Abstract
BACKGROUND AND PURPOSE: Gadobenate dimeglumine has proved advantageous compared with other gadolinium-based contrast agents for contrast-enhanced brain MR imaging. Gadobutrol is a more highly concentrated agent (1.0 mol/L). This study intraindividually compared 0.1-mmol/kg doses of these agents for qualitative and quantitative evaluation of brain tumors.
MATERIALS AND METHODS: Adult patients with suspected or known brain tumors underwent 2 identical MR imaging examinations at 1.5T, 1 with gadobenate dimeglumine and the other with gadobutrol, both at a dose of 0.1-mmol/kg body weight. The agents were injected in randomized order separated by 3–14 days. Imaging sequences and acquisition timing were identical for the 2 examinations. Three blinded readers evaluated images qualitatively for diagnostic information (lesion extent, delineation, morphology, enhancement, global preference) and quantitatively for CNR and LBR.
RESULTS: One hundred fourteen of 123 enrolled patients successfully underwent both examinations. Final diagnoses were intra-axial tumors, metastases, extra-axial tumors, “other” tumors, and “nontumor” (49, 46, 8, 7, and 4 subjects, respectively). Readers 1, 2, and 3 demonstrated preference for gadobenate dimeglumine in 46 (40.7%), 54 (47.4%), and 49 (43.0%) patients, respectively, compared with 6, 7, and 7 patients for gadobutrol (P < .0001, all readers). Highly significant (P < .0001, all readers) preference for gadobenate dimeglumine was demonstrated for all other qualitative end points. Inter-reader agreement was good for all evaluations (κ = 0.414–0.629). Significantly superior CNR and LBR were determined for gadobenate dimeglumine (P < .019, all readers).
CONCLUSIONS: Significantly greater morphologic information and lesion enhancement are achieved on brain MR imaging with 0.1-mmol/kg gadobenate dimeglumine compared with gadobutrol at an equivalent dose.
ABBREVIATIONS:
- CNR
- contrast-to-noise ratio
- GBCA
- gadolinium-based contrast agent
- GRE
- gradient-recalled echo
- LBR
- lesion-to-background ratio
- NSF
- nephrogenic systemic fibrosis
- SE
- spin-echo
- SI
- signal intensity
Optimal detection and characterization of brain tumors on contrast-enhanced T1-weighted MR imaging depends as much on the contrast agent used as on the sequence parameters applied for image acquisition. Among the GBCAs currently approved by the FDA, gadobenate dimeglumine (MultiHance; Bracco, Milan, Italy) has proved superior to other GBCAs at equivalent dose for MR imaging of tumors of the CNS.1⇓⇓⇓⇓⇓⇓–8 The superior diagnostic performance achievable with gadobenate dimeglumine, which is reflected in a recently updated “Summary of Product Characteristics,”9 is due to high in vivo R1 relaxivity (6.3–7.9 L × mmol−1 × sec−1 at 1.5T,10,11) which derives from weak and transient interactions of the gadobenate contrast-effective molecule with serum albumin.12,13 The increased R1 relaxivity leads to increased SI enhancement and thus significantly improved lesion visualization and better depiction of morphologic features relative to those achieved with GBCAs, which do not interact with serum protein, when these agents are administered at an equivalent dose of 0.1 mmol/kg of body weight.1⇓⇓⇓⇓⇓⇓–8
Recently, gadobutrol (Gadavist [Gadovist]; Bayer Healthcare, Berlin, Germany) has been approved by the FDA for imaging of the CNS.14 This GBCA has a reported R1 relaxivity of 4.7–5.2 L × mmol−1 × sec−1 in human blood plasma at 1.5T10,11 and differs from gadobenate dimeglumine and other approved GBCAs in that it is formulated at twice the concentration (1 mol/L rather than 0.5 mol/L), meaning that twice the concentration of gadolinium is present per unit volume. Accordingly, gadobutrol should be administered at half the volume (0.1 mL/kg) to attain the approved dose of 0.1 mmol/kg of body weight.14 Studies in human subjects have suggested that gadobutrol may have advantages over other conventional GBCAs (ie, GBCAs that do not interact with serum albumin) for imaging of brain metastases.15⇓–17 However, to our knowledge, no studies have been performed to compare the more highly concentrated gadobutrol with the higher relaxivity agent gadobenate dimeglumine for MR imaging of brain tumors.
The aim of this study was to compare these 2 agents by using a rigorously controlled multicenter double-blind randomized intraindividual crossover study design in which each patient received 0.1-mmol/kg doses of both of these agents in 2 identical MR imaging examinations.
Materials and Methods
The study was Health Insurance Portability and Accountability Act–compliant, was conducted according to Good Clinical Practice standards, and was registered at www.clintrials.gov (ref. NCT00907530). All patients signed an approved informed consent form before enrollment.
Patients
One hundred twenty-three patients referred for contrast-enhanced MR imaging for known or suspected brain tumors were enrolled in a consecutive manner at 12 participating centers between September 2009 and November 2010. The highest number of patients enrolled at any center was 28. Six further centers enrolled between 8 and 16 patients. The remaining 5 centers enrolled between 2 and 5 patients. Patients were ineligible if they had received any investigational drug within 30 days before administration of either study agent. Subjects were also excluded if they were to receive any treatment that could affect lesion visualization before or between the 2 examinations (eg, whole-brain fractionated radiation therapy, steroids, or chemotherapy). Patients were also ineligible if they were pregnant or nursing or had impaired renal function, congestive heart failure, claustrophobia, gadolinium allergy, a cardiac pacemaker, or other contraindications to MR imaging.
One patient withdrew from the study after signing the informed consent form but before administration of either contrast agent. The remaining 122 eligible patients (67 men, 55 women; mean age, 56.1 ± 12.6 years; range, 20–84 years) were prospectively randomized to 2 study groups (A and B) to receive contrast agent according to 1 of 2 administration orders. Group A (n = 59) received gadobenate dimeglumine for the first examination and gadobutrol for the second; group B (n = 63) received the agents in opposite order.
MR Imaging
MR imaging was performed on 1.5T systems from several vendors (Avanto [n = 16], Sonata [n = 18], Symphony [n = 9], Siemens, Erlangen, Germany; Achieva [n = 34], Intera [n = 12], Gyroscan NT [n = 3], Philips Healthcare, Best, the Netherlands; Signa Excite [n = 14], HDx [n = 16], GE Healthcare, Chalfont St. Giles, United Kingdom) by using a standard head coil.
A rigorously controlled imaging protocol comprising T1 SE and T2 fast SE acquisitions before contrast injection and T1 SE and 3D high-resolution T1 GRE acquisitions after injection ensured protocol uniformity across sites and within individual patients. Sequence parameters varied between centers within predefined ranges because of the different imaging systems in use at these centers. However, the same MR imaging scanner, imaging planes, section prescriptions, and sequence parameters were used for both examinations in each patient. The range of parameters for the T1 SE sequence was as follows: TR = 350–680 ms, TE = 7.7–17 ms, excitations = 1–2, section thickness = 3–5 mm, FOV = 16 × 22–26 × 26 cm. The parameters for the T1 GRE sequence ranged as follows: TR = 7–2050 ms, TE = 2.99–6.28 ms, flip angle = 8°–60°, excitations = 1, section thickness = 1–3 mm, FOV = 23 × 17–30 × 23 cm. Parallel imaging was not used for any patient at any of the investigating centers.
Contrast agent administration was performed intravenously in an identical manner in both examinations by using either manual bolus injection (n = 118) or a power injector (n = 4). Both agents were administered at 0.1 mmol/kg of body weight, corresponding to 0.2 mL/kg for gadobenate dimeglumine and 0.1 mL/kg for gadobutrol. To maintain the study blind, an independent drug dispensing person administered each agent in the order determined by a randomization list. Care was taken to ensure that the injection duration was comparable for the 2 injections in each patient. All injections were followed by a saline flush of up to 30 mL.
Acquisition of postcontrast images began at a fixed time point, which was mandated to occur between 3 and 10 minutes after injection but could vary within this range depending on the site-specific protocol. However, the timing and sequence order of postcontrast acquisitions were identical for both examinations in each patient. The interval between the 2 MR imaging examinations was >48 hours in all patients to avoid carryover effects but <14 days to minimize the chance of measurable disease progression or lesion evolution.
Image Evaluation
All images were evaluated by 3 independent experienced radiologists (G.S., S.B., J.R.) who were unaffiliated with the study centers and blinded to the contrast agent used, to all patient clinical and radiologic information, and to all interpretations by on-site investigators. Each reader evaluated the patient images separately and independently.
All images from each patient were evaluated in a global matched-pairs fashion on a multimonitor imaging workstation. For each randomized patient number, all images from the first examination (examination 1) were displayed simultaneously with the corresponding images from the second examination (examination 2). Each reader was able to perform all routine interactive image-manipulation functions (eg, window/level, zoom, pan) on both image sets. If the postinjection images from either examination were considered technically inadequate by any of the 3 readers (eg, if artifacts compromised interpretability), no further assessment was performed for that patient by that reader. Once the readers' assessments were recorded and signed on an electronic case report form, the data base for that reading was automatically locked.
Qualitative Assessment of Diagnostic Information
Technically adequate images were evaluated qualitatively for diagnostic information and scored in terms of the following: 1) lesion border delineation, 2) disease extent, 3) visualization of lesion internal morphology, and 4) lesion contrast enhancement compared with surrounding normal tissue. All assessments were performed by using 3-point scales from −1 (examination 1 superior) through 0 (examinations equal) to +1 (examination 2 superior). For the various end points, superiority for 1 examination was recorded if it allowed better separation of ≥1 lesion from surrounding tissue, structures, or edema; better definition of lesion extent; clearer depiction of intralesion features; better difference in SI between lesions and surrounding normal tissue; or depiction of ≥1 lesion only after that examination.
The readers also indicated whether they had an overall diagnostic preference for 1 examination over the other. When diagnostic preference was expressed, the reader then selected ≥1 of the following reasons for this preference: superior contrast enhancement, better delineation of normal structures, better delineation of at least 1 lesion, better visualization of lesion internal structure, more lesions identified, or greater diagnostic confidence.
Quantitative Assessment
Quantitative evaluation was performed by each reader independently by using a simultaneous matched-pairs approach. SI measurements were made at ROIs positioned on areas of normal brain parenchyma and on up to 3 enhancing lesions per patient identified on postcontrast images from both examinations. Additional SI measurements were made at ROIs placed in selected areas external to the brain to determine the background noise. Each ROI placed on the selected postinjection image from 1 examination appeared simultaneously at identical coordinates on the corresponding image from the other examination. Care was taken to avoid inclusion of vessels, and to ensure that ROIs of equal size were positioned on all corresponding image sets. If multiple lesions were present in a given patient, ROIs were placed on up to 3 of the largest most conspicuous lesions. A multimonitor imaging workstation (AquariusNET server, Version 4.4.1.4; TeraRecon, San Mateo, California) was used to determine SI values on a pixel-by-pixel basis and to calculate the percentage of enhancement of lesions for T1 SE acquisitions and the CNR and LBR for both T1 SE and T1 GRE acquisitions. CNR and LBR were determined by using the equations below, in which SI represents signal intensity, “brain” represents surrounding normal brain parenchyma measured on the same image section, and SD is the standard deviation of the background SI measured in an area of the same image section out of the body and devoid of artifacts.

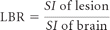
Safety Assessments
Monitoring for adverse events was performed from the moment the patient signed the informed consent form until 24 hours after administration of the first study agent and then from the moment the second study agent was administered until 24 hours after administration of the second agent. Events were classified by the principal investigator at each center as either serious (ie, death, life-threatening, requiring or prolonging hospitalization) or not serious. Any perceived relationship to the agent was recorded.
Statistical Analysis
Power determination was based on the primary efficacy assumption that a 0.1-mmol/kg dose of gadobenate dimeglumine is superior to an equivalent dose of gadobutrol according to reader preference. Based on the results of a recent study5 and assuming an “equal” response in 50% of cases, a ratio of superiority for either agent of 2.8:1, and an effect size of 0.111, enrollment of approximately 118 subjects was needed to demonstrate superiority with 85% of power considering a dropout rate of 15% (nQuery Advisor, Version 6.01; Statistical Solutions, Cork, Ireland).
Analysis of blinded reader evaluations was performed by using the statistical software package SAS, Version 8.2 (SAS Institute, Cary, North Carolina). The distribution of reader preferences for the diagnostic information end points was tested by using the Wilcoxon signed rank test. Inter-reader agreement for diagnostic preference was presented as a percentage agreement and was assessed by using generalized κ statistics. Agreement was classified as excellent (κ values > 0.8), good (κ = 0.61–0.8), moderate (κ = 0.41–0.6), fair (κ = 0.21–0.4), or poor (κ ≤ 0.2).18
Differences between gadobenate dimeglumine and gadobutrol in terms of quantitative enhancement were analyzed by using a mixed-effects model. The change from predose was the response variable, and factors included in the model were patient, period, sequence, study agent, and predose score, where patient nested within sequence was the random effect.
For the comparison of qualitative parameters, a Bonferroni adjusted significance level of P < .01 was used to take into account the multiple qualitative end points. All other statistical tests were conducted at a significance level of P < .05.
Results
Patients
Of the 122 patients randomized and evaluated for safety, 8 (6.5%; 4 after gadobenate dimeglumine; 4 after gadobutrol) were withdrawn prematurely after the first examination. The reasons for discontinuation were initiation of treatment (surgery or steroid therapy, n = 2), deterioration of patient condition (n = 2), or nonenhancing or no lesions seen on the first examination (n = 4). A total of 114 patients (55 in group A, mean age, 57.6 ± 11.9 years; 59 in group B, mean age, 55.0 ± 12.5 years) were, therefore, evaluated for diagnostic efficacy. There were no significant between-group differences in sex (P = .174), age (P = .257), age group (18–64 years, ≥65 years; P = .057), weight (P = .595), height (P = .352), or race (P = .367) distribution.
The diagnoses of these 114 patients were primary glial tumor in 49 (43%) cases (glioma [n = 9], glioblastoma multiforme [n = 17], astrocytoma [n = 11], oligodendroglioma [n = 6], mixed oligoastrocytoma [n = 3], ependymoma [n = 1], ganglioglioma [n = 1], gliomatosis cerebri [n = 1]); secondary metastases in 46 (40%) cases (primary lung cancer [n = 20], breast cancer [n = 8], renal carcinoma [n = 3], melanoma [n = 3], prostate cancer [n = 3], colon cancer [n = 1], esophageal cancer [n = 1], ovarian cancer [n = 1], testicular cancer [n = 1], carcinoma of the maxillary sinus [n = 1], unknown cancer [n = 4]); extra-axial tumors in 8 (7%) cases (meningioma [n = 7], acoustic neuroma/schwannoma [n = 1]); other tumor diagnosis in 7 (6%) cases (brain infiltration by lymphoma [n = 4], leukemia [n = 1], craniopharyngioma [n = 1], glomus tumor [n = 1]); or “other diagnosis” in 4 (4%) cases (postoperative scar/fibrosis [n = 1], leukoaraiosis [n = 1], radiation necrosis [n = 1]; and white matter microangiopathic ischemic disease [n = 1]).
Qualitative Image Assessment
Readers 2 and 3 considered all image sets from each of the 114 evaluable patients to be technically adequate, while reader 1 considered the postdose T1 SE images of 1 patient to be technically inadequate. Qualitative assessments were, therefore, performed for 114 patients by readers 2 and 3 but for 113 patients by reader 1.
The findings of the 3 readers for global diagnostic preference and each of the individual end points are shown in Table 1. Readers 1, 2, and 3 reported global preference for gadobenate dimeglumine in 46 (40.7%), 54 (47.4%), and 49 (43.0%) patients, respectively, compared with 6 (5.3%), 7 (6.1%), and 7 (6.1%) patients for gadobutrol (P < .0001, all readers). Similar highly significant preference (P < .0001; all evaluations, all readers) was demonstrated for each individual diagnostic information end point. κ values for 3-reader agreement ranged from κ = 0.414 for definition of disease extent to κ = 0.629 for visualization of lesion internal morphology (Table 1). All 3 readers agreed completely for 61.9%–73.5% of the patients, depending on the diagnostic information end point under consideration. Agreement between 2 of the 3 readers was obtained for ≥97.3% for all end points.
Qualitative assessment of patients with brain tumorsa
Further evaluation of patients for whom a preference was expressed revealed that in most cases, this was due to superior contrast enhancement and better delineation of lesions and/or lesion internal structures (Table 2). Examples of the improved imaging performance achieved with gadobenate dimeglumine are shown in Figs 1–3.
Reasons for global diagnostic preference as expressed by blinded readers
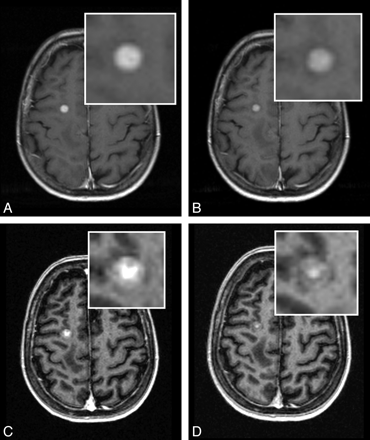
A 58-year-old woman with metastasis from melanoma undergoing MR imaging for definite staging of metastatic disease. T1 SE (A and B) and T1 GRE (C and D) images reveal a metastasis in the right superior frontal gyrus. However, the lesion appears larger and shows more conspicuous enhancement with gadobenate dimeglumine (A and C) than with gadobutrol (B and D).
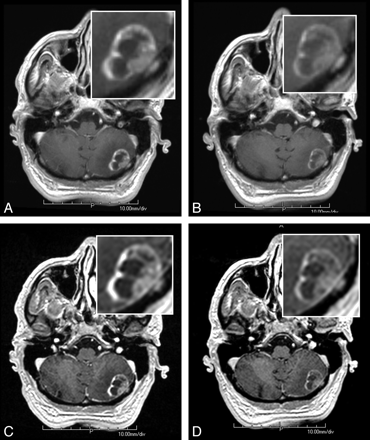
A 70-year-old man with metastasis from lung carcinoma undergoing MR imaging for identification and location of metastatic disease. T1 SE (A and B) and T1 GRE (C and D) images reveal a large metastasis in the posterior aspect of the left cerebellar hemisphere. The lesion appears larger, shows more conspicuous enhancement, and is better demarcated with gadobenate dimeglumine (A and C) than with gadobutrol (B and D).
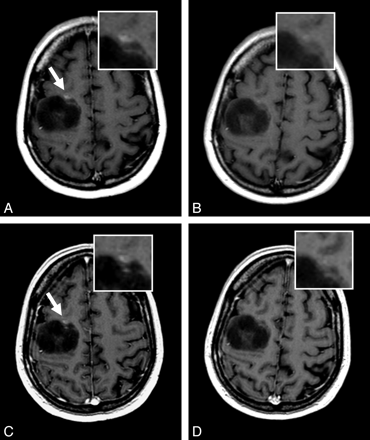
A 49-year-old woman with juxta-/prerolandic right frontal glioma undergoing MR imaging for assessment of tumor evolution. T1 SE (A and B) and T1 GRE (C and D) images reveal a large inhomogeneous tumor. The presence of a small enhancing focus (arrow) in the ventral portion of the tumor is seen clearly only on gadobenate dimeglumine–enhanced images (A and C). The enhancing area is nearly undetectable on gadobutrol-enhanced images (B and D).
Quantitative Evaluation
Readers 1, 2, and 3 recorded lesion SI measurements relative to normal brain parenchyma and background noise for 116, 103, and 92 lesions, respectively, on T1 SE images, and for 116, 102, and 92 lesions, respectively, on T1 GRE images. The mean percentage of enhancement of lesions on T1 SE images was highly significantly greater with gadobenate dimeglumine compared with gadobutrol for all 3 readers (reader 1, 119.9 ± 68.7 versus 97.3 ± 58.2; reader 2, 121.6 ± 65.9 versus 99.8 ± 56.9; reader 3, 111.9 ± 62.3 versus 89.7 ± 55.9; P < .0001 for all 3 readers) with no obvious differences across different scanner manufacturers.
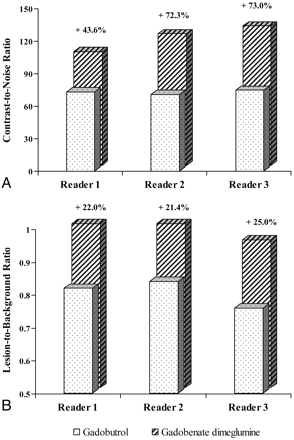
Determinations of CNR and LBR based on SI measurements on T1 SE images relative to precontrast images are shown in Fig 4. Highly significant increases in quantitative enhancement for gadobenate dimeglumine was noted by all 3 readers for both CNR (P = .0186, P < .0001, P = .0007; readers 1, 2, and 3, respectively) and LBR (P < .0001, all 3 readers).
Blinded reader comparison of CNR (A) and LBR (B) after 0.1-mmol/kg doses of gadobenate dimeglumine and gadobutrol. Highly significant increases in quantitative enhancement for gadobenate dimeglumine are noted by all 3 readers for both CNR (P < .02) and LBR (P < .0001).
On postdose T1 GRE images readers 1, 2, and 3 determined CNR values of 67.49, 73.58, and 79.59, respectively, for gadobenate dimeglumine and 31.23, 33.16, and 35.47, respectively, for gadobutrol. The greater CNR achieved with gadobenate dimeglumine (+116.11%, +121.89%, and +124.39%, respectively) was highly significant (P < .0001) for each reader. Similar highly significant (P < .0001) differences between gadobenate dimeglumine and gadobutrol were noted by each reader for postdose comparisons of LBR.
Safety
No clinically meaningful differences were noted in terms of the incidence of adverse events: Eight of 118 (6.8%) patients reported 12 nonserious events after gadobenate dimeglumine compared with 7/118 (5.9%) patients who reported 10 nonserious events after gadobutrol. Minor gastrointestinal symptoms and injection site conditions were the most frequent events reported with both agents. All contrast-related adverse events were mild and self-resolving, apart from 1 case of injection site inflammation after gadobutrol, which was considered moderate in intensity.
Discussion
Gadobutrol differs from other approved GBCAs in that it is formulated at a 2-fold higher concentration.14 It has a macrocyclic structure similar to that of gadoteridol (ProHance, Bracco, Princeton, New Jersey)19,20 and physicochemical properties that resemble those of other GBCAs that have conventional in vivo R1 relaxivity.10,11,18.19 Consistent with the results of previous intraindividual comparisons,1⇓⇓⇓⇓⇓⇓–8 the results of this study show that when administered at its approved dose of 0.1 mmol/kg, gadobutrol is preferred in significantly (P < .0001) fewer patients than gadobenate dimeglumine at the same dose. Specifically, 3 blinded independent readers reported superiority for gadobenate dimeglumine in significantly (P < .0001) more patients for all evaluated end points. Most important, the opinions of the 3 readers were identical for 61.9%–73.5% of the patients, resulting in κ values of κ = 0.414–0.629 for inter-reader agreement. κ values of this magnitude are very good for qualitative evaluations of this type.
Concerning quantitative lesion enhancement, our results are again consistent with those in previous studies1⇓⇓⇓⇓⇓⇓–8 in showing a significantly greater percentage of enhancement of lesions and significantly superior CNR and LBR with gadobenate dimeglumine. Although the magnitude of the difference in CNR was slightly lower for reader 1 (43.6% greater CNR with gadobenate dimeglumine compared with 72.3% and 73.0% greater CNR with gadobenate dimeglumine for readers 2 and 3, respectively), the differences were in all cases significant. As noted in comparisons with other GBCAs,5⇓⇓–8 the increase in CNR with gadobenate dimeglumine compared with gadobutrol can be considered comparable with the magnitude typically observed with a double dose of GBCA compared with a single dose.21,22 Although comparable enhancement to that achieved with gadobenate dimeglumine might have been obtained with a double dose of gadobutrol (ie, an identical volume), injections of high doses of GBCAs are not to be recommended, given the risk of NSF in certain patients with severe renal impairment. In this regard, although gadobutrol is a macrocyclic agent and thus widely considered one of the “safer” GBCAs, cases of NSF following its sole injection have been reported.23,24
Our study suggests that gadobutrol, though reported to have slightly higher relaxivity compared with established standard relaxivity agents,11,25 provides little or no benefit over other GBCAs for brain tumor imaging when compared specifically with the imaging performance achieved with 0.1 mmol/kg of gadobenate dimeglumine. In support of this conclusion, the package insert for gadobutrol describes an as yet unpublished comparison of gadobutrol and gadoteridol and states that “performances of Gadavist and gadoteridol for visualization parameters were similar.”14 Although a recent study by Katakami et al17 suggests that a single dose of gadobutrol is noninferior to a double dose of gadoteridol for detection of brain metastases, their study was inherently flawed in that no comparison was performed between a single dose of gadobutrol and a single dose of gadoteridol. Thus it is not possible to say whether a single dose of gadoteridol would have been equally noninferior to a single dose of gadobutrol by using their study design and statistical methodology.
Two conclusions can be drawn from our findings. First, it is clear that contrast agent concentration in the vial has no effect on imaging performance because injection of the more highly concentrated (1.0 mol/L) GBCA at 0.1 mmol/kg of body weight provides no appreciable clinical advantage relative to published findings for conventional GBCAs at a standard concentration (0.5 mol/L) when compared with gadobenate dimeglumine at an identical dose of 0.1 mmol/kg of body weight.1⇓⇓⇓⇓⇓⇓–8 The lack of any appreciable benefit with gadobutrol can be ascribed to the fact that the SI during the interstitial phase (ie, at postinjection acquisition times of 3–10 minutes as typically performed for brain tumor imaging) depends solely on the total amount of gadolinium in the lesion (ie, the total number of gadolinium molecules) and its relaxivity rather than on the gadolinium solution concentration. Second, it is clear that higher R1 relaxivity is instrumental in improving diagnostic performance relative to that achievable with conventional GBCAs at equivalent dose. In support of both conclusions are the findings of a recent study by Achenbach et al,26 in which gadobutrol and gadobenate dimeglumine were compared at equivalent total volume (0.1 mL/kg of body weight, corresponding to a full dose of gadobutrol [0.1 mmol/kg of body weight] but only a half dose of gadobenate dimeglumine [0.05 mmol/kg of body weight]) for contrast-enhanced MRA of the peripheral arteries. In their study no differences were found in terms of quantitative enhancement, image quality, or diagnostic accuracy, indicating that the higher T1 shortening by gadobutrol per unit of time due to the double concentration was of no benefit compared with the greater relaxivity of gadobenate dimeglumine at only half the dose.26
To appreciate the role that relaxivity plays in improving diagnostic performance, one must understand the molecular properties of the various GBCAs and the influence each agent has on shortening the T1 relaxation time during image acquisition. Gadobenate dimeglumine differs from the widely used conventional GBCA gadopentetate dimeglumine (Magnevist; Bayer Healthcare, Berlin, Germany) through the presence of a hydrophobic benzyloxymethyl substituent on the gadobenate molecule.20 This substituent confers 2 unique properties: First, it results in the gadobenate contrast-effective molecule undergoing elimination from the body in part through the hepatobiliary route, thereby rendering this agent appropriate for hepatobiliary applications.27,28 Second and more pertinent for extrahepatic applications, this substituent causes the gadobenate molecule to interact weakly and transiently with serum albumin.12,29 These interactions result in a slowing of the tumbling rate of the gadobenate molecule, leading to a longer rotational correlation time with inner shell water protons and hence an increase in the T1 relaxation rate.29,30 Thus, while the R1 relaxivity values of gadobenate dimeglumine and gadopentetate dimeglumine are roughly similar in water,11,29 the relaxivity of gadobenate dimeglumine is considerably higher when evaluated in vivo or in solutions containing plasma proteins.10,11,29 Moreover, the R1 relaxivity of gadobenate dimeglumine is consistently higher across all magnetic field strengths,10,11 resulting in improved imaging performance even at higher (3T) field strengths.8,31 In contrast, the molecular structure of gadobutrol differs from that of gadoteridol only in that a hydroxypropyl group on the gadoteridol molecule is replaced by a trihydroxybutyl group on the gadobutrol molecule.20 Unlike gadobenate dimeglumine, gadobutrol does not interact to any appreciable extent with serum proteins and is excreted almost exclusively through the kidneys.14,32,33 As a consequence, its R1 relaxivity in vivo is due entirely to the size and innate T1 shortening capacity of the gadobutrol molecule itself rather than to any augmentation of relaxivity and T1 shortening through interaction with serum albumin. Although reported relaxivity values vary slightly across publications depending on experimental conditions,10,11,13 the results of well-conducted intraindividual comparative studies confirm that differences in relaxivity in vivo lead clinically to significantly better outcomes.1,5⇓⇓–8,34⇓–36
A final consideration concerns the fact that this study was performed at 1.5T only. Although significant superiority for gadobenate dimeglumine compared with gadopentetate dimeglumine for brain tumor imaging has previously been demonstrated at 3T,8 to our knowledge no studies have yet compared gadobenate dimeglumine and gadobutrol at higher field strengths. Nevertheless, given that the reported R1 relaxivity of gadobenate dimeglumine at 3T (5.5–5.9 L × mmol−1 × sec−1) is again higher than that of gadobutrol (4.5–5.0 L × mmol−1 × sec−1)10,11 and given that the same issues apply with regard to SI dependence on total gadolinium dose (number of gadolinium molecules) rather than gadolinium concentration, it is to be expected that superior imaging performance would again be achieved with gadobenate dimeglumine relative to gadobutrol at this higher field strength.
Conclusions
Our results confirm expectations based on theoretic considerations of GBCA molecular structures and properties.20,37 Thus, 3 blinded readers found no relevant benefit for the more highly concentrated gadobutrol in terms of lesion conspicuity, visualization, or sensitivity for detection. Rather, our intraindividual comparison of gadobenate dimeglumine and gadobutrol at an identical dose of 0.1 mmol/kg of body weight confirms that significantly improved qualitative and quantitative enhancement of brain tumors is achieved with gadobenate dimeglumine without any difference in safety.
Acknowledgments
The authors would like to thank Dr. MUDR. Manuela Vaněčková, PhD, Stanislav Burval, MD, Sean Symons, MD, Susan Crisp, Jennifer Giglia and all other persons involved in this study for their invaluable contributions toward patient enrollment and the acquisition of data.
Footnotes
This work was supported by Bracco, who provided support to institutions for fees related to submission of study protocol to the Ethics Committee as well as a fee per patient enrolled.
Disclosures: Josef Vymazal—UNRELATED: Consultancy: NovoCure (Israel); Grants/Grants Pending: Ministry of Health (Czech Republic),* Comments: research grants, Payment for Development of Educational Presentations: Bayer (Germany), Travel/Accommodations/Meeting Expenses Unrelated to Activities Listed: Siemens,* Nycomed (Czech Republic).* Mayank Goyal—UNRELATED: Payment for Lectures (including service on Speakers Bureaus): Penumbra, Comments: for speaking engagements. Stock/Stock Options: Calgary Scientific. Cesare Colosimo—RELATED: Consulting Fee or Honorarium: Catholic University School of Medicine,* UNRELATED: Consultancy: Bracco, Bayer, Comments: Scientific consultant, Expert Testimony: Bracco, Payment for Lectures (including service on Speakers Bureaus): Bracco, Bayer. Brian Yemen—RELATED: Grant: Hamilton Health Sciences, Juravinski Cancer Center clinical trials,* Comments: All discussions related to finances of this clinical trial were handled by the clinical trials department. I have neither seen a copy nor am I aware of the specific contents of that contract, Consulting Fee or Honorarium: Hamilton Health Sciences, Juravinski Cancer Center clinical trials,* Comments: Previous contracts have included compensation for interpretation of the clinical trials component of the examination. I am not aware that this occurred with this study but have answered yes in case I am in error. Nicoletta Anzalone—UNRELATED: Board Membership: Bayer, Comments: advisory boards sponsored by Bayer, Consultancy: Bayer, Expert Testimony: Bayer, Grants/Grants Pending: Bayer, Payment for Lectures (including service on Speakers Bureaus): Bayer. Guenther Schneider—RELATED: Fees for Participation in Review Activities such as Data Monitoring Boards, Statistical Analysis, Endpoint Committees, and the Like: Bracco, Comments: off-site reading of the study; Payment for Lectures (including service on Speakers Bureaus): Siemens, Bracco, Guerbet, Bayer-Schering. Stefano Bastianello—RELATED: Consulting Fee or Honorarium: Bracco, Comments: The fee was for my participation as a blinded reader on the study as compensation for time off work. Jordi Ruscalleda—RELATED: Consulting Fee or Honorarium: Bracco, Comments: fee for blinded assessment. *Money paid to the institution.
References
- Received July 1, 2011.
- Accepted after revision September 5, 2011.
- © 2012 by American Journal of Neuroradiology