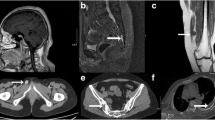
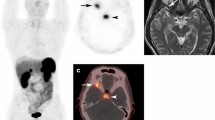
Abstract
Objective
To examine the CT and MR imaging features of phosphaturic mesenchymal tumors (PMTs).
Materials and methods
With IRB approval, our institutional radiology/pathology database was reviewed for pathologically-proven PMTs. CT and MRI examinations were reviewed in consensus noting several imaging features, and if available, comparative molecular imaging tests were analyzed.
Results
We identified 39 patients (21 male, 18 females) with 40 PMTs [mean age, 52.9 ± 14.9 years (range, 14–78)], including 20 bone and 20 soft tissue lesions. Mean maximal lesion diameter was 3.4 ± 2.0 cm (range, 1.1–9.8). 12/18 primary bone lesions (66.6%) were osteolytic and 15/20 (75.0%) had a narrow zone of transition. Internal matrix was present in 18/32 (56.3%) lesions. PMTs were most commonly T1 isointense (31/37, 83.8%), T2 hyperintense (14/36, 38.9%), and solidly enhancing (21/30, 70.0%). The majority (32/36, 88.9%) contained areas of dark T2 signal. 8/9 PMTs were positive by 99mTc-sestamibi scintigraphy, 2/4 by 111In-pentetreotide scintigraphy, 2/2 by 68Ga-DOTATATE PET/CT and 11/13 by 18F-FDG PET/CT. On FDG PET/CT, the mean SUVmax was 4.1 ± 2.5 (range, 1.5–10.8).
Conclusions
Osseous PMTs are commonly osteolytic with a narrow zone of transition. Both bone and soft tissue PMTs often contain matrix and areas of dark T2 signal on MRI, independent of the presence of matrix. However, PMTs may mimic other bone and soft tissue neoplasms, including fibrous dysplasia, tenosynovial giant cell tumor, and even atypical lipomatous tumor. As such, clinical presentation and laboratory correlation are critical to PMT recognition and accurate diagnosis.










Similar content being viewed by others
References
Kumar R, Folpe AL, Mullan BP. Tumor-induced osteomalacia. Transl Endocrinol Metab 2015;7(3):1871.
McCance RA. Osteomalacia with Looser's nodes (Milkman's syndrome) due to a raised resistance to vitamin D acquired about the age of 15 years. Q J Med. 1947;16:33–46.
Prader A, Illig R, Uehlinger RE, Stalder G. Rachitis infolge knochentumors (rickets caused by bone tumors). Helv Pediatr Acta. 1959;14:554–65.
Folpe AL, Fanburg-Smith JC, Billings SD, Bisceglia M, Bertoni F, Cho JY, et al. Most osteomalacia-associated mesenchymal tumors are a single histopathologic entity: an analysis of 32 cases and a comprehensive review of the literature. Am J Surg Pathol. 2004;28(1):1–30.
Sundaram M, McCarthy EF. Oncogenic osteomalacia. Skelet Radiol. 2000;29(3):117–24.
Evans DJ, Azzopardi JG. Distinctive tumours of bone and soft tissue causing acquired vitamin-D-resistant osteomalacia. Lancet. 1972;1(7746):353–4.
Weidner N, Santa Cruz D. Phosphaturic mesenchymal tumors. A polymorphous group causing osteomalacia or rickets. Cancer. 1987;59(8):1442–54.
CDM F. WHO classification of tumours of soft tissue and bone. Lyon: IARC Press; 2013.
Gardner KH, Shon W, Folpe AL, Wieland CN, Tebben PJ, Baum CL. Tumor-induced osteomalacia resulting from primary cutaneous phosphaturic mesenchymal tumor: a case and review of the medical literature. J Cutan Pathol. 2013;40(9):780–4. quiz 779
Woo VL, Landesberg R, Imel EA, Singer SR, Folpe AL, Econs MJ, et al. Phosphaturic mesenchymal tumor, mixed connective tissue variant, of the mandible: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(6):925–32.
Agrawal K, Bhadada S, Mittal BR, Shukla J, Sood A, Bhattacharya A, et al. Comparison of 18F-FDG and 68Ga DOTATATE PET/CT in localization of tumor causing oncogenic osteomalacia. Clin Nucl Med. 2015;40(1):e6–e10.
Clifton-Bligh RJ, Hofman MS, Duncan E, Sim Ie W, Darnell D, Clarkson A, et al. Improving diagnosis of tumor-induced osteomalacia with Gallium-68 DOTATATE PET/CT. J Clin Endocrinol Metab. 2013;98(2):687–94.
Ho CL. Ga68-DOTA peptide PET/CT to detect occult mesenchymal tumor-inducing osteomalacia: a case series of three patients. Nucl Med Mol Imaging. 2015;49(3):231–6.
Jadhav S, Kasaliwal R, Lele V, Rangarajan V, Chandra P, Shah H, et al. Functional imaging in primary tumour-induced osteomalacia: relative performance of FDG PET/CT vs somatostatin receptor-based functional scans: a series of nine patients. Clin Endocrinol. 2014;81(1):31–7.
Zhang J, Zhu Z, Zhong D, Dang Y, Xing H, Du Y, et al. 68Ga DOTATATE PET/CT is an accurate imaging modality in the detection of culprit tumors causing osteomalacia. Clin Nucl Med. 2015;40(8):642–6.
Duet M, Kerkeni S, Sfar R, Bazille C, Liote F, Orcel P. Clinical impact of somatostatin receptor scintigraphy in the management of tumor-induced osteomalacia. Clin Nucl Med. 2008;33(11):752–6.
Garcia CA, Spencer RP. Bone and In-111 octreotide imaging in oncogenic osteomalacia: a case report. Clin Nucl Med. 2002;27(8):582–3.
Jan de Beur SM, Streeten EA, Civelek AC, McCarthy EF, Uribe L, Marx SJ, et al. Localisation of mesenchymal tumours by somatostatin receptor imaging. Lancet. 2002;359(9308):761–3.
Nguyen BD, Wang EA. Indium-111 pentetreotide scintigraphy of mesenchymal tumor with oncogenic osteomalacia. Clin Nucl Med. 1999;24(2):130–1.
Rhee Y, Lee JD, Shin KH, Lee HC, Huh KB, Lim SK. Oncogenic osteomalacia associated with mesenchymal tumour detected by indium-111 octreotide scintigraphy. Clin Endocrinol. 2001;54(4):551–4.
Chong WH, Andreopoulou P, Chen CC, Reynolds J, Guthrie L, Kelly M, et al. Tumor localization and biochemical response to cure in tumor-induced osteomalacia. J Bone Miner Res. 2013;28(6):1386–98.
Hodgson SF, Clarke BL, Tebben PJ, Mullan BP, Cooney WP 3rd, Shives TC. Oncogenic osteomalacia: localization of underlying peripheral mesenchymal tumors with use of Tc 99m sestamibi scintigraphy. Endocr Pract. 2006;12(1):35–42.
Andreopoulou P, Dumitrescu CE, Kelly MH, Brillante BA, Cutler Peck CM, Wodajo FM, et al. Selective venous catheterization for the localization of phosphaturic mesenchymal tumors. J Bone Miner Res. 2011;26(6):1295–302.
Dupond JL, Mahammedi H, Prie D, Collin F, Gil H, Blagosklonov O, et al. Oncogenic osteomalacia: diagnostic importance of fibroblast growth factor 23 and F-18 fluorodeoxyglucose PET/CT scan for the diagnosis and follow-up in one case. Bone. 2005;36(3):375–8.
Jiang Y, Xia WB, Xing XP, Silva BC, Li M, Wang O, et al. Tumor-induced osteomalacia: an important cause of adult-onset hypophosphatemic osteomalacia in China: report of 39 cases and review of the literature. J Bone Miner Res. 2012;27(9):1967–75.
Koplas MC, Rubin BP, Sundaram M. Phosphaturic mesenchymal tumor: two contrasting cases. Skelet Radiol. 2014;43(6):841–5.
Nakanishi K, Sakai M, Tanaka H, Tsuboi H, Hashimoto J, Hashimoto N, et al. Whole-body MR imaging in detecting phosphaturic mesenchymal tumor (PMT) in tumor-induced hypophosphatemic osteomalacia. Magn Reson Med Sci. 2013;12(1):47–52.
Avila NA, Skarulis M, Rubino DM, Doppman JL. Oncogenic osteomalacia: lesion detection by MR skeletal survey. AJR Am J Roentgenol. 1996;167(2):343–5.
Nelson AE, Mason RS, Robinson BG, Hogan JJ, Martin EA, Ahlstrom H, et al. Diagnosis of a patient with oncogenic osteomalacia using a phosphate uptake bioassay of serum and magnetic resonance imaging. Eur J Endocrinol. 2001;145(4):469–76.
Seo HJ, Choi YJ, Kim HJ, Jeong YH, Cho A, Lee JH, et al. Using (18)F-FDG PET/CT to detect an occult mesenchymal tumor causing oncogenic osteomalacia. Nucl Med Mol Imaging. 2011;45(3):233–7.
Dissanayake AM, Wilson JL, Holdaway IM, Reid IR. Oncogenic osteomalacia: culprit tumour detection whole-body magnetic resonance imaging. Intern Med J. 2003;33(12):615–6.
Bahrami A, Weiss SW, Montgomery E, Horvai AE, Jin L, Inwards CY, et al. RT-PCR analysis for FGF23 using paraffin sections in the diagnosis of phosphaturic mesenchymal tumors with and without known tumor induced osteomalacia. Am J Surg Pathol. 2009;33(9):1348–54.
Flanagan AM, Delaney D, O'Donnell P. Benefits of molecular pathology in the diagnosis of musculoskeletal disease: part II of a two-part review: bone tumors and metabolic disorders. Skelet Radiol. 2010;39(3):213–24.
Slot-Steenks MM, Hamdy NA, van de Sande MA, Vriens D, Cleven AH, Appelman-Dijkstra NM. Identifying the culprit lesion in tumor induced hypophosphatemia, the solution of a clinical enigma. Endocrine. 2016;54(3):642–7.
Agaimy A, Michal M, Chiosea S, Petersson F, Hadravsky L, Kristiansen G, et al. Phosphaturic mesenchymal tumors: clinicopathologic, immunohistochemical and molecular analysis of 22 cases expanding their morphologic and immunophenotypic spectrum. Am J Surg Pathol. 2017;41(10):1371–80.
Houang M, Clarkson A, Sioson L, Elston MS, Clifton-Bligh RJ, Dray M, et al. Phosphaturic mesenchymal tumors show positive staining for somatostatin receptor 2A (SSTR2A). Hum Pathol. 2013;44(12):2711–8.
Breer S, Brunkhorst T, Beil FT, Peldschus K, Heiland M, Klutmann S, et al. 68Ga DOTA-TATE PET/CT allows tumor localization in patients with tumor-induced osteomalacia but negative 111In-octreotide SPECT/CT. Bone. 2014;64:222–7.
Jagtap VS, Sarathi V, Lila AR, Malhotra G, Sankhe SS, Bandgar T, et al. Tumor-induced osteomalacia: a single center experience. Endocr Pract. 2011;17(2):177–84.
Sent-Doux KN, Mackinnon C, Lee JC, Folpe AL, Habeeb O. Phosphaturic mesenchymal tumor without osteomalacia: additional confirmation of the “non-phosphaturic” variant, with emphasis on the roles of FGF23 chromogenic in situ hybridization and FN1-FGFR1 fluorescence in situ hybridization. Hum Pathol. 2018. https://doi.org/10.1016/j.humpath.2018.02.022.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
The need for informed consent was waived by the Institutional Review Board.
Rights and permissions
About this article
Cite this article
Broski, S.M., Folpe, A.L. & Wenger, D.E. Imaging features of phosphaturic mesenchymal tumors. Skeletal Radiol 48, 119–127 (2019). https://doi.org/10.1007/s00256-018-3014-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-018-3014-5