Abstract
BACKGROUND AND PURPOSE: We prospectively studied the incidence and natural history of Horner syndrome following stent-supported percutaneous angioplasty of the carotid artery (SPAC). We assessed the hypothesis that postinterventional Horner syndrome is related to ipsilateral carotid wall hematoma.
METHODS: We performed duplex sonography of the carotid arteries and clinical examination in 28 consecutive patients before and 4 hours ± 2, 24 hours ± 2, and 7 days ± 1 after SPAC.
RESULTS: Within 24 hours ± 2 after SPAC, 11 of the 28 patients developed ipsilateral Horner syndrome. Ten of these 11 patients had a postinterventional sonographic appearance of a carotid wall hematoma, whereas only two of the 17 patients without Horner syndrome had this finding (P < .001). In all patients, both Horner syndrome and carotid wall hematoma had resolved 7 days ± 1 after SPAC.
CONCLUSION: Horner syndrome appears to be a common yet transient event after SPAC. Although postinterventional Horner syndrome is significantly related to ipsilateral carotid wall hematoma, causality of the procedure-related formation of a carotid wall hematoma and oculosympathetic disturbance remains unproven.
The use of balloon angioplasty and stent placement for the treatment of carotid artery stenoses has gained increasing popularity. Over a period of 6 months, we saw several cases of Horner syndrome in patients who had been treated with stent-supported percutaneous angioplasty of the carotid artery (SPAC). Some of these patients did not have a complete Horner syndrome, but rather, pupillary dilatation in absence of eyelid ptosis (ie, partial Horner syndrome). We therefore saw the need for a prospective study of the incidence and natural history of Horner syndrome following SPAC. We assessed the hypothesis that Horner syndrome is related to the postinterventional appearance of an ipsilateral carotid wall hematoma.
Methods
Twenty-eight consecutive patients (four female, 24 male; mean age ± SD, 64.4 years ± 11.3) scheduled to undergo SPAC were enrolled in the study. The patients had a proximal, symptomatic, high-grade (≥70%; mean, 86.3% ± 8.3) internal carotid stenosis, according to the European Carotid Surgery Trial methods of measuring carotid stenosis (1). All carotid stenoses were due to atherosclerotic disease. None of the patients had undergone prior carotid artery interventions.
Pretreatment anticoagulation and antiplatelet medication regimens consisted of daily applications of aspirin (300 mg), clopidogrel (75 mg), and enoxaparin (1 mg per kilogram of body weight) for at least 3 days before SPAC. Immediately before the intervention, the activated clotting time was elevated to a minimum of 250 seconds by means of intravenous application of unfractionated heparin. Endovascular treatment was initiated by navigating a 7F introducer catheter into the common carotid artery. A 0.014-inch, high torque guidewire (Guidant; Guidant Europe S.A., Diegem, Belgium) was navigated through the stenosis into the distal internal carotid artery. A self-expandable stent (Carotid Wallstent, 7.0 × 30 mm; Boston Scientific Corporation, Natick, MA) was deployed inside the stenosis. After stent delivery, the stent catheter was removed. With the guidewire maintained in the distal internal carotid artery, a balloon dilation catheter (Bypass Speedy, 20 mm in length, 5.0–6.0 mm in diameter; Boston Scientific Corporation) was navigated to the residual stenosis inside the stent and was inflated with 7–12 bar for 5 seconds. In all cases, the final angiogram demonstrated complete vessel restoration. No predilation was performed, and no distal protection device was used in any of the cases.
Duplex sonography of the extracranial carotid arteries and perivascular tissue and clinical examination was performed before and 4 hours ± 2, 24 hours ± 2, and 7 days ± 1 after SPAC. Duplex sonography was performed in axial and sagittal planes by using the Sonoline Elegra sonography system and a 7.5L40 linear-array 7.5-MHz transducer (Siemens Medical Systems, Erlangen, Germany). Sonographic criteria for carotid wall hematoma were: 1) an imaging appearance of a homogeneous, echolucent thickening of the vessel wall that was crescent-shaped and spindle-shaped in the axial and sagittal planes, respectively; and 2) its resolution during follow-up sonography. Sonographic evaluations were performed by physicians with explicit expertise in neurovascular sonography (M.R., U.S.). Since vascular stents are visualized by means of duplex sonography, the sonography investigators were not blinded to the side of the carotid intervention. Horner syndrome was assumed to be present in case of miosis with or without eyelid ptosis, as diagnosed by a physician who was blinded to the side of SPAC (W.-D.N.).
The study was approved by the local institutional review board. The Fisher exact test and Mann-Whitney U test were used for statistical analysis, and statistical significance was set at P ≤ .05.
Results
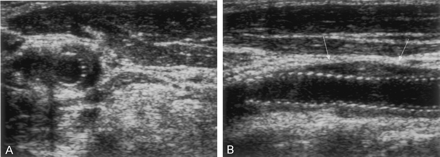
Before SPAC, none of the 28 patients had a carotid wall hematoma, as detected with sonography or an incidence of Horner syndrome. Within 24 hours ± 2 after SPAC, 11 of the 28 patients had developed ipsilateral Horner syndrome (patients 1–11, Table). Ten of these 11 patients had a sonographic appearance of a carotid wall hematoma (Fig 1), whereas only two of the 17 patients without Horner syndrome had this finding (Fisher exact test, P < .001). None of the 28 patients had carotid dissection outside of the stented portion of the vessel, as shown in any of the sonography investigations.
Axial (A) and sagittal (B) duplex sonograms obtained during tissue harmonic imaging of the left internal carotid artery after SPAC in a patient with postinterventional Horner syndrome. The crescent-shaped (A) and respective spindle-shaped (B), homogeneous, echolucent thickening of the vessel wall indicates a carotid wall hematoma.
Characteristics of study patients and the postinterventional appearance of Horner syndrome and carotid wall hematoma
All cases of carotid wall hematoma and 10 of the 11 cases of Horner syndrome had developed within 4 hours ± 2 after SPAC. However, one of the patients with a carotid wall hematoma did not have Horner syndrome during the examination performed 4 hours ± 2 after SPAC; however, it was observed in the examination performed 24 hours ± after SPAC (patient 10, Table). In all patients, both carotid wall hematoma and Horner syndrome had resolved at 7 days ± 1 after SPAC.
The age of the patients with Horner syndrome and of those with carotid wall hematoma was not significantly different from that of the patients who did not develop Horner syndrome and of those without carotid wall hematoma, respectively (Mann-Whitney U Test).
Discussion
In this study, we found ipsilateral Horner syndrome after SPAC in 11 of 28 patients. Horner syndrome was significantly related to a postinterventional appearance of an ipsilateral carotid wall hematoma. In all patients, both Horner syndrome and carotid wall hematoma had resolved within 7 days ± 1 day. Thus, Horner syndrome appears to be a common yet transient event following SPAC. The rather small number of patients in this study, however, limits conclusions regarding the incidence of procedure-related complications.
Before we consider the pathophysiologic mechanisms of Horner syndrome during carotid interventions, we should highlight the sympathetic innervation of the eye. The sympathetic pathway to the eye consists of a three-neuron chain (2). The first-order neuron originates from the posterolateral hypothalamus and descends through the brain stem to reach the second-order neuron located in the intermediolateral cell column of the lower cervical and the upper thoracic cord (C8-T2). The second-order neuron leaves the cord with the spinal nerve roots and ascends along the cervical sympathetic chain to synapse with the third-order neuron located in the superior cervical ganglion. The postganglionic third-order neuron ascends to the cranium as part of the internal carotid plexus and innervates the dilator of the iris and the superior and inferior tarsal muscles. The fibers involved in facial sweating travel along the external carotid artery (2). Interruption of postganglionic sympathetic fibers along the carotid artery may result in ipsilateral oculosympathetic dysfunction with miosis and facultative eyelid ptosis (ie, Horner syndrome).
Horner syndrome is a common sign of spontaneous or traumatic carotid artery dissection (3–5), which poses a risk of ischemic injury to the brain (6, 7). Moreover, carotid dissection associated with Horner syndrome has been reported to complicate cerebrovascular interventions (eg, carotid endarterectomy, cerebral angiography, and carotid artery stent placement) (8–10). Therefore, in the event of Horner syndrome following SPAC, carotid dissection outside of the stented portion of the vessel must be ruled out immediately.
In this study, Horner syndrome following SPAC was significantly related to a postinterventional appearance of an ipsilateral carotid wall hematoma. This observation suggests that the hematoma itself may have caused sympathetic disturbance due to stretching or compression of periarterial sympathetic fibers traveling along the carotid artery. However, two of the 28 patients had a carotid wall hematoma without a sympathetic disturbance, and one of the 11 patients who developed Horner syndrome did not have an imaging appearance of a carotid wall hematoma. Thus, although a causal relation of Horner syndrome and carotid wall hematoma appears to be obvious, both Horner syndrome and carotid wall hematoma could independently result from mechanical stretch injury.
We used self-expandable stents and primary stent placement in all cases. With this technique, the stent is unfolded within the stenosis before angioplasty itself is performed; this process allows dilation of the residual stenosis during stent protection. Thus, potential dissections during and after balloon dilation are fixed to the vessel wall and prevented from obstructing the vessel (11).
A nonobstructive carotid wall hematoma may be diagnosed by means of MR imaging or sonography (12, 13). However, MR imaging cannot be used to visualize a wall hematoma after stent placement because of extensive stent artifacts. In contrast, sonography is not affected by the use of ferromagnetic material, and thus, it is suitable for an examination of stented vessels. Although data about the sonographic diagnosis of carotid wall hematoma with a normal carotid angiogram is limited, complete resolution of echolucent wall thickening within 7 days ± 1 after intervention allowed us to differentiate a wall hematoma from compressed plaque debris.
Conclusion
Horner syndrome appears to be a common yet transient event after SPAC. Although postinterventional Horner syndrome is significantly related to ipsilateral carotid wall hematoma, whether the formation of the hematoma itself is responsible for sympathetic disturbances remains unproven.
References
- Received February 26, 2003.
- Accepted after revision May 11, 2003.
- Copyright © American Society of Neuroradiology