Abstract
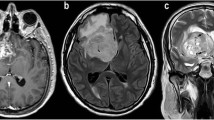
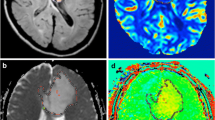
Purpose The purpose of our study was to determine the statistical significance of thresholds of relative cerebral blood volume (rCBV), apparent diffusion coefficient (ADC) and ADC ratios in grading cerebral gliomas. Materials and methods In this retrospective study, 51 patients with histopathologically confirmed primary cerebral gliomas who had undergone conventional MR imaging, dynamic contrast-enhanced T2*-weighted perfusion MR imaging and diffusion MR imaging were included. A retrospective blinded analysis of the imaging findings including the perfusion and diffusion parameters was done. The rCBV measurements were obtained from regions of maximum perfusion. Minimum ADC values were obtained from the region of maximum hypointensity within the tumor and from the corresponding opposite white matter. Tumor grade determined with the two methods were then compared with the histopathologic grade. Mann–Whitney tests were performed to compare the DWI and PWI between tumor types. Receiver operating characteristic analyses were performed to determine optimum thresholds for tumor grading and also to calculate the sensitivity, specificity, PPV, and NPV for identifying high-grade gliomas. Results Statistical analysis demonstrated a threshold value of 2.91 for rCBV to provide sensitivity, specificity, PPV, and NPV of 94.7, 93.75, 90.0, and 96.8%, respectively, in determining high-grade gliomas. An ADC value of 98.50 mm2/s was defined as a threshold below which tumors were classified as high-grade gliomas and a sensitivity, specificity, PPV, and NPV of 90, 87.1, 81.81 and 93.10% respectively, were obtained. Significant differences were noted in the rCBV ratios, ADC and ADC ratios between low- and high-grade gliomas (P < 0.0001). Conclusion Combining PWI and DWI with conventional MR imaging increases the accuracy of pre-operative imaging grading of glial neoplasms. The rCBV measurements had the most superior diagnostic performance in predicting glioma grade. Absolute ADC values or ADC ratios were also helpful in preoperative grading of gliomas. Threshold values can be used in a clinical setting to evaluate tumors preoperatively for histologic grade and provide a means for guiding treatment and predicting postoperative patient outcome.



Similar content being viewed by others
References
Daumas-Duport C, Scheithauer B, O’Fallon J, Kelly P (1988) Grading of astrocytomas: a simple and reproducible method. Cancer 62:2152–2165. doi:10.1002/1097-0142(19881115)62:10<2152::AID-CNCR2820621015>3.0.CO;2-T
Law M, Yang S, Babb JS, Edmond A et al (2004) Comparison of cerebral blood volume and vascular permeability from dynamic susceptibility contrast-enhanced perfusion MR imaging with glioma grade. Am J Neuroradiol 25:746–755
Rees J (2003) Advances in magnetic resonance imaging of brain tumours. Curr Opin Neurol 16:643–650. doi:10.1097/00019052-200312000-00001
Christoforidis GA, Grecula JC, Newton HB et al (2002) Visualization of microvascularity in glioblastoma multiforme with 8-T high-spatial-resolution MR imaging. Am J Neuroradiol 23:1553–1556
Brant-Zawadzki M, Berry I, Osaki L et al (1986) Gd-DTPA in clinical MR of the brain: I. Intraaxial lesions. Am J Roentgenol 147:1223–1230
Brant-Zawadzki M, Badami JP, Mills CM et al (1984) Primary intracranial tumor imaging: a comparison of magnetic resonance and CT. Radiology 150:435–440
Bydder GM, Steiner RE, Young IR et al (1982) Clinical NMR imaging of the brain: 140 cases. Am J Roentgenol 139:215–236
Just M, Thelen M (1988) Tissue characterization with T1, T2, and proton density values: results in 160 patients with brain tumors. Radiology 169:779–785
Felix R, Schorner W, Laniado M et al (1985) Brain tumors MR imaging with gadolinium-DTPA. Radiology 156:681–688
Moller-Hartmann W, Herminghaus S, Krings T et al (2002) Clinical application of proton magnetic resonance spectroscopy in the diagnosis of intracranial mass lesions. Neuroradiology 44:371–381. doi:10.1007/s00234-001-0760-0
Dean BL, Drayer BP, Bird CR et al (1990) Gliomas classification with MR imaging. Radiology 174:411–415
Watanabe M, Tanaka R, Takeda N (1992) Magnetic resonance imaging and histopathology of cerebral gliomas. Neuroradiology 34:463–469. doi:10.1007/BF00598951
Kondziolka D, Lunsford LD, Martinez AJ (1993) Unreliability of contemporary neurodiagnostic imaging in evaluating suspected adult supratentorial (low-grade) astrocytoma. J Neurosurg 79:533–536
Knopp EA, Cha S, Johnson G et al (1999) Glial neoplasms: dynamic contrast-enhanced T2*-weighted MR imaging. Radiology 211:791–798
Kurki T, Lundbom N, Kalimo H, Valtonen S (1995) MR classification of brain gliomas: value of magnetization transfer and conventional imaging. Magn Reson Imaging 13:501–511. doi:10.1016/0730-725X(95)00006-3
Mihara F, Numaguchi Y, Rothman M, Kristt D, Fiandaca M, Swallow L (1995) Non-enhancing supratentorial malignant astrocytomas: MR features and possible mechanisms. Radiat Med 13:11–17
Scott JN, Brasher PM, Sevick RJ, Rewcastle NB, Forsyth PA (2002) How often are nonenhancing supratentorial gliomas malignant? A population study. Neurology 59:947–949
Burger PC, Vogel FS, Green SB et al (1985) Glioblastoma multiforme and anaplastic astrocytoma: pathologic criteria and prognostic implications. Cancer 56:1106–1111. doi:10.1002/1097-0142(19850901)56:5<1106::AID-CNCR2820560525>3.0.CO;2-2
Burger P (1986) Malignant astrocytic neoplasms: classification, pathology, anatomy, and response to therapy. Semin Oncol 13:16–20
Petrella JR, Provenzale JM (2000) MR perfusion imaging of the brain: techniques and applications. Am J Roentgenol 175:207–219
Aronen HJ, Gazit IE, Louis DN et al (1994) Cerebral blood volume maps of gliomas: comparison with tumor grade and histologic findings. Radiology 191:41–51
Bruening R, Kwong KK, Vevea MJ et al (1996) Echo-planar MR determination of relative cerebral blood volume in human brain tumors: T1 versus T2 weighting. Am J Neuroradiol 17:831–840
Sugahara T, Korogi Y, Kochi M et al (1998) Correlation of MR imaging-determined cerebral blood volume maps with histologic and angiographic determination of vascularity of gliomas. Am J Roentgenol 171:1479–1486
Sugahara T, Korogi Y, Shigematsu Y et al (1999) Value of dynamic susceptibility contrast magnetic resonance imaging in the evaluation of intracranial tumors. Top Magn Reson Imaging 10:114–124. doi:10.1097/00002142-199904000-00004
Wong ET, Jackson EF, Hess KR et al (1998) Correlation between dynamic MRI and outcome in patients with malignant gliomas. Neurology 50:777–781
Wong JC, Provenzale JM, Petrella JR (2000) Perfusion MR imaging of brain neoplasms. Am J Roentgenol 174:1147–1157
Cha S, Knopp EA, Johnson G et al (2002) Intracranial mass lesions: dynamic contrast-enhanced susceptibility-weighted echo-planar perfusion MR imaging. Radiology 223:11–29. doi:10.1148/radiol.2231010594
Lev MH, Rosen BR (1999) Clinical applications of intracranial perfusion MR imaging. Neuroimaging Clin N Am 9:309–331
Shin JH, Lee HK, Kwun BD et al (2002) Using relative cerebral blood flow and volume to evaluate the histopathologic grade of cerebral gliomas: preliminary results. Am J Roentgenol 179:783–789
Chenevert TL, McKeever PE, Ross BD (1997) Monitoring early response of experimental brain tumors to therapy using diffusion magnetic resonance imaging. Clin Cancer Res 3:1457–1466
Krabbe K, Gideon P, Wagn P, Hansen U, Thomsen C, Madsen F (1997) MR diffusion imaging of human intracranial tumours. Neuroradiology 39:483–489. doi:10.1007/s002340050450
Filippi CG, Edgar MA, Ulug AM, Prowda JC, Heier LA, Zimmerman RD (2001) Appearance of meningiomas on diffusion-weighted images: correlating diffusion constants with histopathologic findings. Am J Neuroradiol 22:65–72
Stadnik TW, Chaskis C, Michotte A et al (2001) Diffusion-weighted MR imaging of intracerebral masses: comparison with conventional MR imaging and histologic findings. Am J Neuroradiol 22:969–976
Eis M, Els T, Hoehn-Berlage M, Hossmann KA (1994) Quantitative diffusion MR imaging of cerebral tumor and edema. Acta Neurochir Suppl (Wien) 60:344–346
Eis M, Els T, Hoehn-Berlage M (1995) High resolution quantitative relaxation and diffusion MRI on three different experimental brain tumors in rat. Magn Reson Med 34:835–844. doi:10.1002/mrm.1910340608
Els T, Eis M, Hoehn-Berlage M, Hossmann KA (1995) Diffusion-weighted MR imaging of experimental brain tumors in rats. MAGMA 3:13–20. doi:10.1007/BF02426396
Gupta RK, Sinha U, Cloughesy TF, Alger JR (1999) Inverse correlation between choline magnetic resonance spectroscopy signal intensity and the apparent diffusion coefficient in human glioma. Magn Reson Med 41:2–7. doi:10.1002/(SICI)1522-2594(199901)41:1<2::AID-MRM2>3.0.CO;2-Y
Sugahara T, Korogi Y, Kochi M et al (1999) Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 9:53–60. doi:10.1002/(SICI)1522-2586(199901)9:1<53::AID-JMRI7>3.0.CO;2-2
Gupta RK, Cloughesy TF, Sinha U et al (2000) Relationships between choline magnetic resonance spectroscopy, apparent diffusion coefficient and quantitative histopathology in human glioma. J Neurooncol 50:215–226. doi:10.1023/A:1006431120031
Castillo M, Smith JK, Kwock L, Wilber K (2001) Apparent diffusion coefficients in the evaluation of high-grade cerebral gliomas. Am J Neuroradiol 22:60–64
Gauvain KM, McKinstry RC, Mukherjee P et al (2001) Evaluating pediatric brain tumor cellularity with diffusion-tensor imaging. AJR Am J Roentgenol 177:449–454
Nonomura Y, Yasumoto M, Yoshimura R et al (2001) Relationship between bone marrow cellularity and apparent diffusion coefficient. J Magn Reson Imaging 13:757–760. doi:10.1002/jmri.1105
Kono K, Inoue Y, Nakayama K et al (2001) The role of diffusion-weighted imaging in patients with brain tumors. Am J Neuroradiol 22:1081–1088
Guo AC, Cummings TJ, Dash RC, Provenzale JM (2002) Lymphomas and high-grade astrocytomas: comparison of water diffusibility and histologic characteristics. Radiology 224:177–183. doi:10.1148/radiol.2241010637
Tien RD, Felsberg GJ, Friedman H, Brown M, MacFall J (1994) MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echoplanar pulse sequences. Am J Roentgenol 162:671–677
Fan G, Zang P, Jing F, Wu Z, Guo Q (2005) Usefulness of diffusion/perfusion-weighted MRI in rat gliomas: correlation with histopathology. Acad Radiol 12:640–651. doi:10.1016/j.acra.2005.01.024
Fan G, Deng QL, Wu ZH et al (2006) Usefulness of diffusion/perfusion-weighted MRI in patients with non-enhancing supratentorial brain gliomas: a valuable tool to predict tumour grading? Br J Radiol 79:652–658. doi:10.1259/bjr/25349497
Catalaa I, Henry R, Dillon WP, Graves EE et al (2006) Perfusion, diffusion and spectroscopy values in newly diagnosed cerebral gliomas. NMR Biomed 19(4):463–475. doi:10.1002/nbm.1059
Ringertz J (1950) Grading of gliomas. Acta Pathol Microbiol Scand 27:51–64
Batra A, Tripathi RP, Singh AK (2004) Perfusion magnetic resonance imaging and magnetic resonance spectroscopy of cerebral gliomas showing imperceptible contrast enhancement on conventional magnetic resonance imaging. Australas Radiol 48:324–332. doi:10.1111/j.0004-8461.2004.01315.x
Maia AC Jr, Malheiros SM, da Rocha AJ et al (2005) MR cerebral blood volume maps correlated with vascular endothelial growth factor expression and tumor grade in nonenhancing gliomas. Am J Neuroradiol 26:777–783
Peters AM (1998) Fundamentals of tracer kinetics for radiologists. Br J Radiol 71:1116–1129
Guyton AC (1991) Cerebral blood flow, the cerebrospinal fluid, and brain metabolism. Basic neuroscience: anatomy and physiology, 2nd edn. Saunders, Philadelphia, pp 285–287
Roberts HC, Roberts TP, Brasch RC, Dillon WP (2000) Quantitative measurement of microvascular permeability in human brain tumors achieved using dynamic contrast-enhanced MR imaging: correlation with histologic grade. Am J Neuroradiol 21:891–899
Law M, Yang S et al (2003) Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. Am J Neuroradiol 24:1989–1998
Lev MH, Ozsunar Y, Henson JW et al (2004) Glial tumor grading and outcome prediction using dynamic spin-echo MR susceptibility mapping compared with conventional contrast-enhanced MR: confounding effect of elevated rCBV of oligodendrogliomas. Am J Neuroradiol 25:214–221. Corrected, published erratum appears in Am J Neuroradiol 2004;25:B1
Bulakbasi N, Kocaoglu M, Örs F (2003) Combination of single-voxel proton MR spectroscopy and apparent diffusion coefficient calculation in the evaluation of common brain tumors. Am J Neuroradiol 24:225–233
Author information
Authors and Affiliations
Corresponding author
Additional information
This article, published in Volume 94, Issue 1, pages 87-96, DOI 10.1007/s11060-009-9807-6, has been retracted, as it contains portions of other authors' writings on the same topic in other publications, without sufficient attribution to these earlier works being given. The principal authors of the paper acknowledged that text from background sources was mistakenly used without proper reference to the original source.
A retraction note to this article is available at http://dx.doi.org/10.1007/s11060-013-1205-4.
An erratum to this article is available at http://dx.doi.org/10.1007/s11060-013-1205-4.
About this article
Cite this article
Arvinda, H.R., Kesavadas, C., Sarma, P.S. et al. RETRACTED ARTICLE: Glioma grading: sensitivity, specificity, positive and negative predictive values of diffusion and perfusion imaging. J Neurooncol 94, 87–96 (2009). https://doi.org/10.1007/s11060-009-9807-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-009-9807-6