Abstract
Purpose
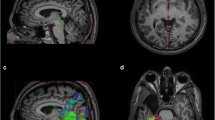
We evaluated optic radiation (OR) damage in patients with glaucoma by using fractional anisotropy (FA) of diffusion-tensor magnetic resonance imaging.
Methods
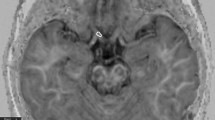
We studied 29 patients with glaucoma and 19 healthy controls. Regions of interest were placed over the bilateral anterior and posterior ORs on the FA maps, and the FA value of each region was measured.
Results
FA values of the bilateral anterior and posterior ORs were significantly lower in patients with glaucoma (anterior right, P = 0.0002; anterior left, P = 0.00028; posterior right, P = 0.0004; and posterior left, P = 0.0001) than in the healthy controls. In glaucoma patients, significant correlations were observed between the FA values and the average of the total deviation of the contralateral hemifields of both eyes (left hemifield and anterior right OR, correlation coefficient [r] = 0.46 [P = 0.013]; right hemifield and anterior left OR, r = 0.43 [P = 0.021]; left hemifield and posterior right OR, r = 0.54 [P = 0.0027]; and right hemifield and posterior left OR, r = 0.46 [P = 0.012]).
Conclusions
FA values of the entire OR were decreased in glaucoma patients and correlated with the degree of visual field defect.


Similar content being viewed by others
References
Quigley HA. Neuronal death in glaucoma. Prog Retin Eye Res. 1999;18:39–57.
Gupta N, Yücel YH. Glaucoma as a neurodegenerative disease. Curr Opin Ophthalmol. 2007;18:110–4.
Yücel YH, Zhang Q, Gupta N, Kaufman PL, Weinreb RN. Loss of neurons in magnocellular and parvocellular layers of the LGN in glaucoma. Arch Ophthalmol. 2000;118:378–84.
Qing G, Zhang S, Wang B, Wang N. Functional MRI signal changes in primary visual cortex corresponding to the central normal visual field of patients with primary open-angle glaucoma. Invest Ophthalmol Vis Sci. 2010;51:4627–34.
Garaci FG, Bolacchi F, Cerulli A, Melis M, Spanò A, Cedrone C, et al. Optic nerve and optic radiation neurodegeneration in patients with glaucoma: in vivo analysis with 3-T diffusion-tensor MR imaging. Radiology. 2009;252:496–501.
Engelhorn T, Michelson G, Waerntges S, Struffert T, Haider S, Doerfler A. Diffusion tensor imaging detects rarefaction of optic radiation in glaucoma patients. Acad Radiol. 2011;18:764–9.
Engelhorn T, Michelson G, Waerntges S, Otto M, El-Rafei A, Struffert T, et al. Changes of radial diffusivity and fractional anisotropy in the optic nerve and optic radiation of glaucoma patients. Sci World J. 2012;2012:849632.
Li C, Ling X, Liu S, Xu A, Zhang Y, Xing S, et al. Early detection of secondary damage in ipsilateral thalamus after acute infarction at unilateral corona radiata by diffusion tensor imaging and magnetic resonance spectroscopy. BMC Neurol. 2011;11:49.
Chang CC, Lui CC, Wang JJ, Huang SH, Lu CH, Chen C, et al. Multi-parametric neuroimaging evaluation of cerebrotendinous xanthomatosis and its correlation with neuropsychological presentations. BMC Neurol. 2010;10:59.
Li M, Li J, He H, Wang Z, Lv B, Li W, et al. Directional diffusivity changes in the optic nerve and optic radiation in optic neuritis. Br J Radiol. 2011;84:304–14.
Wu TC, Wilde EA, Bigler ED, Li X, Merkley TL, Yallampalli R, et al. Longitudinal changes in the corpus callosum following pediatric traumatic brain injury. Dev Neurosci. 2010;32:361–73.
Foster PJ, Buhrmann R, Quigley HA, Johnson GJ. The definition and classification of glaucoma in prevalence surveys. Br J Ophthalmol. 2002;86:238–42.
Anderson DR, Patella VM. Interpretation of single field. In: Anderson DR, Patella VM, editors. Automated static perimetry. 2nd ed. St. Louis: Mosby; 1999. p. 121–90.
Giorgio A, Santelli L, Tomassini V, Bosnell R, Smith S, De Stefano N, et al. Age-related changes in grey and white matter structure throughout adulthood. Neuroimage. 2010;51:943–51.
Suzuki Y, Kiyosawa M, Wakakura M, Mochizuki M, Ishii K. Gray matter density increase in the primary sensorimotor cortex in long-term essential blepharospasm. Neuroimage. 2011;56:1–7.
Hernowo AT, Boucard CC, Jansonius NM, Hooymans JM, Cornelissen FW. Automated morphometry of the visual pathway in primary open-angle glaucoma. Invest Ophthalmol Vis Sci. 2011;52:2758–66.
Kashiwagi K, Okubo T, Tsukahara S. Association of magnetic resonance imaging of anterior optic pathway with glaucomatous visual field damage and optic disc cupping. J Glaucoma. 2004;13:189–95.
Gupta N, Ang LC, Noël de Tilly L, Bidaisee L, Yücel YH. Human glaucoma and neural degeneration in intracranial optic nerve, lateral geniculate nucleus, and visual cortex. Br J Ophthalmol. 2006;90:674–8.
Boucard CC, Hernowo AT, Maguire RP, Jansonius NM, Roerdink JB, Hooymans JM, et al. Changes in cortical grey matter density associated with long-standing retinal visual field defects. Brain. 2009;132:1898–906.
Ito Y, Shimazawa M, Chen YN, Tsuruma K, Yamashima T, Araie M, et al. Morphological changes in the visual pathway induced by experimental glaucoma in Japanese monkeys. Exp Eye Res. 2009;89:246–55.
Woodward WR, Coull BM. Studies of effects of kainic acid lesions in the dorsal lateral geniculate nucleus of rat. J Comp Neurol. 1982;211:93–103.
Engelhorn T, Haider S, Michelson G, Doerfler A. A new semi-quantitative approach for analysing 3T diffusion tensor imaging of optic fibres and its clinical evaluation in glaucoma. Acad Radiol. 2010;17:1313–6.
Acknowledgments
This research was supported by Grants-in-Aid for Scientific Research (B) 21791725 (Y.S.) and (C) 20591038 (K.I.) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Murai, H., Suzuki, Y., Kiyosawa, M. et al. Positive correlation between the degree of visual field defect and optic radiation damage in glaucoma patients. Jpn J Ophthalmol 57, 257–262 (2013). https://doi.org/10.1007/s10384-013-0233-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-013-0233-0