Abstract
BACKGROUND AND PURPOSE: Our aim was to explore the changes of CSF and spinal pathomorphology after high-concentration ozone injection into a pig's subarachnoid space.
MATERIALS AND METHODS: Eight experimental pigs were randomly divided into 4 groups (n=2 in each group). In groups A, B, C, and D, animals were sacrificed for pathology 1 day, 1 week, 1 month, and 3 months after the injection, respectively. Ozone (10 mL, 90 μg/mL) was injected along the lumbar 4–5 interspace into the subarachnoid space with a 21-ga Chiba needle under fluoroscopy. CSF collection and CT and MR imaging were performed before injection and 30 minutes, 1 week, 1 month, and 3 months after injection, respectively. The spinal cords were observed macroscopically and microscopically.
RESULTS: No serious behavior abnormalities were observed in any animals. There were no abnormalities in CSF and spinal cords pathologically and radiologically after injection.
CONCLUSION: Our initial study suggests that high-concentration ozone injection into a pig's subarachnoid space will not cause serious sequelae such as chemical arachnoiditis and necrosis of the spinal cord, and this finding provides evidence of the safety of ozone inadvertently injected into subarachnoid space in the clinical treatment of disk herniation.
In recent years, ozone injection for the treatment of lumbar or cervical disk herniation has been reported by several authors with good results.1–2 Sufficient ozone concentration and dose as well as full contact with the nucleus pulposus are crucial to obtain good results, and precise puncture is essential for full ozone contact with the nucleus pulposus. The commonly used trajectories include the posterolateral and paramidline approach under fluoroscopy and CT. Whichever trajectory is introduced, there is a probability of puncture of the subarachnoid space and entrance of ozone into subarachnoid space. To our knowledge, there is no report yet of the effect of ozone on the CSF and spinal cord when it is inadvertently injected into the subarachnoid space. In this study, we injected high-concentration ozone (10 mL, 90 μg/mL) into the subarachnoid space of 8 healthy pigs and observed its effects on the CSF and spinal cord. The purpose of our study was to provide evidence of the safety of ozone inadvertently injected into subarachnoid space in the clinical treatment of disk herniation.
Materials and Methods
Animals and Experimental Groups
Our institutional animal research committee gave protocol approval for this study. All policies on humane care and use of laboratory animals were followed. Eight experimental pigs (4 male, 4 female) from the Experimental Animal Center of Chinese PLA General Hospital (mean weight, 27.5 kg; range, 25–30 kg) were divided into 4 groups. In groups A, B, C, and D, animals were sacrificed for pathology 1 day, 1 week, 1 month, and 3 months after the injection, respectively.
Procedure
Animals were anesthetized with an intramuscular injection of 10 mg per kilogram of body weight of ketamine hydrochloride and 1-mg/kg diazepam hydrochloride. Under fluoroscopy, a 21-ga Chiba needle (Maiteli Medical Technology, Beijing, China) was punctured into the subarachnoid space along the lumbar 4–5 interspace, and 10 mL, 90 μg/mL of ozone from an ozone generator (CHY-11-β, Chinese YiDeKang Medical Technology, Shandong Province, China) was pumped and injected into it. CSF collection was made before injection and 30 minutes, 1 week, 1 month, and 3 months after injection, respectively.
Observation of Animal Behavior after the Procedure
After the procedure, the status of all animals, including eating, drinking, moving, and neurologic behavior, was observed and recorded.
Imaging Examination
CT was performed with a LightSpeed 16-spiral scanner (GE Healthcare, Milwaukee, Wis) at the same time as CSF collection. In all pigs, lumbar and thoracolumbar columns were scanned, and sagittal multiplanar reconstructions were used in a workstation (Advantage Workstation 4.2, GE Healthcare). Imaging parameters included a section thickness of 1.5 mm, kilovolt peak of 120 kVp, x-ray tube current of 300–330 mAs, pitch factor of 1.375, matrix of 512 × 512, and tube rotation time of 0.8 seconds. MR imaging was performed with a Signa Excite 1.5T imager (GE Healthcare) at the same time as CT. The coil was an 8-channel spinal coil. All MR imaging examinations included sagittal and axial T2-weighted fast-recovery fast spin-echo (FSE) imaging (TR/TE, 3000–4000/90–110 ms; echo-train length, 10–20; section thickness, 4.0 mm; matrix, 256 × 256; FOV, 30 × 30 cm) as well as contrast-enhanced T1-weighted spin-echo imaging (TR/TE, 400–500/10–15 ms; 90° flip angle; matrix, 256 × 256; section thickness, 4 mm; FOV, 30 × 30 cm) after rapid-bolus intravenous injection of gadopentetate dimeglumine (0.1 mmol per kilogram of body weight; Magnevist; Schering, Berlin, Germany).
CSF Assay and Pathologic Evaluation
The opening pressure of the CSF was observed. Routine assay (including color, transparency, and qualitative analysis) and biochemical assay (including protein, glucose, chloride, white blood cell count, and total cell count) were analyzed. After sacrificing the pigs, we removed the spinal cords with the meninges intact. A 1-cm segment from lumbar 5–6, lumbar 3–4, and lumbar 1–2 was cut, respectively, and stored in formalin for complete fixation. After being embedded in paraffin, transversely cut in 5-μm-thick sections, mounted on glass slides, and stained with hematoxylin-eosin (H&E), the segments were observed under light microscopy.
Statistical Analysis
The biochemical assay data of CSF were collected and intergroup comparisons were made by using a t test. P values less than .05 were considered to indicate a significant difference. All analyses were performed by using SPSS 11.0 statistical software (SPSS, Chicago, Ill).
Results
Behavior of the Animals after the Procedure
All the animals resumed eating and drinking the next day after the procedure; no animals demonstrated claudication, paralysis, and any other abnormal neurologic behavior.
CSF Assay
All the samples of CSF were colorless, lucid, and negative in protein qualitative analysis. The concentrations of glucose, protein, and chloride as well as white blood cell count and total cell count at various time points were in the normal range. There was no statistically significant difference among them (Table).
The values of the composition of the CSF in a biochemical examination (Mean ± SD)
Imaging Examination
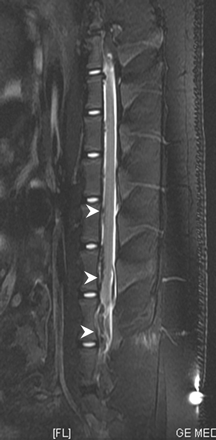
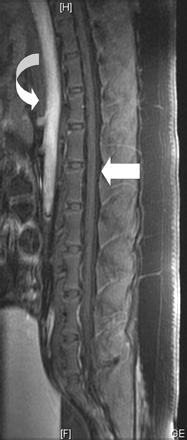
CT and MR imaging 30 minutes after injection showed that ozone was injected successfully into the subarachnoid space (Fig 1). MR imaging without and with contrast material showed that there was no abnormal signal intensity in spinal cord and leptomeninges at various time points (Figs 1 and 2).
Sagittal FSE T2-weighted MR image (TR/TE, 4000/110 ms) scanned 30 minutes after ozone (10 mL, 90 μg/mL) injection into the subarachnoid space shows gas (white arrowheads) in the subarachnoid space. There are no signal intensity changes in the spinal cord.
Sagittal FSE T1-weighted contrast-enhanced MR image (TR/TE, 500/15 ms) scanned 1 week after ozone (10 mL, 90 μg/mL) injection into the subarachnoid space shows gas absorbed in the subarachnoid space. There is no abnormal enhancement in the spinal cord, dural sac, arachnoid, or nerve roots (white straight arrow) during the arterial phase. Aorta enhancement also can be seen (white curved arrow).
Gross Pathologic Examination
The appearance of the spinal cords was grossly normal. There were no swelling, adhesion, discoloring, and tarnishing. The vessels of the dural sac were vivid and visible. The color and texture of nerve roots were normal in appearance (Fig 3).
Animals were sacrificed 1 week after ozone (10 mL, 90 μg/mL) injection into the subarachnoid space, and the spinal cord was obtained. The appearance of the spinal cords is normal. There is no discoloring or tarnishing, and the vessel is vivid and visible.
Micropathologic Examination
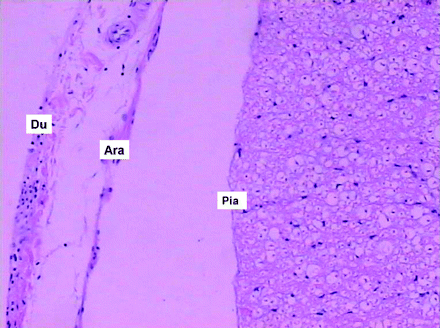
There were no serious changes micropathologically in the spinal cords, dural sac, arachnoid, nerve roots, and nerve tracts. No histologic evidence of degeneration or necrosis of nerve cell or fiber (eg, focal demyelination, axonal edema, cellular swelling) was found. There was only individual slight congestion in spinal cord vessels, probably due to puncture for the CSF sample before sacrifice (Fig 4).
Histologic sample from the spinal cord in the lumbar 5–6 level 1 week after injection shows no abnormal changes in the dura (Du), arachnoid (Ara), pia mater (Pia), nerve tracts, or nerve roots (H&E, original magnification, ×40).
Discussion
Ozone has a very strong oxidation property that can oxidize the nucleus pulposus of intervertebral disks.3–5 When the proteoglycans in the nucleus pulposus are oxidized, it decomposes and releases water molecules, resulting in subsequent degeneration of the matrix, which is replaced by fibrous tissues; these events result in a reduction in disk volume. A reduction in herniated disk volume may reduce nerve root compression. On the other hand, the analgesic and anti-inflammatory effects of ozone may counteract disk-induced pain.6–11 At present, the concentration and dose of ozone have not been standardized; we usually use 60 μg/mL for intradisk injection and 40 μg/mL for paraspinal tissue injection. Generally speaking, the concentration of ozone is classified into 3 types: low (10–30 μg/mL), medium (40–60 μg/mL), and high (70–90 μg/mL); the higher the concentration of ozone, the stronger is the oxidation of the tissue. Our study showed that there was no obvious bad result after intra-subarachniod space injection of high-concentration ozone (10 mL, 90 μg/mL). The values of the compounds in the CSF sampled at various time points were in normal ranges, and the spinal cords showed no serious injury pathologically or radiologically. No changes occurred like those in intra-subarachniod space injection of collagenase, such as chemical arachnoiditis, and there were no meningitis, myelitis, radiculitis, or microlesion of infarction of the spinal cord. In our clinical practice, we have not yet found severe sequelae when minimal ozone is inadvertently injected into subarachnoid space. However, the concentrations and doses in this study were much higher than those in clinical inadvertent injection. There was no serious damage but only slight arteriolar hyperemia in some spinal cords at different time points. There was no chronologic relationship with the injection of ozone, but rather with repeated puncture. We speculate that the hyperemia is correlated with repeated sampling of CSF before animal sacrifice.
The reasons for no serious nerve damage may be interpreted as follows: 1). Ozone decomposes more rapidly in watery CSF than in the disk or paraspinal muscles and avoids injury of structures in the subarachnoid space such as leptomeninges, nerve roots, and spinal cord. 2). Buffering systems in CSF like those in plasma palliate the oxidation of ozone for structures in the subarachnoid space. It is well known that CSF is a watery solution containing ions and different substances. Despite the different composition in relation to plasma, the osmolality (289 mOsm/L) and the main composition of the 2 fluids remain the same. Therefore, we can speculate that there are buffering systems in CSF just like in plasma, including some deoxidizers such as albumin and reduced glutathione, that can counteract the oxidative effect of ozone on other tissue, and the structures such as leptomeninges, nerve roots, and spinal cord remain untraumatized.
This study had several limitations. First, the routine and biochemical assays could not include all the compositions in CSF because it is well known that they are complicated. Even if no changes occur in the routine and biochemical assays, components may still change; especially the detection of antigens, enzymes, and other metabolites that may play an important role in the palliation of ozone oxidation was not included in this study. Second, electron microscopy was not used in this study because we found that there were no obvious changes occurring under light microscopy. Although there were no overt neurologic symptoms to suggest nerve damage, perhaps there was evidence of more subtle damage that would only be evident with electron microscopy or more sophisticated tissue processing for light microscopy. Alternatively, nerve-conduction studies or other functional studies may have been appropriate to evaluate damage related to the ozone exposure. Last, there was only a total of 8 animals in this study, 2 at each time point. One of these points (the 3-month point) only had 2 values, and this limited the meaning at this point.
Conclusion
Our initial study suggests that high-concentration ozone injection into a pig's subarachnoid space will not cause serious sequelae such as chemical arachnoiditis and necrosis of the spinal cord, and this provides evidence of the safety of inadvertent injection of ozone into the subarachnoid space in the clinical treatment of disk herniation.
References
- Received July 18, 2006.
- Accepted after revision September 22, 2006.
- Copyright © American Society of Neuroradiology