Abstract
Objectives
To compare intraindividually two macrocyclic contrast agents - gadobutrol and gadoterate meglumine (Gd-DOTA) - for dynamic and quantitative assessment of relative enhancement (RE) in benign and malignant breast lesions.
Methods
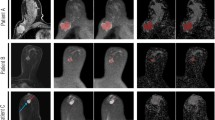
This was an ethically approved, prospective, single-centre, randomized, crossover study in 52 women with suspected breast lesions referred for magnetic resonance imaging (MRI). Each patient underwent one examination with gadobutrol and one with Gd-DOTA (0.1 mmol/kg BW) on a 1.5 T system 1 – 7 days apart. Dynamic, T1-weighted, 3D gradient echo sequences were acquired under identical conditions. Quantitative evaluation with at least three regions of interest (ROI) per lesion was performed. Primary endpoint was RE during the initial postcontrast phase after the first and second dynamic acquisition, and peak RE. All lesions were histologically proven; differences between the examinations were evaluated.
Results
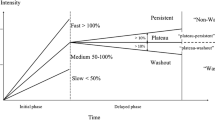
Forty-five patients with a total of 11 benign and 34 malignant lesions were assessed. Mean RE was significantly higher for gadobutrol than Gd-DOTA (p < 0.0001). Gadobutrol showed significantly less washout (64.4 %) than Gd-DOTA (75.4 %) in malignant lesions (p = 0.048)
Conclusions
Gadobutrol has higher RE values compared with Gd-DOTA, whereas Gd-DOTA shows more marked washout in malignant lesions. This might improve the detection of breast lesions and influence the specificity of breast MRI-imaging.
Key Points
• Contrast agents differ in terms of peak enhancement and postinitial contrast behaviour.
• Gadobutrol results in greater peak RE in lesions, likely due to relaxivity.
• Gd-DOTA shows more pronounced washout in postinitial contrast behaviour of malignant lesions.
• Further investigations of the differences among GD-CM may improve sensitivity and specificity.




Similar content being viewed by others
References
Kuhl CK, Mielcareck P, Klaschik S et al (1999) Dynamic breast MR imaging: are signal intensity time course data useful for differential diagnosis of enhancing lesions? Radiology 211:101–110
Montemurro F, Martincich L, Sarotto I et al (2007) Relationship between DCE-MRI morphological and functional features and histopathological characteristics of breast cancer. Eur Radiol 17:1490–1497
Morris EA, Harms S (2004) ACR practice guideline for the performance of magnetic resonance imaging (MRI) of the breast. American College of Radiology, Reston (VA)
Heywang-Kobrunner SH, Viehweg P, Heinig A, Kuchler CH (1997) Contrast-enhanced MRI of the breast: accuracy, value, controversies, solutions. Eur J Radiol 24:94–108
Lehman CD, Isaacs C, Schnall MD et al (2007) Cancer yield of mammography, MR, and US in high-risk women: prospective multi-institution breast cancer screening study. Radiology 244:381–388
Saslow D, Boetes C, Burke W et al (2007) American Cancer Society Guidelines for Breast Screening with MRI as an Adjunct to Mammography. CA Cancer J Clin 57:75–89
Schelfout K, Van Goethem M, Kersschot E et al (2004) Preoperative breast MRI in patients with invasive lobular breast cancer. Eur Radiol 14:1209–1216
Van Goethem M, Schelfout K, Dijckmans L et al (2004) MR mammography in the pre-operative staging of breast cancer in patients with dense breast tissue: comparison with mammography and ultrasound. Eur Radiol 14:809–816
Mann RM, Kuhl CK, Kinkel K, Boetes C (2008) Breast MRI: guidelines from the European Society of Breast Imaging. Eur Radiol 18:1307–1318
Flickinger FW, Allison JD, Sherry RM, Wright JC (1993) Differentiation of benign from malignant breast masses by time-intensity evaluation of contrast enhanced MRI. Magn Reson Imaging 11:617–620
Heywang SH, Hahn D, Schmidt H et al (1986) MR imaging of the breast using gadolinium-DTPA. J Comput Assist Tomogr 10:199–204
Kaiser WA, Zeitler E (1989) MR imaging of the breast: fast imaging sequences with and without Gd-DTPA. Preliminary Observations. Radiology 170:681–686
Stack JP, Redmond OM, Codd MB, Dervan PA, Ennis JT (1990) Breast disease: tissue characterization with Gd-DTPA enhancement profiles. Radiology 174:491–494
Weinmann HJ, Laniado M, Mutzel W (1984) Pharmacokinetics of GdDTPA/dimeglumine after intravenous injection into healthy volunteers. Physiol Chem Phys Med NMR 16:167–172
Allard M, Doucet D, Kien P, Bonnemain B, Caille JM (1988) Experimental study of DOTA-gadolinium. Pharmacokinetics and pharmacologic properties. Investig Radiol 23:S271–S274
Le Mignon MM, Chambon C, Warrington S, Davies R, Bonnemain B (1990) Gd-DOTA. Pharmacokinetics and tolerability after intravenous injection into healthy volunteers. Investig Radiol 25:933–937
Bourasset F, Dencausse A, Bourrinet P, Ducret M, Corot C (2001) Comparison of plasma and peritoneal concentrations of various categories of MRI blood pool agents in a murine experimental pharmacokinetic model. MAGMA 12:82–87
Pintaske J, Martirosian P, Graf H et al (2006) Relaxivity of Gadopentetate Dimeglumine (Magnevist), Gadobutrol (Gadovist), and Gadobenate Dimeglumine (MultiHance) in human blood plasma at 0.2, 1.5, and 3 Tesla. Investig Radiol 41:213–221
Rohrer M, Bauer H, Mintorovitch J, Requardt M, Weinmann H-J (2005) Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Investig Radiol 40:715–724
Attenberger UI, Runge VM, Morelli JN, Williams J, Jackson CB, Michaely HJ (2010) Evaluation of gadobutrol, a macrocyclic, nonionic gadolinium chelate in a brain glioma model: comparison with gadoterate meglumine and gadopentetate dimeglumine at 1.5T, combined with an assessment of field strength dependence, specifically 1.5 versus 3T. J Magn Reson Imaging 31:549–555
Knopp MV, Bourne MW, Sardanelli F et al (2003) Gadobenate dimeglumine-enhanced MRI of the breast: analysis of dose response and comparison with gadopentetate dimeglumine. AJR Am J Roentgenol 181:663–676
Martincich L, Faivre-Pierret M, Zechmann CM et al (2011) Multicenter, double-blind, randomized, intraindividual crossover comparison of gadobenate dimeglumine and gadopentetate dimeglumine for Breast MR imaging (DETECT Trial). Radiology 258:396–408
Pediconi F, Catalano C, Padula S et al (2008) Contrast-enhanced MR mammography: improved lesion detection and differentiation with gadobenate dimeglumine. AJR Am J Roentgenol 191:1339–1346
Gillis A, Gray M, Burstein D (2002) Relaxivity and diffusion of gadolinium agents in cartilage. Magn Reson Med 48:1068–1071
Wiener E, Woertler K, Weirich G, Rummeny EJ, Settles M (2007) Contrast enhanced cartilage imaging: Comparison of ionic and non-ionic contrast agents. Eur J Radiol 63:110–119
Marotta M, D'Armiento FP, Martino G et al (1985) Glycosaminoglycans in human breast cancer: morphological and biochemical study. Appl Pathol 3:164–169
Olsen EB, Trier K, Eldov K, Ammitzboll T (1988) Glycosaminoglycans in human breast cancer. Acta Obstet Gynecol Scand 67:539–542
Takeuchi JSM, Sato E, Shamoto M, Miura K, Nakagaki S (1976) Variation in Glycosaminoglycan Components of Breast Tumors. Cancer Res 36:2133–2139
van der Molen AJ, Bellin MF (2008) Extracellular gadolinium-based contrast media: differences in diagnostic efficacy. Eur J Radiol 66:168–174
Renz DM, Durmus T, Bottcher J et al (2014) Comparison of Gadoteric Acid and Gadobutrol for Detection as Well as Morphologic and Dynamic Characterization of Lesions on Breast Dynamic Contrast-Enhanced Magnetic Resonance Imaging. Investig Radiol. doi:10.1097/RLI.0000000000000039
Mussurakis S, Buckley DL, Coady AM, Turnbull LW, Horsman A (1996) Observer variability in the interpretation of contrast enhanced MRI of the breast. Br J Radiol 69:1009–1016
Committee ACoRB-R, Radiology ACo (2003) Breast imaging reporting and data system. American College of Radiology
Li SP, Padhani AR, Taylor NJ et al (2011) Vascular characterisation of triple negative breast carcinomas using dynamic MRI. Eur Radiol 21:1364–1373
Veltman J, Mann R, Kok T et al (2008) Breast tumor characteristics of BRCA1 and BRCA2 gene mutation carriers on MRI. Eur Radiol 18:931–938
Schmitz AC, Peters NH, Veldhuis WB et al (2008) Contrast-enhanced 3.0-T breast MRI for characterization of breast lesions: increased specificity by using vascular maps. Eur Radiol 18:355–364
Pediconi F, Catalano C, Occhiato R et al (2005) Breast lesion detection and characterization at contrast-enhanced MR mammography: gadobenate dimeglumine versus gadopentetate dimeglumine. Radiology 237:45–56
Pediconi F, Kubik-Huch R, Chilla B, Schwenke C, Kinkel K (2013) Intra-individual randomised comparison of gadobutrol 1.0 M versus gadobenate dimeglumine 0.5 M in patients scheduled for preoperative breast MRI. Eur Radiol 23:84–92
Anzalone N, Gerevini S, Scotti R, Vezzulli P, Picozzi P (2009) Detection of cerebral metastases on magnetic resonance imaging: intraindividual comparison of gadobutrol with gadopentetate dimeglumine. Acta Radiol 50:933–940
Ibrahim MA, Haughton VM, Hyde JS (1995) Effect of disk maturation on diffusion of low-molecular-weight gadolinium complexes: an experimental study in rabbits. AJNR Am J Neuroradiol 16:1307–1311
Jansen SA, Shimauchi A, Zak L et al (2009) Kinetic curves of malignant lesions are not consistent across MRI systems: need for improved standardization of breast dynamic contrast-enhanced MRI acquisition. AJR Am J Roentgenol 193:832–839
Pabst T, Kenn W, Kaiser WA, Hahn D (2001) Understanding why contrast enhancement in dynamic MRI is not reproducible: illustration with a simple phantom. Breast J 7:166–170
Sardanelli F, Fausto A, Esseridou A, Di Leo G, Kirchin MA (2008) Gadobenate dimeglumine as a contrast agent for dynamic breast magnetic resonance imaging: effect of higher initial enhancement thresholds on diagnostic performance. Investig Radiol 43:236–242
Prince MR, Zhang HL, Roditi GH, Leiner T, Kucharczyk W (2009) Risk factors for NSF: a literature review. J Magn Reson Imaging 30:1298–1308
Sardanelli F, Boetes C, Borisch B et al (2010) Magnetic resonance imaging of the breast: recommendations from the EUSOMA working group. Eur J Cancer 46:1296–1316
Acknowledgments
The scientific guarantor of this publication is Prof. Bernd Hamm. The authors of this manuscript declare relationships with the following companies: Prof. B. Hamm nearly all medical companies. Dr. A. Huppertz, Bayer Healthcare, Siemens Healthcare. Dr. E.M. Fallenberg, travel support and speaker’s fee by Bayer Healthcare, Guerbet, Siemens Healthcare, GE Healthcare. This study has received funding from Bayer Healthcare and Siemens Healthcare. The investigators had exclusive control of all data, manu- script drafting, and submission of this study. One of the authors has significant statistical expertise. Institutional review board approval was not required because the study was approved by the Federal Institute for Drugs and Medical Products and the local ethics committee (Clinical study phase: IIIb, EudraCT No.: 2009-013432-20). Written informed consent was obtained from all subjects (patients) in this study. Some study subjects or cohorts have been previously reported in Investigative Radiology. Renz DM, Durmus T, Bottcher J, Taupitz M, Diekmann F, Huppertz A, Pfeil A, Maurer MH, Streitparth F, Bick U, Hamm B, Fallenberg EM (2014) Comparison of Gadoteric Acid and Gadobutrol for Detection as Well as Morphologic and Dynamic Characterization of Lesions on Breast Dynamic Contrast-Enhanced Magnetic Resonance Imaging. Investigative Radiology. Methodology: prospective, randomised controlled trial, performed at one institution. We are grateful to Nikola Bangemann, MD, Tahir Durmus, MD, Elke Keil, MD, Christiane Richter-Ehrenstein, MD, Achim Schneider, MD and Klaus- Jürgen Winzer MD for their contribution in the patient recruitment and inclusion. We are thankful to Bettina Herwig for editorial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fallenberg, E.M., Renz, D.M., Karle, B. et al. Intraindividual, randomized comparison of the macrocyclic contrast agents gadobutrol and gadoterate meglumine in breast magnetic resonance imaging. Eur Radiol 25, 837–849 (2015). https://doi.org/10.1007/s00330-014-3426-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3426-0