Summary
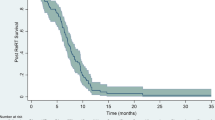
Twenty patients with malignant supratentorial gliomas progressing after radiation therapy and chemotherapy with nitrosoureas received intravenous carboplatin, 450 mg/m2. Courses of therapy were repeated every four weeks. Therapeutic evaluation was performed monthly using neurologic examination and CT scan of the brain. Of 19 patients evaluable for response, 2 (10%) responded to therapy and 6 (30%) had stable disease. The estimated median time to tumor progression for responding and stable patients was 6 months. Median duration of survival was 6 months for all patients. Of 20 patients evaluable for toxicity, none had renal or auditory toxicity. Side effects consisted of hematologic toxicity in 4 patients (20%): one patient had grade 4 toxicity requiring discontinuation of carboplatin and 3 patients had grade 2–3 toxicity.
Similar content being viewed by others
Abbreviations
- HECNU:
-
1-(2-chloroethyl)-1-nitroso-3-(2-hydroxyethyl) urea
References
E.O.R.T.C. Brain tumor group: Effect of CCNU on survival rate of objective remission and duration of free interval in patients with malignant brain glioma-final evaluation. Eur J Cancer 14: 851–856, 1978
Aida T, Bodell WJ: Cellular resistance to chloroethylnitrosoureas, nitrogen mustard and cis-diamminedichloroplatinum II) in human gial-derived cell lines. Cancer Res 47: 1361–1366, 1987
Feun LG, Wallace S, Stewart DJ, Chuang VP, Yung WKA, Leavens ME, Burgess MA, Savaraj N, Benjamin RS, Young SE, Tang RA, Handel S, Mavligit G, Fields WS: Intracarotid infusion of cis-diamminedichloroplatinum in the treatment of recurrent malignant brain tumors. Cancer 54: 794–799, 1984
Walker RW, Allen JC: Treatment of recurrent primary intracranial childhood tumors with cis-diamminedichloroplatinum. Ann Neurol 14: 371, 1983
Canetta R, Rozencweig, Carter SK: Carboplatin: the clinical spectrum to date. Cancer Treat Rev 12: 125–136, 1985
Takahaski H, Sasaki Y, Saijo N, Sakurai M, Nakano H, Nagakawa K, Hoshi A, Jett JR, Hong NS: In vitro colony inhibition of carboplatin against stomach and lung cancer cell lines in comparison with cisplatin. Cancer Chemother Pharmacol 19: 197–200, 1987
Rose WC, Schurig JE: Preclinical antitumor and toxicologic profile of carboplatin. Cancer Treat Rev 12: 1–19, 1985
Boven E, Van der Vijgh WJF, Nauta MM, Schlüper HMM, Pinedo HM: Comparative activity and distribution studies of five platinum analogues in nude mice bearing human ovarian carcinome xenografts. Cancer Res 45: 86–90, 1985
Koeller JM, Trump DL, Tutsch KD, Earhart RH, Davis TE, Tormey DC: Phase I clinical trial and pharmacokinetics of carboplatin (NSC 241240) by single monthly 30-minute infusion. Cancer 57: 222–225, 1986
Allen JC, Walker R, Lucks E, Jennings M, Barfoot S, Tan C: Carboplatin and recurrent childhood brain tumors. J Clinic Oncol 5: 459–463, 1987
Walker RW, Dantis E, Shapiro WR: Treatment of recurrent glioma with carboplatin (CBDCA). Proc Am Soc Clin Oncol 6: 72, 1987
Follezou JY, Fauchon F, Chiras J: Intraarterial infusion of carboplatin in the treatment of malignant gliomas: A phase II study. Neoplasma 36: 349–352, 1989
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Poisson, M., Péréon, Y., Chiras, J. et al. Treatment of recurrent malignant supratentorial gliomas with carboplatin (CBDCA). J Neuro-Oncol 10, 139–144 (1991). https://doi.org/10.1007/BF00146875
Issue Date:
DOI: https://doi.org/10.1007/BF00146875