Abstract
BACKGROUND AND PURPOSE: Currently, no specific method exists to measure arteriovenous malformation (AVM) nidus size, a requirement in assessing the success of treatment. Additionally, the commonly used evaluation provides only a linear one-dimensional measurement of this three-dimensional entity. The purpose of this study was to devise an improved method for measuring AVM nidus size, an irregularly shaped radiologic entity, that provides objective and reproducible results.
METHODS: The procedure involved digitizing angiograms obtained before and after treatment, making the gray scale uniform, printing images on standard bond paper, delineating the nidus area, measuring the nidus area with a polar planimeter, and finally, correcting for geometric magnification. Three observers made the measurements. The corrected nidus areas were tabulated, and the mean, standard deviation, interobserver variability, and confidence intervals (CIs) were calculated.
RESULTS: On both anteroposterior and lateral views, the Kendall coefficient of concordance (a measure of interobserver variability) was equal to 0.97, signifying excellent agreement. Additionally, these values were within the 95% CIs; this result showed that they were unlikely the result of chance.
CONCLUSION: Precise measurements of an AVM nidus are required to properly analyze changes in the lesion after endovascular embolization (ie, to evaluate treatment success). Because of the irregular contours of an AVM nidus, measuring an area with planimetry, rather than with the usual linear dimensions, should yield more exact results.
Neuroradiologists often encounter cerebral arteriovenous malformations (AVMs), a common vascular pathologic condition. Referring clinicians, both neurosurgeons and neurologists, depend on radiologic imaging, particularly cerebral angiography, to fully evaluate these lesions. Accurate information regarding nidus size and location, arterial feeding pedicles, and venous drainage patterns have a substantial effect on the lesion’s natural history, potential treatment paradigms, and prognostic implications. Additionally, an objective, reproducible method to analyze AVM nidus size is required to fully assess patient improvement or change after surgical resection, endovascular embolization, or radiation therapy ablation.
The Spetzler-Martin grading scale (1) is the generally accepted method for analyzing an AVM because it provides important surgical prognostic information based on nidus size (<3 cm, 3–6 cm, or >6 cm), location (eloquent or non-eloquent), and venous drainage (superficial or deep). However, the nidus size measurement is a linear dimension and provides only an estimate of the actual nidus volume because the nidus is rarely a sphere. Therefore, although this classification is extremely helpful in the presurgical patient examination, it does not provide the precision needed to evaluate therapeutic success. We devised a method to achieve this result and tested the reproducibility of the measurements.
Cerebral angiography remains the criterion standard for AVM evaluation. Use of vascular contrast material injection provides further detailed information regarding feeding arterial pedicles, nidus location, and venous drainage, as well as associated vascular pathologic conditions such as arterial aneurysms, venous varices, and venous stenoses. Therefore, angiography is the radiologic technique of choice in the assessment of nidus size.
Methods
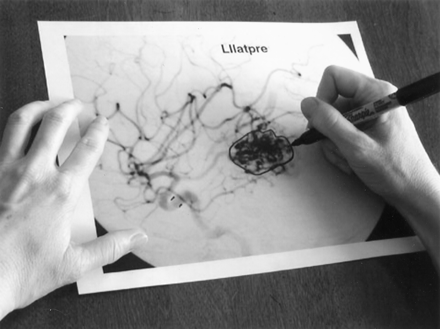
In this study, 11 consecutive patients with brain AVMs of varying size and location were examined. Cerebral angiograms were obtained in both anteroposterior and lateral projections. The 22 images that showed maximal nidus size were digitized, and the background gray scale was made uniform by means of computer manipulation (Photoshop; Adobe) before the nidus was measured. Multiple copies of the images were then printed on standard paper. Three experienced neuroradiologists independently outlined the AVM nidus (Fig 1).
Lateral angiogram with an outline of the AVM nidus to be subsequently measured.
A mechanical polar planimeter (Model L-20; Lasico Corp, Los Angeles, CA) was calibrated by using a planar object of known dimensions. The nidus area was then measured with the planimeter (Fig 2). The polar planimeter is a device that scientific and engineering personnel use to measure planar areas, for example, the area under a curve in a spectrum such as that produced at gas chromatography or MR spectroscopy. The outline of the object to be measured is traced with the lever arm of the planimeter. The measurement is then registered in the device. Because the planimeter produces only a numerical dial reading (Fig 3), this reading must be multiplied by a correction factor particular to each planimeter model; the product represents the area in centimeters squared. This measured area value can then be corrected for the geometric magnification of the radiograph by using the parent vessel (such as the basilar artery or carotid artery) as an internal control.
Mechanical polar planimeter tracing the outline of the AVM nidus.
Numerical dial reading produced by the planimeter.
The average diameters of the internal carotid and basilar arteries, as well as the statistical deviation resulting from normal anatomic variation, have been previously described (2–4). If the AVM is supplied by the anterior circulation, the intracavernous internal carotid artery is considered to be a 4-mm-diameter structure. If the AVM is supplied by the posterior circulation, the basilar artery is considered to be a 3-mm-diameter structure. These standards are then compared with the actual diameter measurement of the corresponding vessel on the radiograph. With these correction factors, the actual dimensions of the AVM nidus area can be calculated for any magnification used during patient imaging. This is achieved with ratios as shown in Equation 1, as follows: 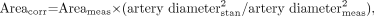 1) where Areacorr is the corrected nidus area, Areameas is the measured nidus area, Artery diameterstan is the standard artery diameter (3 or 4 mm), and Artery diametermeas is the measured artery diameter.
1) where Areacorr is the corrected nidus area, Areameas is the measured nidus area, Artery diameterstan is the standard artery diameter (3 or 4 mm), and Artery diametermeas is the measured artery diameter.
To determine the statistical significance of the differences between these measured values and to evaluate the reliability and reproducibility of the described method, the interobserver variability was determined. The Kendall coefficient of concordance, which is used in a nonparametric method of assessing agreement between two or more observers, was calculated. To account for the role of chance in estimating the Kendall coefficient of concordance, test-based, two-sided 95% confidence intervals (CIs) were also calculated. The coefficient of concordance was determined for each of the two planes (5). Equation 2 illustrates the method used to calculate the coefficients, as follows:  2) where W′ is the sample estimate of the Kendall coefficient of concordance; Rj is the sum of the ranks for all the observers for the jth observation; n is the number of groups; k is the number of variables (both dependant and independent); and ΣT is the correction for ties, where ti is the number of tied ranks in the ith group of ties, and ΣT = Σ(t3 − ti),
2) where W′ is the sample estimate of the Kendall coefficient of concordance; Rj is the sum of the ranks for all the observers for the jth observation; n is the number of groups; k is the number of variables (both dependant and independent); and ΣT is the correction for ties, where ti is the number of tied ranks in the ith group of ties, and ΣT = Σ(t3 − ti),
The test-based, two-sided 95% CIs were defined (6) as shown in Equation 3: 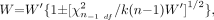 3) where W is the interval estimates of the Kendall coefficient of concordance, and χ2n−1 df is χ2 with (n − 1) degrees of freedom.
3) where W is the interval estimates of the Kendall coefficient of concordance, and χ2n−1 df is χ2 with (n − 1) degrees of freedom.
Results
The corrected areas in both the anteroposterior and lateral planes for each of the 11 patients were calculated and tabulated. Table 1 shows the corrected areas measured by each observer, as well as the arithmetic mean and standard deviation for each patient on images in both planes. The values measured were similar among the three observers, as shown by the relatively low calculated standard deviations. Occasional disparate values were measured when the nidus had poorly defined borders, in particular, in one patient who had a relatively large ill-defined nidus. The Kendall coefficient of concordance and the test-based, two-sided 95% CIs were then calculated. The resultant point and interval estimates of the Kendall coefficient of concordance are shown in Table 2. A coefficient of unity (ie, 1.0) signifies perfect agreement, and a coefficient greater than 0.75 represents excellent agreement (7).
Corrected area values measured by each observer*
Interobserver agreement in the measurement of nidus size
Discussion
We devised a new method of AVM nidus measurement to better approximate the true nidus size by measuring the actual nidus area in two projections (ie, anteroposterior and lateral). The measurements were easily performed and reproducible. Occasionally, the readers disagreed in individual measurements when the nidus border was not apparent; however, these differences were not statistically significant. When the nidus was clearly imaged, the individual areas were similar. The only potential source of error was the use of a standard arterial diameter measurement to correct for geometric magnification. Although this may have slightly altered the true individual lesion size, the potential error was eliminated when pre- and posttreatment images were compared because the measurements in the same artery were used in each examination (with the assumption that the parent vessel did not change in size and that a substantial change in the diameter immediately after embolization was unlikely).
Regarding the reliability and reproducibility of this method in measuring nidus size, the data were statistically evaluated to determine the interobserver variability. For both planes, the Kendall coefficient of concordance was 0.97. Therefore, despite the small sample size, the statistical analysis revealed excellent agreement among readers in the evaluation of AVM nidus size. Additionally, the Kendall coefficients of concordance in both the anteroposterior and lateral planes were within the calculated 95% CIs. Thus, it is unlikely that the statistical conclusions were the result of chance.
Neuroradiologists, in particular, interventional neuroradiologists, are commonly required to evaluate and treat cerebral AVMs. Cerebral angiography is necessary to define nidus size and location, arterial feeding pedicles, and venous drainage patterns. Although detecting the arterial feeders and draining veins is usually easy, precise delineation of the vascular nidus can be difficult because of the inherent three-dimensional shape of the lesion, which is further complicated by the commonly irregular borders of the nidus. Given this irregular shape, the common practice of using a linear measurement for the nidus size is not accurate. Additionally, when therapeutic success is compared by using pre- and posttreatment angiograms, this linear measurement error is compounded (ie, nidus volume reduction is a linear measurement to the third power).
The accuracy of this method in the isolated individual values was not proved in this study because this confirmation requires comparison to a criterion standard, a standard that has not yet been established for the planar measurement of an AVM nidus. Yet, the utility of this method is in the comparison of pre- and post-treatment images to better analyze therapeutic success. Ultimately, the best evaluation of AVM nidus size is a volumetric measurement. However, presently, this is an extremely difficult task. Even evaluation with MR or CT imaging does not provide true volumetric assessment. A three-dimensional reconstruction of the nidus, such as the production of a model from stereolithography, is required for these lesions with extremely irregular borders. The actual volume could then be determined by using a volumetric displacement method such as immersion in a fluid. Future projects will be aimed toward this goal. Nonetheless, although the use of planimetry (a two-dimensional technique) does not enable true volumetric assessment, it is a notable improvement compared with the current, common practice of using linear (one-dimensional) measurements in the evaluation of AVM nidus size.
Because multiple modalities of treatment for AVMs with many new therapeutic agents are currently on the horizon, particularly in the neurointerventionalist’s realm, this objective, reproducible, and reliable method for measuring AVM nidus size should be a welcome addition to the ever-expanding armamentarium needed to evaluate treatment success of such complicated and detrimental lesions.
Conclusion
Cerebral angiography remains the technique of choice in the evaluation of intracranial AVMs. More precise measurement of the AVM nidus with planimetry allows objective, reproducible assessment of the lesion. This easily performed method should improve appraisal of the success of treatment for this difficult disease entity.
Acknowledgments
The authors gratefully acknowledge Dr Robert Hirsch for his invaluable assistance with the statistical analysis of the data.
References
- Received June 6, 2001.
- Accepted after revision October 12, 2001.
- Copyright © American Society of Neuroradiology