Abstract
BACKGROUND AND PURPOSE: Few collected series of cerebral microarteriovenous malformations (micro-AVMs) have been reported. Our propose was to assess the unique diagnostic and therapeutic challenges posed by these lesions and their influence on outcomes.
METHODS: The clinical presentation, diagnostic features, principles of endovascular or surgical treatment, and outcomes for a consecutive series of 10 patients (five male, five female; mean age, 48.8 years; age range, 31–65 years) with angiographically demonstrated cerebral micro-AVMs were retrospectively analyzed.
RESULTS: All patients presented with a cerebral hematoma (supratentorial in eight, infratentorial in two, intraventricular in one, subarachnoid in one; mean volume, 11.6 cm3), which was superficially situated in nine patients. Neurologic deficits were observed in nine patients, and three patients had seizures. The mean delay between the onset of symptoms and diagnosis was 129.8 days (range, 6 days to 1 year). Superselective angiography was performed in seven patients and followed by successful acrylic embolization of the lesion in five. Five patients underwent surgical intervention, which led to definitive resection. Although long-term neurologic problems were present in eight patients, they were able to return to their previous activities and employment.
CONCLUSION: The diagnosis of cerebral micro-AVMs requires a high index of suspicion, especially in young adults with atypical hemorrhaging. Single-shot embolization of micro-AVMs may be a safe alternative to the established surgical therapy in select cases. Outcomes depend mostly on the clinical conditions at admission.
Cerebral microarteriovenous malformations (micro-AVMs) were defined by Yasargil (1) as a subgroup of pial AVMs characterized by a nidus with a dimension of 1 cm or less. Micro-AVMs are a rare entity, accounting for about 8% of all cerebral AVMs in surgical series, but they are usually a source of clinically relevant cerebral hemorrhage in young adults (2), representing a diagnostic and therapeutic challenge. Furthermore, few collected series of cerebral micro-AVMs (3–4) have been reported. Most recently, major technical progresses in the field of endovascular therapy have expanded the armamentarium of treatment strategies for cerebral AVMs. The purpose of our study was to assess the diagnostic and treatment issues posed by surgical and endovascular therapy in a series of consecutive patients with cerebral micro-AVMs.
Methods
We retrospectively reviewed findings in a series of 10 patients (five male, five female; mean age, 48.8 years; age range, 31–65 years) with cerebral micro-AVMs who were treated at our institution between January 1996 and December 2000. All clinical, diagnostic, and therapeutic data were obtained by evaluating the office and hospital charts, imaging studies, and surgical and/or endovascular reports. Inclusion criteria consisted of angiographic findings of pial, plexiform arteriovenous shunting with a nidus diameter of less than 1 cm. Patients with Rendu-Osler-Weber disease, those with cerebral neoplasias, and those with partially thrombosed AVMs were excluded. All patients underwent contrast-enhanced CT of the brain at admission and then one or more control examinations, for a total of 24 CT examinations. Contrast-enhanced cerebral MR images were available in seven patients (five examinations performed after episodes of hemorrhaging and two performed before). Two patients also underwent three-dimensional time-of-flight (TOF) MR angiography (MRA) of the intracranial vessels performed prior to hemorrhaging. All patients underwent one or more cerebral digital subtraction angiographic (DSA) examinations with selective catheterization of the internal carotid, external carotid, and vertebral arteries. One or more superselective angiographic examinations were performed in seven patients. Overall, a total of 25 conventional (n = 15) and superselective (n = 10) angiographic examinations were performed.
Two neuroradiologists (S.M., M.C.) reviewed all imaging studies in a nonblinded fashion. All CT and MR images were evaluated for the presence of abnormal foci of enhancement or flow voids. The volume of parenchymal hematomas was calculated according to the following formula: (4/3)π(abc), where a, b, and c represent half of the maximal diameters of the lesion along the three main spatial axes. Selective and superselective cerebral DSA were analyzed for the presence and angioarchitectural features of the nidus, phase of visualization, and characteristics of the feeding vessels (eg, hypertrophy, aneurysms) and draining vessels (eg, venous ectasia and/or stenosis, superficial and/or deep venous drainage).
Treatment consisted of surgical resection in five patients and endovascular embolization with a polymerizing agent in five patients. The decision about performing surgery or embolization was always made after a case-by-case discussion with the neurosurgeons (H.N., F.A., P.M.) and based on the patient’s clinical picture, the size of the parenchymal hematoma, and the location and characteristics of the micro-AVM. The same neuroradiologist (S.M.) performed the embolization procedures in all patients under general anesthesia. A 6F groin sheath was inserted into the right common femoral artery. A loading bolus of 5000 IU heparin followed by an infusion of 1000 IU/h was administered. A 6F guiding catheter (Envoy; Cordis Endovascular Systems, Miami Lakes, FL) was placed in one of the four cerebral vessels, and a microcatheter (Flow rider; MTI, Irvine CA) primed with a 0.010-inch guidewire (Silver Speed 0.010 inch) was subsequently advanced proximal to the nidus. A single 0.2–0.4-mL injection of a 20% mixture of a polymerizing agent (Histoacryl; Braun, Melsungen, Germany) and Lipiodol (Guerbet, Roissy, France) was delivered via a microcatheter that was wedged in the nidus or placed in the terminal arterial feeder just proximal to the nidus. Procedural times, which included patient preparation, ranged from 2.30 to 3.30 hours (mean, 3.05 hours). All patients underwent immediate angiographic posttreatment studies. In eight patients, a delayed follow-up cerebral angiography was also performed after 3–6 months. All patients were available for clinical follow-up. The mean duration of follow-up monitoring was 23.0 months (range, 2 months to 4 years).
Results
The clinical presentations, diagnostic workup results, angiographic findings, course of treatment, and clinical and angiographic follow-up results for all 10 patients are reported in Tables 1 and 2.
Overview of clinical and diagnostic features and therapy in the 10 patients with cerebral micro-AVMs
Overview of diagnostic workup results, therapeutic course, angiographic follow-up findings, and outcome
Clinical Features
All patients were admitted to hospital because of a hemorrhagic ictus. Neurologic prodromes were characterized by a partial right hemisensory-motor seizure that preceded a left temporoparietal hemorrhage by 1 week in one patient, and an episode of severe headache and vomiting followed by intermittent visual disturbances that preceded a left occipital hematoma with subarachnoid extension by 1 month in another patient. Two patients reported histories consistent with a previous hemorrhagic ictus 8 and 13 years earlier. Nine patients had neurologic deficits with the hemorrhagic ictus; these consisted of a decreased level of consciousness (n = 3), monohemiplegia (n = 4), speech disturbances (n = 1), hemisensory deficits (n = 1), and visual deficits (n = 2). One patient had headache, dizziness, and vomiting, without neurologic deficits. Seizure accompanied the hemorrhagic ictus in three patients. Three patients were in a coma: one had a GCS score of 4; one, a GCS score of 8; and one, a GCS score of 12. In all patients, the mean delay between the onset of neurologic symptoms and the diagnosis of the micro-AVM was 129.8 days (range, 6 days to 1 year).
CT and MR Findings
In both patients who underwent contrast-enhanced MR examination of the brain prior to the hemorrhagic ictus, images yielded no evidence of abnormal findings that could possibly be related to the micro-AVM. Similarly, none of the CT and MR imaging–MRA examinations performed at admission revealed any findings consistent with the micro-AVM. In all patients, CT and MR studies demonstrated a parenchymal hematoma with a mean volume of 11.6 cm3 (range, 3.0–35.15 cm3). The hematoma was associated with a notable mass effect and a contralateral shift of the midline structures (1 cm) in three patients. In nine patients, the hematomas were superficial and either supratentorial (left parietal lobe, n = 2; left frontoparietal lobes, n = 1; left frontal lobe, n = 1; left occipitoparietal lobe, n = 1; right temporoparietal lobe, n = 1; right temporal lobe, n = 1) or infratentorial (inferior cerebellar vermis, n = 1; right cerebellar tonsil, n = 1). In one patient, the hematoma was deep in the wall of the right ventricular temporal horn and was associated with an intraventricular hemorrhage.
Angiography
In the acute-subacute settings, four-vessel cerebral DSA findings were diagnostic of the micro-AVM in five patients and yielded either questionable (n = 3) or negative (Fig 1A and B) results (n = 2) in the other patients. In all cases, diagnostic findings consisted of a small feeding artery, a tiny, poorly defined nidus, and an early draining vein to superficial or deep venous structures (Figs 1C, 2A and B, 3A). The micro-AVM opacified late in the arterial phase before filling in the capillary phase. Questionable findings consisted of a capillary blush (n = 1); prolonged distalarterial opacification (n = 1); venous pooling (n = 1); and, most important, early venous filling (n = 3) in the absence of a clearly defined nidus or feeding artery. In all patients but one with questionable or negative results, evidence of slight mass effect was present on CT and/or MR images, and control DSA was scheduled after hematoma resorption (1–2 months later). Control DSA showed diagnostic findings in three patients and questionable findings in two patients, who underwent superselective exploration a few days later. The arterial feeder did not show evidence of hypertrophy in any case and the only occasional a posteriori feature was represented by prolonged distal filling. The arterial feeder usually had the same caliber as the draining vein, and in five patients, the distal tract of the feeder and the proximal tract of the draining vein had a parallel course (Fig 2B). In one patient, two consecutive flow-related small aneurysms were seen in the arterial feeder. No dural contribution for any micro-AVM was observed. The nidus was best characterized with superselective angiography and consisted of a plexiform agglomerate of fine vessels that constituted multiple arteriovenous shunts (Figs 1D, 2C and D, 3B). Venous drainage involved single (nine patients) or double (one patient) draining veins through cortical (seven patients) or deep (three patients) veins. In one patient, two consecutive stenoses of the draining vein was noted (Fig 3A and B). In one patient, a small venous pouch was seen, and in another, the venous drainage occurred through a slightly enlarged or prominent vein.
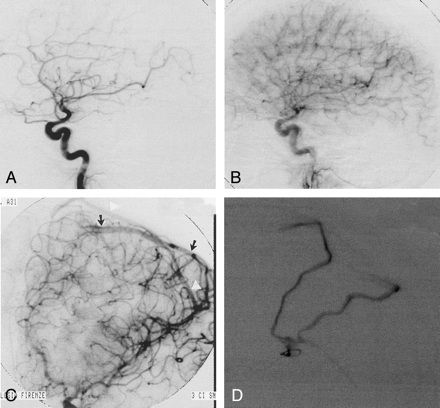
Imaging studies in a 31-year-old man (case 2) with a cortical hematoma in the left angular and supramarginal gyri.
A and B, At admission, lateral early (A) and late (B) arterial-phase angiograms of left internal carotid artery do not show evidence of arteriovenous shunting.
C, Oblique left internal carotid arterial-phase angiogram obtained 1 month later, after hemorrhage resorption, shows early venous filling of two cortical veins that drain in the superior sagittal sinus (arrows and arrowheads).
D, Subsequent superselective exploration of the angular branch of the left middle cerebral artery reveals the feeder, the nidus, and the double superficial venous drainage of the micro-AVM. After stable microcatheter positioning, treatment of the lesion was performed by using a single injection (ie, single shot) of a polymerizing agent.
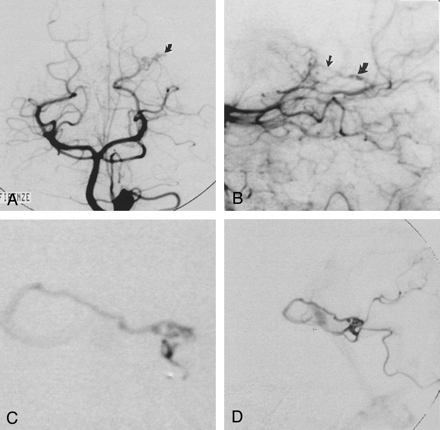
Imaging studies in a 43-year-old woman (case 1) with a large hematoma in the left occipitoparietal lobe.
A and B, Frontal (A) and lateral (B) late arterial-phase left vertebral angiograms show a small tangle of arterial blush (curved arrow) and a questionable early venous filling (straight arrow) along the parieto-occipital branches of the left posterior cerebral artery.
C, Superselective exploration of the parieto-occipital branch demonstrates the plexiform structure of the nidus of the micro-AVM, which is characterized by multiple arteriovenous shunts and a single deep draining vein coursing toward the vein of Galen.
D, The lesion could not be embolized because microcatheter tip instability and opacification of functional vessels originated from the distal tract of the feeder. The patient subsequently underwent successful surgery.
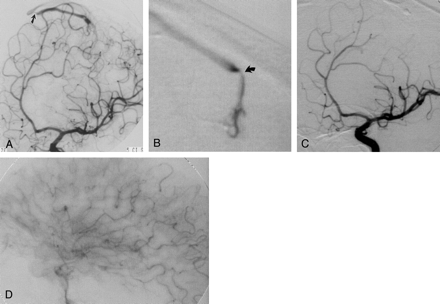
Imaging studies in a 41-year-old man (case 4) with a large hemorrhage of the left paracentral lobule.
A, Oblique arterial-phase left internal carotid angiogram shows early venous drainage in the absence of a clearly defined nidus. Note stenosis of the draining vein at the junction with the superior sagittal sinus (arrow).
B, Superselective exploration of the paracentral branch of the left callosomarginal artery allows identification of the nidus with demonstration of another stenosis (arrow) in the proximal tract of the draining vein.
C and D, After successful embolization, left internal carotid angiograms show the glue cast of the nidus and the draining vein (C) in the absence of any residual arteriovenous shunting (D). C is an arterial-phase oblique view; D, a capillary-phase lateral view.
Treatment
Six patients underwent surgical intervention (group B), and resection of the micro-AVM was successful in five patients. Superselective exploration of the lesion was performed in seven patients (group A), and it was followed by single-shot embolization (Figs 3C and D) of the lesion in five patients (Table 2). Overall, 10 superselective procedures were performed. In two group A patients, endovascular treatment of the lesion failed because of the occurrence of a clinically uncomplicated vasospasm of the feeding vessel in one patient and suboptimal microcatheter positioning in the other (Fig 2D). Both patients subsequently underwent surgery. In one patient, the final postembolization angiograms revealed residual early venous drainage, and a second session of embolization was successfully repeated 6 months later. In any case, obliteration of the draining vein by using glue emboli occurred without complete exclusion of the nidus.
In two group B patients, the micro-AVM was identified by exploring the walls of the cavity after hematoma decompression. In two other patients, the nidus was identified by following the arterialized draining vein, in a retrograde fashion, into the sulcal spaces, which revealed the site of the arteriovenous shunting. In one patient, limited corticectomy was necessary to expose the nidus. After resection of the micro-AVM nidus, the arterialized draining vein turned blue in both cases. In one patient with deep venous drainage, the nidus was identified as a result of surgical acumen and intraoperative angiography. In one patient with a deep-seated micro-AVM, surgical identification of the lesion failed, and the patient subsequently underwent successful endovascular treatment.
Outcome
Micro-AVM obliteration was verified at delayed DSA in all group A and three group B patients. All patients had improvements after hemorrhaging. Neurologic problems persisted in eight patients (visual disturbances, n = 1; hemimonoplegia, n = 4, language disturbances; n = 1; cognitive impairment, n = 1; new seizure disorder after surgery, n = 1). Neurologic deficits were of moderate severity, allowing all but two patients to return to his or her previous activities and employment (Barthel index, 95–100). One patient was admitted with a coma and score of GCS 4, and although he recovered from deep coma to resume an independent lifestyle, he had a residual left-sided hemiplegia (Barthel index, 50). The other patient with a residual right-hand weakness and numbness was not able to perform the skilled handwork his work as a goldsmith required. However, he had only undergone 2 months of follow-up and was expected to improve.
Discussion
Tiny arteriovenous shunts that are histologically consistent with micro-AVMs may represent a possible source of major cerebral hemorrhaging, especially in young adults (5–10). In two patients of our series, however, the hemorrhagic ictus was preceded by prodromal symptoms that were probably related to small sites of bleeding that were not documented with neuroimaging. Although the tendency of micro-AVMs to hemorrhage cannot be calculated because of the lack of data, the predominant presentation of micro-AVMs involves intracranial hemorrhage. This finding is supported by the observation that small AVMs hemorrhage more frequently than larger ones (11–13). Higher pressures in the feeding pedicles of small AVMs and a higher tendency for hemorrhaging in low-shunt AVMs compared with high-shunt AVMs have been reported (14–16). Micro-AVM bleeding has been related to small bridging arteries that anchor the draining vein to the surrounding parenchyma, resulting in mechanical venous outflow hindrance and hyperpressure (3). On the other hand, their small size and angiographic features, consistent with those of low-flow low-volume AVMs, explain the lack of compressive and/or ischemic symptoms due to steal phenomena. Our findings are in agreement with the data in the literature in that micro-AVM hemorrhaging causes substantial neurologic impairment (3). The effect seems to be ascribed to the dimensions of the parenchymal hematoma and, most of all, its cortical location that frequently involves eloquent areas.
Diagnosis of micro-AVMs can be difficult and challenging. Micro-AVMs can be angiographically occult, and only careful surgical exploration of the hematoma cavity reveals the lesion on the walls of intracranial hematomas (17). Since the introduction of CT and MR imaging into everyday clinical practice, occult vascular malformations (including capillary telangiectasias, venous and cavernous angiomas, and AVMs), also called cryptic malformations, have considerably shrunk, depending on the size, the type of the malformation, and the presence of accompanying bleeding.
In our series, it was interesting to note that cerebral MR images obtained before the hemorrhagic ictus did not show evidence of either abnormal flow-voids or hemosiderin deposits related to the presence of the micro-AVM or chronic microbleedings. This fact limits their usefulness as incidental findings. Conventional DSA findings may be subtle or even completely negative, especially in the acute phase; these findings are probably related to the mass effect of the concomitant hematoma with transitory displacement and compression of the micro-AVM. In our series, this explanation could not be sustained in one patient whose hematoma did not show evidence of mass effect; in patients such as this one, other mechanisms are needed to explain the normal cerebral angiographic findings at admission. It is our opinion that transitory thrombosis or extremely slow flow phenomena in the nidus after bleeding may possibly hinder diagnosis. Furthermore, the diagnosis of micro-AVMs requires high-quality angiography with a large matrix (1024 pixels), rapid frame acquisition (more than three frames per second), magnification, and multiplanar imaging. In our experience, negative or questionable conventional DSA findings in young adults with atypical hemorrhaging should prompt a repetition of the examination after complete resorption of the cerebral hematoma, and superselective exploration should eventually follow. Micro-AVMs can be missed at DSA because of the absence of substantial differences between the arterial feeder and the draining vein, both in terms of their caliber and course; this lack can hinder recognition of early venous filling. This finding, however, is nonspecific and occurs in inflammatory processes, as well as in trauma, infarction (luxury perfusion), and epileptic foci; it may also represent an epiphenomenon of the hematoma itself. In our series, superselective exploration of the lesion increased the diagnostic yield, allowing definite diagnosis in two patients with questionable or negative conventional DSA findings. Furthermore, superselective exploration of the arterial pedicle enabled precise anatomic location of the lesion and characterization of the angioarchitecture of the nidus. The first finding resulted in easier localization and depiction of the lesion in the operating field, which facilitated the surgeon’s task. The second finding allowed differentiation between AVMs in which the nidus was a plexiform agglomerate of multiple arteriolovenular shunts and cortical fistulae characterized by pial arteriovenous shunts in a single vein. Such differentiation is usually not possible with conventional DSA because micro-AVMs usually involve a single compartment, and therefore, a single draining vein, as usually happens in cortical fistulae. This differentiation could entail major therapeutic consequences. Complete resection or obliteration of the nidus is the therapeutic aim of definitive treatment of AVMs, and cutting off only the venous drainage proximal to the nidus, as usually performed in arteriovenous fistulae, is insufficient. Venous drainage was also better characterized with superselective DSA, which allowed the neurosurgeon to more easily recognize the arterialized draining vein to be followed to the nidus in retrograde fashion.
Surgical resection of small and micro-AVMs represents the consolidated therapy, especially with superficial lesions that present with hemorrhage. Endovascular embolization or radiosurgery may be considered as alternative therapeutic modalities; these are suggested as primary therapies for deep-seated lesions or for cases in which functionally important brain tissue is interposed between the nidus and the cortical surface. Experience with both radiosurgical and endovascular treatment for micro-AVMs is limited, and the effectiveness of both modes of treatment remains to be demonstrated. One patient in the series reported by Willinsky et al (3) was primarily treated with radiosurgery, and rebleeding was not observed at follow-up. Another patient in the surgical series reported by Stiver and Ogilvy (2) was treated with stereotactic Bragg-peak proton-beam radiosurgery. and persistence of the shunt was demonstrated at follow-up angiography 2 years later. In our series,endovascular treatment resulted in stable obliteration of the nidus without peri- or postprocedural clinical complications. Safe glue injection requires the extremity of the microcatheter to be wedged into the nidus or at least positioned in the terminal feeding pedicle just proximal to the nidus. The major technical limiting factor to successful embolization of micro-AVMs in our series was the absence of feeder hypertrophy. Microcatheter navigation was also hindered by the feeder length and tortuosity. These conditions resulted in the failure to reach a safe injection site for the glue in two patients, who underwent surgery. However, in the near future, such constraints may be overcome by the technologic improvement of materials for endovascular therapy. Because micro-AVMs are tiny lesions, their accurate localization represents one of the major problems encountered during surgical resection. Empiric exploration of the hematoma cavity may produce both false-positive and false-negative results, and it may be dangerous for the cortex overlying the walls of the hematoma cavity. In our series, the preferred surgical approach consisted of following the arterialized vein to the nidus in retrograde fashion. Although this approach may sometimes imply a limited corticectomy (usually in the absence of clinical consequences, as was the case in one of our patients) it allows us to confirm total resection of the lesion by demonstrating transformation of the arterialized draining vein from red to blue. Unfortunately, this technique was possible only in patients whose arterialized draining veins had a superficial course. Superselective exploration of the lesion, with surgical acumen and intraoperative angiography, was an extremely valuable adjunct for accurate localization of the nidus when the draining vein of the micro-AVM did not traverse the cortical surface but had a deep course.
Neurologic morbidity rates after AVM hemorrhage average 20–30% (11, 12, 16, 18). Our series was too small to allow us to draw any statistical conclusions about the relationships between micro-AVM localization and outcomes. However, the findings are in agreement with reported data in that, even if micro-AVM hemorrhaging represents a devastating event, we observed a marked recovery after treatment in most cases. Of the three patients who were admitted in a coma, all recovered to resume an independent lifestyle. Residual neurologic deficits occurred in more than 50% of our patients and were primarily attributable to the initial hemorrhage correlated to the size and location of the hematoma. One patient had new late-onset seizures. Perihemorrhage and delayed-onset seizures are a well known occurrence in micro-AVMs haemorrhaging. Cortical location and histories of previous hemorrhage are reported to be risk factors for late-onset seizures (19, 20). In our series, all patients presenting with simple partial seizures had a cortical nidus, and one of the patients who had new late-onset seizures had a history suggestive of a previous hemorrhagic episode.
Conclusion
The diagnosis of a cerebral micro-AVM requires a high index of suspicion. CT and MR results are usually inconclusive, and conventional DSA findings may be questionable or negative, especially in the acute phase. Because micro-AVMs represent a treatable cause of major cerebral hemorrhaging, more aggressive diagnostic workup should be advocated, especially in young adults with a large atypical parenchymal hematoma in the absence of any other obvious source of bleeding. In our experience, superselective exploration of the lesion increases the diagnostic yield, enables differentiation between AVM and cortical fistulae, and facilitates the surgeon’s task. Although surgical excision of the nidus still represents the consolidated therapy, endovascular acrylic embolization may be a possible alternative in select cases. The outcome depends mostly on the clinical conditions at admission, which are a direct reflection of the volume of the hematoma and its location. No neurologic deficits developed in the patients described herein because of neurosurgical or endovascular treatment.
References
- Received June 20, 2001.
- Accepted after revision February 18, 2002.
- Copyright © American Society of Neuroradiology