We enjoyed reading the recent article by Zhou et al (1) on the application of quantitative diffusion imaging to the differential diagnosis of benign versus metastatic vertebral compression fractures. This article and the accompanying editorial provided useful insights into both the clinical dilemma posed by this problem and the limitations of current MR imaging methodology in addressing it. As we see it, the major “talking point” from the article is that quantitative diffusion imaging improves upon the use of qualitative diffusion-weighted imaging by eliminating the confounding effects of “T2 shine-through.” Although this improvement on the use of diffusion-weighted imaging alone is appreciated, it is our concern that the article under consideration and the few previous works in this arena (2–4) have either ignored or failed to appreciate adequately the extent to which lipid signal intensity within vertebral marrow may contribute to quantitative measurements of the tissue diffusion coefficient. Indeed, the discussions in these articles regarding the differences between diffusion characteristics of benign versus metastatic vertebral compression fractures tend to focus on the same biophysical mechanisms used to describe diffusion characteristics in brain and brain abnormalities where, of course, any lipid component can be safely ignored. This is not so in vertebral marrow; we demonstrate in this letter how even a fairly small fraction of lipid within a voxel can drastically affect diffusion coefficient measurements.
Healthy vertebral marrow contains some 20–70% lipid (5–12) with, according to the early spectroscopic studies of De Bisschop et al (5), approximately 7% increase in fat percentage per decade of life. Thus, a voxel of healthy vertebral marrow will contain a substantial signal intensity component from lipid. With the infiltration of malignant tumor or a more benign edematous process, the lipid component of the proton signal intensity can be expected to diminish. In our opinion, however, complete replacement of the lipid component with water will represent only an extreme condition. Thus, the tendency to ignore the lipid contribution—for either qualitative interpretations of diffusion-weighted imaging (2–4) or quantitative tissue diffusion measurements (1) within vertebral marrow, particularly when fat saturation or selective water excitation is not included in the pulse sequence design (1–4)—is simply not appropriate.
To justify this opinion requires consideration of how the lipid signal intensity can be anticipated to affect quantitative tissue diffusion coefficient measurements in vertebral marrow. Here we rely on our own measurements of the lipid diffusion coefficient in human scalp in vivo, which revealed that the large, slowly diffusing triglyceride molecules have a low diffusion coefficient Df, of approximately 0.05 μm2/ms (13). Let us now assume for the sake of argument that the water tissue diffusion coefficient Dw, takes a value of 1.9 μm2/ms as reported by Zhou et al for metastatic fractures within vertebral marrow (1). Because water and lipid protons do not exchange, the overall decay of signal intensity with b factor will then be described by a biexponential function of the form 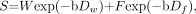 1
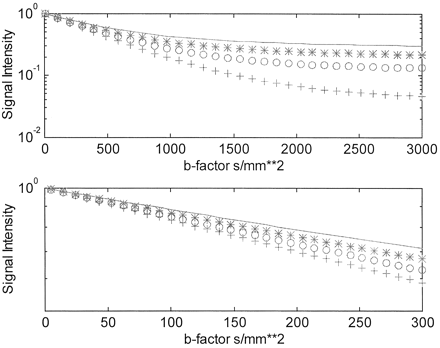
1
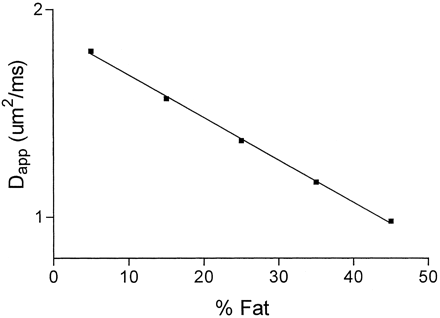
where W and F are the apparent amplitudes of the water and fat protons, respectively. The term “apparent” is used, because W and F do not represent solely the respective water and fat proton densities, but also T1 and T2 weightings. These in turn depend on the specifics of the pulse sequence (echo and reetition times) in combination with the fat and water relaxation times for which there is an established literature (9–12). Letting F + W = 1 for normalization purposes, simulations based on equation 1 were generated for both an extended b factor range up to 3000 s/mm2 and for the more limited range below 300 s/mm2 employed by Zhou et al and others to study diffusion in vertebral marrow (1–4). Semilog plots of S versus b factor are provided in Figure 1, where the different curves in each plot represent different fat fractions from 0.05 (+), 0.15 (0), 0.25 (*),and 0.35 (−). Over the extended b factor range (top plot) the nonmonoexponential nature of equation 1 reveals itself quite clearly as a curvature over the extended b factor range. Over the more limited b factor range relevant to the current clinical studies (lower plot), the decays actually appear monoexponential, but with lower diffusion coefficients (smaller slopes) observed with increasing fat fraction. Closer analysis of the simulated slopes reveals a nearly linear decrease with fat fraction in the apparent diffusion coefficient Dapp, in this low b factor range. Figure 2 shows how a “monoexponential”-based Dapp would be measured by using four b factors of 5, 70, 135, and 200 s/mm2 as the fat fraction increases from 0.05 to 0.45. The correlation coefficients r2 for the linear regressions of the ln(S) versus b factor fits used to obtain the Dapp values were all above 0.999, effectively disguising the biexponential nature of the signal intensity decay due to the lipid component in this b factor range. Rather, the primary effect of increasing fat fraction in this low b factor range is to “artificially” lower Dapp. The simulated results of Figure 2 are well represented by a linear regression fit as 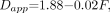 2
2
with an r2 > 0.99.
To conclude, we were concerned that Zhou et al and others who have begun to explore the potential of diffusion imaging in vertebral marrow have paid too little attention to the consequences of a lipid component on diffusion-weighted images and quantitative diffusion measurements. Figure 2 demonstrates how even small percentages of lipid within a voxel can dramatically affect the measured value of the diffusion coefficient. Because the non-echo-planar–based pulse sequences used to date for the vertebral marrow studies have not employed water selective or fat suppression methods (1–4), many of the forwarded interpretations of diffusion characteristics based on water specific models common to brain discussions seem premature.
Simulated semilog plots of signal intensity versus b factor for marrow with varying fractions of fat for extended b factor range (top plots) and the limited b factor range used to date in most studies of vertebral marrow.
Plot of the apparent diffusion coefficient as a function of fat fraction in vertebral marrow when measured over the limited b factor range from 0 to 200 s/mm2.
References
Reply:
We thank Drs. Mulkern and Schwartz for their careful review of our article (1) and appreciate the opportunity to respond to their comments.
Drs. Mulkern and Schwartz expressed concern that our article, as well as several others on the same subject (2, 3), did not discuss the influence of lipid signal on diffusion-weighted images and on measurement of apparent diffusion coefficient (ADC) of vertebral bodies. On the basis of a two-compartment model that accounts for water and fat, they presented an empirical relationship between the ADC and the lipid fraction F (see Equation 2 of Drs. Mulkern and Schwartz’s letter). This relationship indicates that the presence of lipid signal can contaminate the results in quantitative diffusion analysis, an aspect we did not consider in our article.
In healthy vertebral marrow, lipid content can be as high as 70% (4). Therefore, it is not negligible. For the two diseases we studied, however, the lipid fraction in the vertebral marrow can be considerably less. According to a study by Yuh et al (5), approximately 88% of vertebral metastasis cases exhibited total replacement of normal bone marrow. Similarly, edema in benign vertebral compression fractures can also replace normal marrow, resulting in substantially decreased lipid content in the lesion (6). In our study, the ADC values were calculated within a region of interest that contained only the vertebral lesion. Within the region of interest, the signal intensity of diffusion-weighted images arises predominantly from water, as evidenced by the T1-weighted, T2-weighted, and postcontrast T1-weighted images with or without fat suppression (Figs 1 and 2 in our article [1]). Therefore, we think it is appropriate to qualitatively discuss our results by using a water-only model.
Quantitatively, lipid signals within the region of interest can arise from either incomplete bone marrow replacement or the partial volume contamination. Thus, we agree with Drs. Mulkern and Schwartz that the possible presence of lipid should be considered. If we assume that the lipid fraction ranges from 0% to 20% among the regions of interest of the metastatic lesions in our study, equation 2 of Drs. Mulkern and Schwartz’s letter indicates that the ADC value would vary between 1.88 and 1.48 mm2/s, with a standard deviation of 0.2 mm2/s. This standard deviation is not larger than what we observed in the study and is already contained in the error term (±0.3 mm2/s) we reported.
For normal vertebral bodies, wherein the lipid content cannot be neglected, we completely agree with Drs. Mulkern and Schwartz that a two-compartment model should be used to characterize the signals in diffusion-weighted images and determine the ADC values for water and fat separately. The primary focus for our article, however, was to present a possible way to differentiate benign from malignant vertebral compression fractures, not to provide a detailed account of the diffusion process in normal vertebral bodies.
In summary, Drs. Mulkern and Schwartz raised an important question in analyzing diffusion-weighted images and interpreting the ADC values in regions wherein both water and lipid signals are present. Within benign and malignant vertebral compression fractures, the impact of the lipid signal intensity is minimal. Thus, our explanation based on water diffusion remains valid and our conclusions unchanged. To extend quantitative diffusion imaging to other vertebral lesions, such as hemangiomas, we recommend that fat suppression techniques (eg, by using chemical suppression or spatial spectral excitation radio-frequency pulses) be employed to reduce the effect of lipid signal intensity on ADC quantification.
- Copyright © American Society of Neuroradiology