H.A. (2000) Elevated matrix metalloprotease and angiostatin levels in integrin alpha 1 knockout mice cause reduced tumor vascularization. Proc. Natl. Acad. Sci. USA 97, 2202–2207.
Aoudjit F., Ptoworowski E.F., and St-Pierre Y. (1998) Bi-directional induction of matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 during T lymphoma/endothelial cell contact: implication of ICAM-1. J. Immunol. 160, 2967–2973.
May A.E., Kalsch T., Maasberg S., Herouy Y., Schmidt R., and Gawaz M. (2002) Engagement of glycoprotein IIb/IIa (alpha-IIb/beta-3) on platelets upregulates CD40L and triggers CD40L-dependent matrix degradation by endothelial cells. Circulation 106, 2111–2117.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X., Lo, E.H. Triggers and mediators of hemorrhagic transformation in cerebral ischemia. Mol Neurobiol 28, 229–244 (2003). https://doi.org/10.1385/MN:28:3:229
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/MN:28:3:229