Abstract
BACKGROUND AND PURPOSE: Diffusion tensor imaging (DTI) is a noninvasive technique that can be used to assess the integrity of cerebral tissue. The purpose of this study was to assess DTI measurements in the hippocampal formation (HF) and to investigate the role of DTI in lateralizing the seizure focus in temporal lobe epilepsy (TLE).
METHODS: We evaluated 12 patients with unilateral TLE and 14 healthy subjects. We collected diffusion-weighted images along six different directions with a b value of 1000 s/mm2, as well as an image acquired without diffusion weighting (b = 0 s/mm2). A 1.5-T imager was used to acquire 17 (3-mm) coronal sections covering the temporal lobes. We compared the mean diffusivity (trace D) and fractional anisotropy (FA) from symmetrical voxels by sampling the anterior HF bilaterally. We compared measurements with the EEG, high-resolution MR imaging, and clinical information.
RESULTS: The patient group had significantly increased diffusivity and decreased FA in the HF ipsilateral to the seizure focus, as compared with values in the contralateral HF. When compared with healthy subjects, patients had significantly higher mean diffusivity in the ipsilateral HF; ipsilateral FA values were lower and did not reach statistical significance. Measurements in the contralateral HF did not show differences. Left-right and absolute diffusivity indices lateralized the abnormal HF in eight and five of 12 patients, respectively.
CONCLUSION: Abnormal DTI measurements and the epileptogenic HF are associated in unilateral TLE. This finding may reflect hippocampal sclerosis and may aid in presurgical evaluation.
Diffusion tensor imaging (DTI) is a new imaging technique that can be used to noninvasively assess the molecular and biochemical environment of cerebral tissue (1–2). DTI can aid in characterizing and measuring the diffusive transport of water molecules by means of an effective diffusion tensor, D. These symmetric tensor measurements contain useful information about the tissue microstructure and architecture. Of the several indices used to characterize diffusion tensor, those most commonly used are the trace of the tensor, which measures mean diffusivity (trace D), and fractional anisotropy (FA) (3–4). These characteristic measurements may represent the changes in cerebral structure that occur in various neurologic conditions (1, 2). DTI has been shown to be useful in the study of diseases, such as cerebral ischemia (5), acute stroke (6), multiple sclerosis (7) and schizophrenia (8).
Noninvasive MR imaging techniques are becoming increasingly important in lateralizing and localizing the seizure focus in a noninvasive manner. Only a few studies have addressed the utility of DTI in epilepsy (9–11). In general, these studies have demonstrated increased diffusivity and decreased FA in cerebral tissue, which corresponded to the seizure focus in the entire group of the patients evaluated. However, these studies included a mixture of temporal epilepsy and extratemporal epilepsy with or without lesions. In addition, these studies failed to demonstrate a high yield of abnormal DTI measurements in individualized patients. A recent study evaluated DTI of white matter in temporal lobe epilepsy (TLE) (12). In that study, significantly lower diffusion anisotropy and higher diffusivity in directions perpendicular to the axons were detected in several white matter structures in the patients, when compared with control subjects. However, none of these measured structures were in the temporal lobes. Another recent study (13) was conducted to evaluate apparent diffusion coefficient (ADC) values in patients with unilateral hippocampal sclerosis. The investigators found abnormal values on the side with hippocampal sclerosis in all patients, as compared with healthy volunteers. That study, however, was limited to unilateral hippocampal sclerosis findings on MR images, and it involved large regions of interest (ROIs) on axial sections, which are subject to partial-volume effects.
The purpose of this study was to obtain DTI measurements of the hippocampal regions from thin coronal sections in patients with clinically proved TLE and in healthy control subjects. In addition, we investigated the diagnostic utility of DTI in detecting hippocampal abnormalities and in lateralizing the temporal-lobe seizure focus in a consecutive series of patients with TLE who have been evaluated for epilepsy surgery at our comprehensive epilepsy center. Furthermore, we compared the DTI measurements and high-resolution brain MR images in these patients.
Methods
We evaluated 12 patients with unilateral nonlesional TLE and 14 healthy subjects by using DTI of the brain. Their age and sex information is provided in Tables 1 and 2. The age range was 27–59 years for the patient group (mean age, 41.42 years; SD, 8.52). The control group had an age range of 24–49 years (mean age, 31.29 years; SD 8.19). Informed consent was obtained from all subjects, per the guidelines of the institutional review board. The diagnosis of unilateral TLE was based on clinical and EEG findings after a careful review of the patient’s medical history, high-resolution brain MR images, routine EEGs, and continuous audiovisual/EEG monitoring results. This information was interpreted by a board-certified epileptologist and electrophysiologist (B.A.A.) who was blinded to the DTI quantitative measurements. This process resulted in the diagnosis of definite unilateral TLE proved with unilateral ictal EEG recordings during continuous audiovisual/EEG monitoring in nine patients and in the diagnosis of probable unilateral TLE based on exclusively unilateral interictal EEG findings during prolonged continuous audiovisual/EEG monitoring and other concordant clinical information in three patients. This classification was similar to that used by Arfanakis et al (12) for TLE diagnosis.
Summary of the patient population evaluated with DTI
Summary of the control-subject population evaluated with DTI
DTI was performed in all patients after they had been seizure-free for at least 24 hours. This approach was intended to ensure measurements from a baseline state as much as possible, because prior studies indicated that seizures from status epilepticus may affect diffusivity measurements (14–15). DTI was performed by using a 1.5-T MR unit (Vision; Siemens Medical Systems, Erlangen, Germany), with a single-shot echo-planar diffusion-weighted imaging sequence. To fully determine the diffusion tensor, we collected diffusion-weighted images along six different directions with a b value of 1000 s/mm2, as well as an image without diffusion weighting (b = 0 s/mm2). Seventeen 3-mm coronal sections were acquired to cover the entire temporal lobes. The imaging parameters included the following: TR/TE, 6000/100; FOV, 240 mm; matrix, 98 × 128; and four acquisitions. The total imaging time to collect the DTI images was 4.5 minutes.
After image acquisition, the data were transferred to an independent workstation (Silicon Graphics, Mountain View, CA) for calculation of the DTI indices. The maps of mean diffusivity (rotationally invariant scalar indices of diffusion) and FA were calculated from the diffusion-weighted images by using a method based on that proposed earlier (3, 4, 16). Software written in IDL (Interactive Data Language, Boulder, CO) was used. See the Appendix for details.
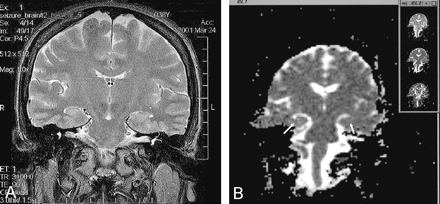
We compared the mean diffusivity and FA indices from symmetrical-voxel sampling regions from the anterior hippocampal formation (HF) on both sides, in patients and in control subjects. The ROIs were selected on the coronal section from a 4 × 8-mm rectangular area drawn in the center of the anterior HF. The selections were made by a neurologist (K.J.A.-K.) and a board-certified neuroradiologist (S.H.F.) who were blinded to the side of the seizure focus, the patient’s history. and other clinical information. Figure 1A shows the coronal T2-weighted image through a section showing the temporal lobes. Figure 1B shows the placement of the ROIs in the left and right HFs.
Images in a patient with left-sided TLE.
A, Coronal T2-weighted MR image. Note the abnormal anterior hippocampus with mild deformation on the left side compared with the right side.
B, DTI at the same coronal level as in A. Note the marking of the ROI in the left and right HFs. The images in the upper right corner correspond to the diffusion (B0, top), trace D (middle), and FA (bottom) images.
Summary data for the patient group and the control subjects, along with the relevant medical history and DTI measurements are shown in Tables 1 and 2. In our analysis, we compared the group HF mean diffusivity and FA measurements ipsilateral with the epileptogenic side to the measurements in the contralateral HF. We also compared the group ipsilateral and contralateral DTI measurements in the patient population with the control values derived from a total of 28 HF measurements on both sides in all 14 healthy subjects. In addition, we calculated the left-right diffusivity and FA indices of the patient group and compared them with the mean left-right index of the normal group. The significance of the differences was assessed by using the Student t test. Only differences with P < .05 were considered significant. Subsequently, we identified the individual subjects with TLE who had a significant difference in left-right DTI measurement and also those with DTI measurements differing from the mean for the control subjects. When identifying these patients with abnormal values, we compared the abnormal side in the patients with the mean value of the corresponding side in the control group. We later compared the DTI measurements with the eventual side of TLE, with the high-resolution MR images, and with the other clinical information (Tables 1 and 2).
Results
The statistical measurements of mean diffusivity and FA ipsilateral and contralateral to the epileptogenic HF in the patient group and the entire measurements in the control group are summarized in Tables 3 and 4. There was no statistically significant difference in diffusivity or FA measurements between the sides in the control measurements, with a mean diffusivity of 0.84 and 0.83 on the left and right sides, respectively, and a mean FA of 0.0146 and 0.0152 on the left and right sides, respectively. The mean diffusivity and FA values for all 28 HF measurements in the control group were 0.84 ± 0.03 and 0.015 ± 0.007, respectively.
Summary of statistical measurements of diffusivity and FA in the patient group
Summary of statistical measurements of diffusivity and FA in the healthy control group
The patient group, however, had significantly increased mean diffusivity and decreased FA values in the HF ipsilateral to the seizure focus, when compared with the contralateral HF measurements, and with T(11) = −2.55 and T(11) = 4.14 (P < .05 and P < .05, respectively). When the patient group was compared with the control subjects, the ipsilateral HF had significantly increased mean diffusivity, with T(24) = 2.73 and P < .05. Ipsilateral FA was decreased, but the difference did attain statistical significance, with T(24) = −1.82 and P = .08. The contralateral HF measurements did not demonstrate differences when compared with those of the control group.
When left-right diffusivity measurements for individual patients were compared with those of healthy subjects, eight of 12 patients (patients 1, 2, 4, 7–10, and 12; sensitivity of 66%) had values that were 2 SDs beyond the mean, while three of 12 patients (patients 1, 7, and 9) had abnormal left-right FA measurements that were 2 SDs below or above the mean. When the individual mean diffusivity measurements were compared with the mean value of healthy subjects on the same side, five of 12 patients (patients 1, 2, 7, 9, and 12 in Table 1; sensitivity of 42%) had increased diffusivity measurements beyond 2 SDs from the mean for the control subjects. The contralateral HF measurements did not demonstrate differences when compared with those of the healthy control group.
We compared the sex and age in both the patient group and the healthy subject group. There was no statistically significant difference in sex, with χ2(1) = 2.48 and P < .12. There was a 10.13-year mean difference in age between the patient group and the control group, with T(23.1) = 3.08 and P < .05. However, age was not correlated with any of the dependent measures (the DTI measurements) therefore; an analysis of covariance for age was not conducted.
The review of clinical information revealed that both high-resolution brain MR imaging results and left-right DTI measurements were lateralizing in eight of 12 patients, whereas they were normal in four cases (Table 1). The results were concordant in six patients and complementary to each other in four other cases. In only two of 12 patients were both measurements normal. Neither imaging technique demonstrated discordant findings with the side of the epilepsy or with the other technique. Three patients (patients 1, 6, and 7) underwent anteromesial temporal lobectomy and became seizure-free. In these patients, pathologic evaluation revealed temporal-lobe sclerosis with gliosis and a diminished neuronal count. Other clinical information is summarized in Table 1.
Discussion
The hippocampus is still an essential structure in the epileptogenicity of TLE, and hippocampal sclerosis is the major pathology underlying nonlesional TLE (17–18). While visual inspection of high-resolution brain MR images and, often, volumetric HF measurements is the standard in evaluating brain MR abnormalities in patients with TLE, hippocampal atrophy on brain MR images is not always indicative of epileptogenicity (19–20). In this study, we investigated the role of DTI in lateralizing the seizure focus and the potential pathophysiology underlying DTI findings.
The patient group analyzed in this study demonstrated a significant difference in the mean diffusivity and FA measurements in the epileptogenic HF, as compared with values on the contralateral side. The finding of significantly increased mean diffusivity and the trend of decreased FA that did not reach statistical significance indicate that diffusivity measurements are more sensitive than FA in detecting the epileptogenic side, when values were compared with those of the healthy group. These findings concur with the results that Rugg-Gunn et al (10) reported in TLE patients. The observation may best be explained by the concept that expansion in the extracellular space, which causes increased diffusivity, is probably associated with a relatively preserved structural organization of the fiber bundle.
This study also demonstrates higher sensitivity with the left-right diffusivity index in lateralizing the epileptogenic temporal lobe than with other indices or findings in previous studies. In eight of our 12 patients (sensitivity of 66%), the left-right diffusivity difference was abnormal, when compared with the difference in the control group. Three of 12 patients had an abnormal left-right FA difference. The sensitivity of the left-right diffusivity index was also higher than that of the HF diffusivity index (sensitivity of 42%) in lateralizing the seizure focus. These represent significant findings in our study, as compared with the results of previous studies. Rugg-Gunn et el (10) found that, despite the sensitivity of the diffusivity index in what they called “acquired epilepsy with lesions,” diffusivity did not permit the identification of a clinically concordant abnormality in most their cases with what they called “cryptogenic epilepsy”; these included 15 patients with TLE. In their series, only one of 15 patients with TLE demonstrated individualized changes when investigators performed a voxel-by-voxel comparison by using the statistical parametric mapping model. While the results from the group comparison of diffusivity and anisotropy were statistically significant in our study as well as in theirs, we believe that the higher sensitivity of individualized left-right and direct HF diffusivity measurement in our study (66% and 42%, respectively) reflects the higher accuracy of our manual method. The selection of ROI in the anterior body of the HF from relatively thin (3-mm) sections is likely to produce accurate and consistent measurement from the HF while avoiding partial-volume effects. In addition, measurements in the cases of unilateral TLE in our study with relatively healthier contralateral HF could have produced left-right diffusivity difference more robust than that observed in a series with mixed unilateral and bilateral TLE. The increased sensitivity of the left-right measurement difference may be related to the inherent control of this index with regard to other variables that may affect the DTI measurements (such as patient age, age of onset, duration of epilepsy, among others).
Yoo et al (13) reported ADC measurements in 18 patients with TLE and unilateral MR imaging–positive hippocampal sclerosis. The researchers found significant abnormalities in all sclerotic hippocampal measurements, when compared with those in healthy subjects. Although DTI is more sensitive than ADC measurement, the higher sensitivity in their study is related to two factors: One pertains to their restricted selection of patients with TLE to those with unilateral hippocampal sclerosis, as shown on brain MR images, and the other factor relates to the selection of the ROI from axial images. This method is likely to produce a partial-volume effect that produces higher ADC values in the sclerotic hippocampi due to associated hippocampal atrophy and CSF sampling (13).
Our analysis was based on manual selection of the ROI as a region in the center of the anterior HF in all patient and healthy subjects. We avoided outlining the hippocampal margins to ensure complete sampling from the HF and to avoid CSF sampling. This method was consistent for multiple measurements from each subject and for the two investigators (K.J.A.-K., S.H.F.). In addition, the section thickness limited to 3 mm ensured sampling from only the HF. The measurements were unlikely to be affected by sampling outside the HF as a result of the convoluted shape of the HF in the healthy population and of the added deformation incurred in the case of mesial temporal sclerosis in TLE. The lack of significant differences in the left and right HF DTI measurements in the healthy population in our study further supports the consistency and validity of this manual method. The consistency and reproducibility of DTI measurements obtained by using the manual selection of ROI is also reported in other DTI studies of TLE in which ROIs were manually selected to measure DTI indices in the white matter (12).
This and other studies (9, 10, 13) indicate a potential role for DTI in localizing the seizure focus in TLE. High-resolution brain MR imaging is still the standard neuroimaging technique for lateralizing the seizure focus in TLE, but negative MR imaging results can be encountered in cases of unilateral TLE. In four of 12 patients, high-resolution brain MR findings were negative. Two of these patients had abnormal left-right HF diffusivity measurements. On the other hand, two of four patients with normal left-right HF diffusivity measurements had abnormal high-resolution brain MR images. This observation supports a complementary role of these two imaging modalities, although a study in larger patient sample may be needed to satisfactorily address this issue. This potential complementary role of DTI may be explained by the fact that each imaging technique is used to measure a relatively different aspect of the seizure focus. We believe that the improved technical design and understanding of the pathophysiology of DTI measurements may further improve the role of this imaging technique in evaluating patients with TLE. The improved sensitivity in our analysis technique probably accounts for the discrepancy between the significant differences with group analysis and the lack of difference between measurements in individualized patients and mean control subjects in other studies (10). In addition, further understanding of other factors influencing the DTI indices, such as the temporal occurrence and frequency of seizures and interictal spikes, is likely to better define the significance of these DTI measurements and will probably increase their sensitivity. Seizure activity in status epilepticus has been shown to decrease the diffusivity of water molecules (14, 15, 21). Furthermore, improved understanding would better clarify the role of DTI in the presurgical workup of patients with intractable TLE, in comparison to the role of high-resolution MR techniques, which have high sensitivity to changes in the mesial structures in mesial TLE (22).
We evaluated only patients with unilateral TLE to assess the sensitivity and utility of this refined DTI technique in a well-defined patient population. While this technique is demonstrably sensitive in identifying the epileptogenic hippocampus in this group of patients, further and larger studies in patients with TLE—including patients with bilateral independent temporal lobe seizures—are needed. Such studies may have to be controlled for seizure and spike occurrence, as well as for other variables (such as subjects’ age, age at seizure onset, and duration of epilepsy) to improve our understanding of the role of this technique in assessing the various TLE entities.
Conclusion
The results of this study demonstrate the ability of DTI to depict changes to the HF architecture and to lateralize the seizure focus in the entire group of patients and in individualized patients with TLE. Further studies are needed to delineate the role of this technique in the evaluation of patients with various categories of TLE.
Appendix
When using a method based on the work of Basser and Pierpaoli (16) to obtain an overall evaluation of the diffusivity in a region, one must avoid the anisotropic diffusion effects and limit the results to an invariant. One such invariant, the trace of the diffusion tensor was used to calculate the mean diffusivity in this study. The trace is given by Equation 1, as follows: 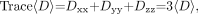 1) where Dxx, Dyy, and Dzz are the three diagonal elements of the diffusion tensor and their average, 〈D〉, is the mean diffusivity. Regarding FA, this invariant index measures the diffusion anisotropy of fiber structure, as shown in Equation 2:
1) where Dxx, Dyy, and Dzz are the three diagonal elements of the diffusion tensor and their average, 〈D〉, is the mean diffusivity. Regarding FA, this invariant index measures the diffusion anisotropy of fiber structure, as shown in Equation 2: 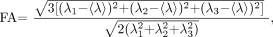 2) where 〈λ〉 = (λ1 + λ2 + λ3)/3 and λ1, λ2, and λ3 are the eigenvalues of the diagonalized diffusion tensor.
2) where 〈λ〉 = (λ1 + λ2 + λ3)/3 and λ1, λ2, and λ3 are the eigenvalues of the diagonalized diffusion tensor.
References
- Received March 6, 2003.
- Accepted after revision May 1, 2003.
- Copyright © American Society of Neuroradiology