Abstract
BACKGROUND AND PURPOSE: Cerebral veins show wide variation, and unexpected veins occasionally limit surgical procedures for clipping of cerebral aneurysms. This study was undertaken to assess the utility of three-dimensional CT angiography (3D-CTA) in the evaluation of venous anatomy before surgical clipping of cerebral aneurysms.
METHODS: 3D-CTA was performed in 19 patients before pterional craniotomy. The venous anatomy visualized by use of 3D-CTA was compared retrospectively with intraoperative findings.
RESULTS: 3D-CTA clearly depicted the superficial sylvian veins (SSVs) in 17 of 19 cases. In three of these 17 cases, 3D-CTA failed to reveal the additional connecting veins from SSVs to the midportion of the sphenoid wing. 3D-CTA also depicted bridging veins from: 1) the basal vein of Rosenthal to the sphenoparietal sinus (SPS) (three cases); 2) a junction of the anterior cerebral vein and the deep middle cerebral vein to the SPS (two cases); 3) the anterior cerebral vein into the cavernous sinus (one case); and 4) the deep middle cerebral vein to the cavernous sinus in one case. Surgical findings corresponded well with the angiographic findings. 3D-CTA depicted bridging veins from the frontal base into the SPS in two cases; retraction of the frontal lobe was severely limited in these cases. 3D-CTA also exhibited anatomic variation of deep cerebral veins, which suggests the utility of 3D-CTA in evaluating the risks carried by sacrifice of some bridging veins. In two patients, aneurysmal dissection was restricted because of adhering veins. 3D-CTA depicted these cerebral veins to be lying in contact with the aneurysm. In one case, the contiguous vein was hidden behind a large middle cerebral artery aneurysm; 3D-CTA was helpful for visualization of this vein.
CONCLUSION: 3D-CTA can clearly depict the venous anatomy of the brain and is useful for surgical planning of a pterional or temporopolar approach for cerebral aneurysms.
The advancing technology of spiral CT imaging has resulted in improved delineation of cerebral aneurysms by three-dimensional CT angiography (3D-CTA) (1–5). Recent studies have suggested that 3D-CTA is equal or superior to conventional angiography in characterizing the aneurysms for surgical planning (6–9). Previous studies have focused mainly on the shape of the aneurysms and the anatomic relation of the aneurysmal sac to the parent artery and its branches (1–3, 5, 7, 9) and have not focused on venous images. 3D-CTA can depict cerebral veins simultaneously with cerebral arteries and skull base structures. The reconstructed 3D-CTA images can be rotated 360° in any plane. Thus, 3D-CTA provides better representation of the spatial relation between the cerebral veins and the surrounding structures than that provided by conventional angiography. Cerebral veins show much more anatomic variation than do cerebral arteries (10, 11), and unexpected cerebral veins in the sylvian fissure occasionally limit surgical procedures for cerebral aneurysms via the pterional approach (12). Sacrifice of these veins, however, can result in hemorrhagic infarction. Thus, preoperative visualization of veins that would limit opening of the sylvian fissure is important in determining the surgical approach to cerebral aneurysms. Furthermore, by evaluating the collateral venous channels with 3D-CTA, we can estimate the risk involved in sacrifice of these veins. A cerebral vein lying contiguous to the aneurysm presents a clipping problem with respect to surgical dissection. Cerebral veins hidden behind large aneurysms are not always detected before application of the clip. Prediction of these veins is also helpful in reducing surgical complications resulting from venous injury or venous occlusion.
At our institution, preoperative 3D-CTA is performed in addition to conventional angiography in patients with incidentally identified aneurysms or in patients with ruptured aneurysms who are scheduled for clipping at the late stage of subarachnoid hemorrhage. In the present study, we compared venous images obtained with 3D-CTA with operative findings. We present our findings herein and discuss the significance of anatomic evaluation of cerebral veins in planning for aneurysmal neck clipping via the pterional surgical approach.
Methods
Patients
Between December 1997 and March 2000, 23 patients were examined by spiral CT before surgery for aneurysms. Of these 23 patients, 19 underwent clipping via the pterional approach; four patients treated via the interhemispheric approach were excluded from the study. Eight of the patients were men and 11 were women (age range, 43–78 years; mean age, 61.8 years). Aneurysms were identified incidentally by “brain check-up” in 13 of these patients. Incidental aneurysms were also found during extensive examinations for polycystic kidney (n = 2), chronic headache (n = 1), previous history of aneurysm (n = 1), and third-nerve paresis (n = 1). In the remaining patient, spiral CT was performed before clipping of a ruptured aneurysm in the late stage of subarachnoid hemorrhage.
Spiral CT, Reconstruction, Maximum Intensity Projection, and 3D-CTA
Spiral CT was performed with a HiSpeed Advantage scanner (General Electric Medical Systems, Milwaukee, WI). Nonionic contrast medium (100 mL of 300-mgI/mL solution) was injected with a pump into an antecubital vein at a rate of 2.5 or 3.0 mL/s. After a delay of 20 seconds, 35 to 40 helical scans were obtained at 1-mm section thickness, 1 mm/s table speed, a 512 × 512 matrix, 120 kV, and 100–150 mA. The axial source images were reconstructed to 0.5 mm and transferred to the Advantage Windows workstation (General Electric Medical Systems). At the workstation, multiplanar reconstruction, maximum intensity projection, and 3D-CTA images were produced. Images for 3D-CTA were obtained mainly by a volume-rendering method with a minimum density threshold of approximately 60–80 HU and maximum density threshold of 300–400 HU. For patients enrolled earliest, a shaded surface display with a lower limit of approximately 80 HU was used for processing of the 3D-CTA images.
Angiography
Cerebral angiograms were obtained by digital subtraction angiography. All angiograms included standard anteroposterior, lateral, and oblique projections, with the addition of other views when indicated. In occasional cases, the conventional cut-film technique was used.
Results
Bridging Veins
3D-CTA clearly depicted the superficial sylvian veins (SSVs) in 17 of 19 patients (Fig 1). The final draining point from the SSVs into the sphenoparietal sinus (SPS) at the midportion of the sphenoid wing was revealed in 11 patients; it was depicted at the inner portion of the sphenoid wing in four. These connections were confirmed at surgery in all cases. 3D-CTA showed that the SSVs connected directly to the cavernous sinus in two patients. In two other patients, the SSVs were not clearly depicted by 3D-CTA; they were found to be hypoplastic during surgery. Intraoperatively, additional connecting veins between the SSVs and the SPS were found at the midportion of the sphenoid wing in one of four patients in whom the SSVs emptied at the inner portion of the sphenoid wing and in both patients in whom the SSVs emptied into the cavernous sinus. However, 3D-CTA had not clearly shown these additional connecting veins.
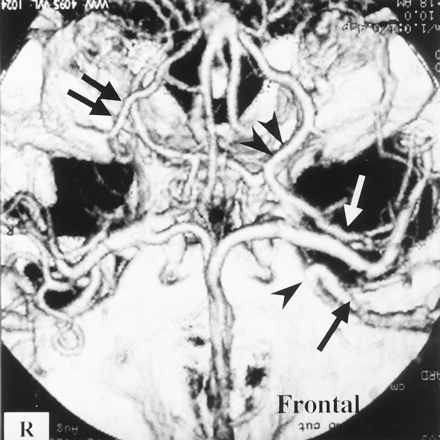
Patient with a left internal carotid artery aneurysm.
3D-CTA (superior view) shows drainage of well-developed SSVs (black arrow) into the SPS at the inner portion of the sphenoid wing (black arrowhead) on the left side. A well-developed DMCV (white arrow) connecting to the BVR (double black arrowheads) is also visualized on the left side. DMCV and BVR development (double black arrows) on the right side is poor compared with that on the left side.
In addition to revealing drainage from the SSVs to the SPS, 3D-CTA revealed bridging veins from the basal vein of Rosenthal (BVR) to the SPS (Figs 2 and 3) in three cases and from the junction of the anterior cerebral vein and the deep middle cerebral vein (DMCV) to the SPS in two (Fig 4). These bridging veins were confirmed during surgery, but they did not preclude retraction of the frontal and temporal lobes in any of the five cases. Venous drainage was depicted by 3D-CTA to flow from the frontal base into the SPS in two cases; the insular veins in one (Fig 5) and the fronto-orbital veins in the other (Fig 6). Retraction of the frontal lobe was limited in both cases. In one case (Fig 5), we were unable to continue clipping the aneurysm via the pterional approach, because the bridging veins anchoring the frontal lobe prevented complete dissection of the anterior communicating artery (AcomA) aneurysm.
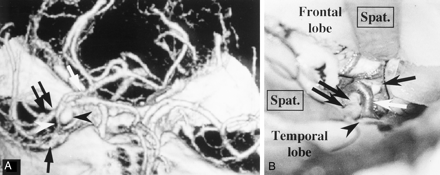
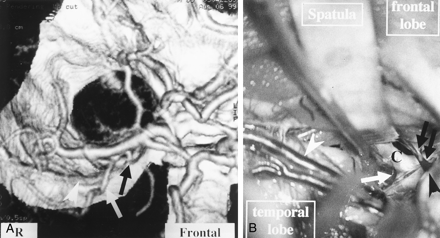
Patient with a right middle cerebral artery aneurysm.
A, 3D-CTA (right anterior view) shows the connecting vein (white arrowhead) between the BVR (white arrow) and the SPS. The connecting vein lies in contact with the aneurysm (black arrowhead). A black arrow and double black arrows indicate the SSVs and right middle cerebral artery, respectively.
B, A microscopic operative photograph shows that the right sylvian fissure is opened by the pterional approach. Spatulas (Spat.) retract the right frontal and right temporal lobes. The connecting vein between the BVR and the SPS (white arrow) adheres strongly to the middle cerebral artery (black double arrows) and the aneurysm (black arrow). A black arrowhead indicates the SSVs.
Patient with a left middle cerebral artery aneurysm. 3D-CTA (superior view) suggests strong adhesion (double black arrows) of an insular vein (white arrow) to the cerebral aneurysm (white arrowhead). In this case, a connecting vein (black arrow) between the BVR (double white arrows) and the SPS, SSVs (black arrowhead), and an aneurysmal bleb (*) are also revealed.
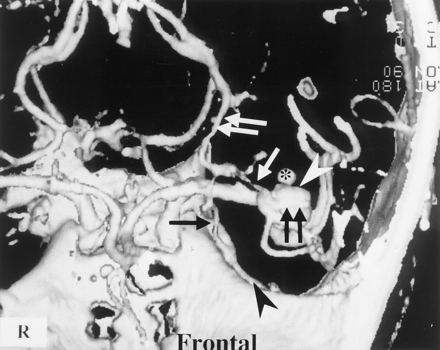
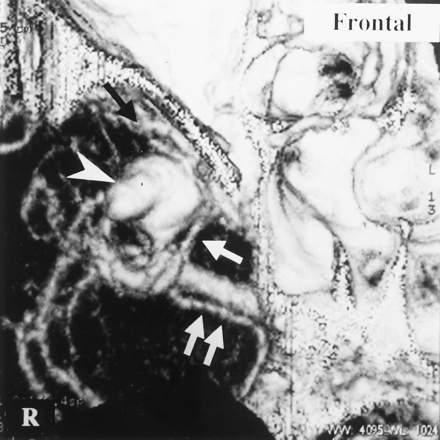
Patient with a left internal carotid artery aneurysm.
A, 3D-CTA (superior view) shows the left DMCV (arrow) emptying into the SPS and poor BVR development on the left side compared with that on the right side (double arrows).
B, 3D-CTA (left anterior view) reveals that the anterior cerebral vein (black arrow) and DMCV (double white arrows) join and empty into the SPS (white arrow). 3D-CTA also shows these veins in front of a left internal carotid artery aneurysm (*) but not in contact with it. C indicates the proximal portion of the left internal carotid artery.
C, A microscopic operative view shows that the left frontal lobe and the left temporal lobe are retracted to expose the left internal carotid artery aneurysm (*). A black arrow indicates the proximal portion of the left internal carotid artery. The connecting vein (white arrow) depicted in B was smoothly detached from the aneurysm and the internal carotid artery, but application of the clip was disturbed by this vein.
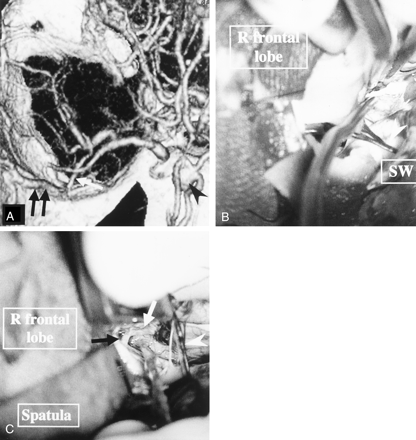
Patient with an AcomA aneurysm.
A, 3D-CTA (superior view) shows the insular vein (white arrow) emptying into the SPS. A black arrowhead and double black arrows indicate the AcomA aneurysm and the SSVs.
B and C, Via the pterional approach to the AcomA aneurysm, dissection of the aneurysm was attempted rostral (B) and caudal (C) to the bridging veins from the insular vein to the SPS (microscopic operative views). The white arrowhead, white arrow, black arrow, and SW indicate the bridging veins, the left anterior cerebral artery, the right anterior cerebral artery, and the sphenoid wing, respectively.
Retraction of the right frontal lobe was restricted, because the right frontal lobe was anchored to these bridging veins. Therefore, the aneurysmal neck was not completely explored. Because 3D-CTA depicted poor venous drainage from the insular cortex to the BVR, as shown in A, we could not sacrifice these veins and halted clipping of the aneurysm with this approach.
Patient with a right internal carotid artery aneurysm.
A, 3D-CTA (superior view) reveals the fronto-orbital vein (white arrow) emptying into the SPS at the inner portion of the sphenoid wing (black arrow) A white arrowhead indicates the SSVs.
B, A microscopic operative photograph shows that the right frontal lobe was retracted by the spatula to expose the right internal carotid artery (C). The aneurysm (white arrow) was barely clipped, although the bridging vein between the fronto-orbital vein and the SPS (black arrows) limited retraction of the frontal lobe. The black arrowhead indicates the draining point of this bridging vein into the SPS. The white arrowhead indicates the SSVs.
Drainage into the cavernous sinus was observed in two cases; one from the anterior cerebral vein and the other from the DMCV. These connecting veins into the cavernous sinus did not preclude retraction of the frontal or temporal lobe.
Variation of venous channels in the deep cerebral veins was also clearly shown by 3D-CTA (Figs 1 and 4A).
Cerebral Veins Contiguous to Aneurysm
3D-CTA depicted cerebral veins contiguous to the aneurysm in eight cases. Strong adhesion of the cerebral vein to the aneurysm was found during surgery in two of these cases. 3D-CTA depicted these veins to be lying in contact with the aneurysm in both cases (Figs 2 and 3). In the other six cases, the cerebral veins were easily dissected from the aneurysms; 3D-CTA had shown gaps between these structures (Fig 7).
Patient with a ruptured right middle cerebral artery aneurysm.
3D-CTA (inferior view) shows a connecting vein (arrow) between the BVR (double arrows) and the SPS contiguous to the right middle cerebral artery aneurysm (white arrowhead), but it also depicts a gap between the aneurysm and the connecting vein. A black arrow indicates the SSVs.
Discussion
3D-CTA depicts the cerebral arteries and veins simultaneously, allowing for stereoscopic correlation of the cerebral veins to the surrounding arteries and bone structures. Some authors (3, 6) have reported that the simultaneous enhancement sometimes makes it difficult to distinguish the veins from the arteries. In our experience, however, this problem can be resolved by rotation of the reconstructed image and comparative evaluation of 3D-CTA and digital subtraction angiography images. A recent study (13) reported that 3D-CTA can clearly delineate cerebral veins in the skull base and indicated that 3D-CTA was useful in the preoperative planning of aneurysmal clipping. However, few studies have correlated the venous images obtained by 3D-CTA to surgically observed findings.
In the pterional approach to aneurysm surgery, opening the sylvian fissure and retracting the frontal or temporal lobe are fundamental to accessing the aneurysm. The cerebral veins across the sylvian fissure sometimes preclude these operative procedures. Connecting veins to the SPS from the frontal lobe or temporal lobe may limit retraction of these structures, because they anchor the frontal or temporal lobe to the skull base. The cerebral veins from the frontal base, such as the fronto-orbital or insular veins usually empty into the BVR (10, 11) (Fig 8A). However, the venous system of the brain is of considerable anatomic variation (10, 11). In two of the present cases, fronto-orbital veins or insular veins emptied into the SPS, and these bridging veins from the frontal base limited retraction of the frontal lobe (Fig 8B). An AcomA aneurysm could not be clipped via the pterional approach in one case (Fig 5). An internal carotid artery aneurysm was barely clipped in another case, and we speculate retrospectively that access to the AcomA in this case also would have been very difficult, because anchoring of the frontal lobe by the bridging vein would strongly limit retraction of the frontal lobe (Fig 6). AcomA aneurysms can be accessed via either side of the pterional approach. They also can be clipped via the interhemispheric approach. Our results suggest that 3D-CTA examination of the venous anatomy surrounding the sylvian fissure may become an important determining factor in the selection of a surgical approach to the AcomA aneurysm.
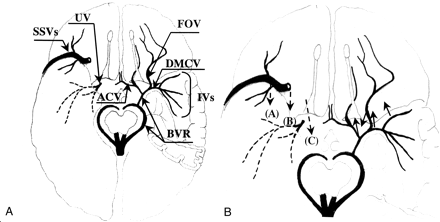
Diagram of the cerebral veins around the sylvian fissure (inferior view).
A, Representative venous-draining system is revealed. ACV, BVR, DMCV, FOV, IVs, SSVs, and UV indicate the anterior cerebral vein, basal vein of Rosenthal, deep middle cerebral vein, fronto-orbital vein, insular veins, superficial sylvian veins, and uncal vein, respectively.
B, Bridging veins from the IVs, DMCV, FOV, and BVR to the SPS are indicated by solid arrows. The draining veins from the SSVs to the SPS or cavernous sinus are revealed by broken arrows. The final draining points are depicted at the midportion of the sphenoid wing in 11 patients (A), at the inner portion of the sphenoid wing in four (B), and the cavernous sinus in two (C).
Bridging veins from the BVR to the SPS were revealed by 3D-CTA in three cases. In these cases, the anatomic relations among the middle cerebral artery, DMCV, BVR, and SPS were clearly depicted by 3D-CTA. In a case of a middle cerebral artery aneurysm (Fig 2), the bridging vein adhered to the aneurysm. In this case, we managed to dissect the vein from the aneurysm and clip the aneurysm without sacrificing the vein. However, 3D-CTA in this case showed a well-developed venous draining system from the DMCV to the vein of Galen via the BVR, suggesting that this bridging vein could have been sacrificed without complication. 3D-CTA showed that the anterior cerebral vein and the DMCV joined and then emptied into the SPS in two cases. In one case (Fig 4), the bridging vein lay in front of the aneurysm of the left internal carotid artery, which precluded application of a clip. We did not sacrifice this vein, however, because the BVR was hypoplastic and the DMCV was shown to connect directly with the SPS. Yasargil (12) suggested that attempts to preserve these delicate vascular structures during dissection would occasionally prove futile and that these veins should be sacrificed only rarely. The present study suggests that anastomosis of the deep venous system is clearly revealed by 3D-CTA and that the risk in sacrificing these veins can be evaluated by this venous imaging technique.
The main venous connections beween the SSVs and SPS or the cavernous sinus were clearly depicted by 3D-CTA in all but two cases (Fig 8B). In the remaining two cases, the SSVs were found to be hypoplastic during surgery and were not verified by 3D-CTA. In three of six patients in whom the SSVs emptied into the SPS at the inner portion of the sphenoid wing or the cavernous sinus, venous connection between the SSVs and the SPS at the midportion of the sphenoid wing was also found during surgery. These additional venous connections were not clearly shown by 3D-CTA, probably because they were hidden under well-developed SSVs. Bridging veins from SSVs to the SPS are not usually sacrificed in the pterional approach for aneurysms of the anterior circulation. In contrast, bridging veins from SSVs to the SPS at the temporal tip must be sacrificed in the temporopolar approach for basilar artery aneurysms (14). The locations of bridging veins and development of anastomotic channels of the venous system should be carefully evaluated in this approach. The present study indicates that 3D-CTA provides useful information in the development of SSVs, final drainage points of the SSVs, and anastomotic venous channels between the SPS, DMCV, and BVR. It cannot, however, clearly depict additional connecting veins between the SSVs and the SPS in patients in whom the SSVs ultimately drain into the inner portion of the SPS or cavernous sinus.
In aneurysm surgery, identification of veins contiguous to aneurysms, especially easily overlooked cerebral veins hidden behind large aneurysms, is important to dissection and placement of the clip without complication. Adhesion of the cerebral vein seriously precludes the dissection of the aneurysm. In six cases, 3D-CTA depicted cerebral veins contiguous to the aneurysm but not in contact with it. In these six cases, the cerebral vein was easily dissected from the aneurysm. In contrast, 3D-CTA showed the cerebral vein to be lying in contact with an aneurysm in two cases, and strong adhesion of the vein to the aneurysm was found during surgery in both cases. We do not think that adhesion of the vein to the aneurysm can be accurately predicted on the basis of imaging. However, cerebral veins in contact with the aneurysm can strongly suggest adhesion of these structures. Although cerebral arteries and veins are simultaneously depicted on 3D-CTA images, we can differentiate veins lying contiguous to aneurysms and arterial branches by comparison of 3D-CTA images with digital subtraction angiograms. Our results also suggest that 3D-CTA can characterize the cerebral vein contiguous to an aneurysm.
Conclusion
Cerebral veins exhibit many anatomic variations, and 3D-CTA can clearly depict the layout of certain veins. Cerebral veins also can be stereoscopically correlated with surrounding arteries and bone structures. We conclude that 3D-CTA will allow for informed selection of the best surgical approach to the AcomA aneurysm. Furthermore, 3D-CTA exploration of anastomotic channels of deep cerebral veins will be helpful in determining the risk of sacrifice. It is also indicated that predicting surgical risk by evaluation of venous anatomy will be helpful in selecting therapeutic procedures (eg, surgical clipping or endovascular obliteration).
References
- Received March 19, 2001.
- Accepted after revision August 16, 2001.
- American Society of Neuroradiology